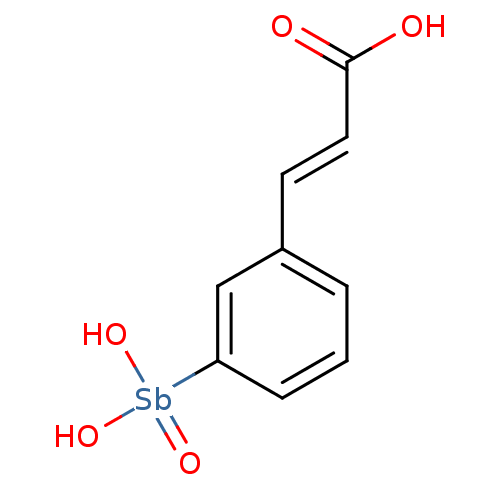
BDBM26613 (2E)-3-{3-[dihydroxy(oxo)--stibanyl]phenyl}prop-2-enoic acid::Compound 13778
SMILES OC(=O)\C=C\c1cccc(c1)[Sb](O)(O)=O
InChI Key InChIKey=ZTQRMGDLUGQTTF-MNPOOLNOSA-L
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 13 hits for monomerid = 26613
Found 13 hits for monomerid = 26613
Affinity DataKi: 72nMAssay Description:Inhibition of N-terminal His-tagged Cdc25a catalytic domain expressed in Escherichia coli BL21(DE3) using 3-O-Methylfluorescein phosphate as substrat...More data for this Ligand-Target Pair
Affinity DataKi: 369nMAssay Description:Inhibition of N-terminal His-tagged Cdc25b catalytic domain expressed in Escherichia coli BL21(DE3) using 3-O-Methylfluorescein phosphate as substrat...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human recombinant His-tagged VHR using 3-O-Methylfluorescein phosphate as substrate incubated for 10 mins prior to substrate addition b...More data for this Ligand-Target Pair
TargetReceptor-type tyrosine-protein phosphatase beta(Homo sapiens (Human))
Imperial College
Curated by ChEMBL
Imperial College
Curated by ChEMBL
Affinity DataIC50: 770nMAssay Description:Inhibition of PTP-beta receptor using 3-O-Methylfluorescein phosphate as substrate incubated for 10 mins prior to substrate addition by fluorometryMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of MKP3 using 3-O-Methylfluorescein phosphate as substrate incubated for 10 mins prior to substrate addition by fluorometryMore data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 1(Homo sapiens (Human))
Imperial College
Curated by ChEMBL
Imperial College
Curated by ChEMBL
Affinity DataIC50: 7.93E+3nMAssay Description:Inhibition of PTP1B using 3-O-Methylfluorescein phosphate as substrate incubated for 10 mins prior to substrate addition by fluorometryMore data for this Ligand-Target Pair
TargetPhosphatidylglycerophosphatase and protein-tyrosine phosphatase 1(Homo sapiens (Human))
Imperial College
Curated by ChEMBL
Imperial College
Curated by ChEMBL
Affinity DataIC50: 1.62E+3nMAssay Description:Inhibition of PTPMT1 using 3-O-Methylfluorescein phosphate as substrate incubated for 10 mins prior to substrate addition by fluorometryMore data for this Ligand-Target Pair
TargetReceptor-type tyrosine-protein phosphatase beta(Homo sapiens (Human))
Imperial College
Curated by ChEMBL
Imperial College
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibition of PTP-beta receptor using para-nitrophenol as substrate by colorimetric analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 110nMAssay Description:Inhibition of N-terminal His-tagged Cdc25a catalytic domain expressed in Escherichia coli BL21(DE3) using 3-O-Methylfluorescein phosphate as substrat...More data for this Ligand-Target Pair
Affinity DataIC50: 600nMAssay Description:Inhibition of N-terminal His-tagged Cdc25b catalytic domain expressed in Escherichia coli BL21(DE3) using 3-O-Methylfluorescein phosphate as substrat...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of N-terminal His-tagged Cdc25c catalytic domain expressed in Escherichia coli BL21(DE3) using 3-O-Methylfluorescein phosphate as substrat...More data for this Ligand-Target Pair
TargetDNA-(apurinic or apyrimidinic site) endonuclease(Homo sapiens (Human))
The Johns Hopkins University
The Johns Hopkins University
Affinity DataIC50: 200nMpH: 7.5 T: 2°CAssay Description:The screening assay was performed using 384-well microtiter plate first spotted with test compounds. Then Ape1 in reaction buffer was added to each w...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 3,4,5-trisphosphate 3-phosphatase and dual-specificity protein phosphatase PTEN(Homo sapiens (Human))
Imperial College
Curated by ChEMBL
Imperial College
Curated by ChEMBL
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of human PTEN using 3-O-Methylfluorescein phosphate as substrate incubated for 10 mins prior to substrate addition by fluorometryMore data for this Ligand-Target Pair