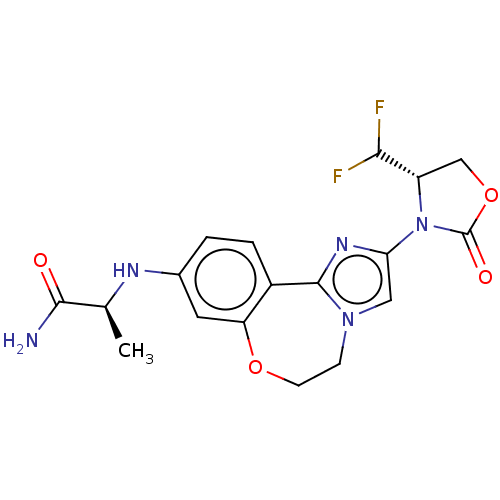
BDBM295665 (S)-2-((2-((S)-4-(difluoromethyl)- 2-oxooxazolidin-3-yl)-5,6- dihydrobenzo[f]imidazo[1,2- d][1,4]oxazepin-9- yl)amino)propanamide::US10112932, Compound 101::US10851091, Compound 101
SMILES C[C@H](Nc1ccc2-c3nc(cn3CCOc2c1)N1[C@@H](COC1=O)C(F)F)C(N)=O
InChI Key InChIKey=SGEUNORSOZVTOL-CABZTGNLSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 295665
Found 4 hits for monomerid = 295665
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Genentech
US Patent
Genentech
US Patent
Affinity DataKi: 0.0340nMAssay Description:The biochemical inhibition of four PI3K isoforms by the Formula I compounds of Table 1. In addition, two clinically tested PI3K compounds, taselisib ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Genentech
US Patent
Genentech
US Patent
Affinity DataKi: 12.2nMAssay Description:The biochemical inhibition of four PI3K isoforms by the Formula I compounds of Table 1. In addition, two clinically tested PI3K compounds, taselisib ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Homo sapiens (Human))
Genentech
US Patent
Genentech
US Patent
Affinity DataKi: 18.2nMAssay Description:The biochemical inhibition of four PI3K isoforms by the Formula I compounds of Table 1. In addition, two clinically tested PI3K compounds, taselisib ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform(Homo sapiens (Human))
Genentech
US Patent
Genentech
US Patent
Affinity DataKi: 99.7nMAssay Description:The biochemical inhibition of four PI3K isoforms by the Formula I compounds of Table 1. In addition, two clinically tested PI3K compounds, taselisib ...More data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)