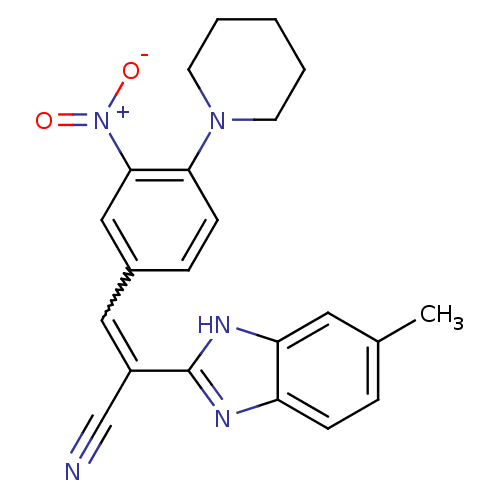
BDBM33459 (Z)-2-(6-methyl-1H-benzimidazol-2-yl)-3-(3-nitro-4-piperidin-1-yl-phenyl)prop-2-enenitrile::(Z)-2-(6-methyl-1H-benzimidazol-2-yl)-3-(3-nitro-4-piperidin-1-ylphenyl)prop-2-enenitrile::(Z)-2-(6-methyl-1H-benzimidazol-2-yl)-3-(3-nitro-4-piperidino-phenyl)acrylonitrile::(Z)-2-(6-methyl-1H-benzimidazol-2-yl)-3-[3-nitro-4-(1-piperidinyl)phenyl]-2-propenenitrile::2-(5-methyl-1H-benzimidazol-2-yl)-3-[3-nitro-4-(1-piperidinyl)phenyl]acrylonitrile::MLS000533042::SMR000140480::cid_5340140
SMILES Cc1ccc2nc([nH]c2c1)C(=Cc1ccc(N2CCCCC2)c(c1)[N+]([O-])=O)C#N
InChI Key InChIKey=WLPODNUCECUFHZ-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 33459
Found 3 hits for monomerid = 33459
Affinity DataIC50: 1.02E+3nMpH: 7.4 T: 2°CAssay Description:HTS was performed on a total of 33,068 compounds of the MLSCN library, 23,017 of which were not in the mixture HTS plates used previously. These comp...More data for this Ligand-Target Pair
TargetCarboxy-terminal domain RNA polymerase II polypeptide A small phosphatase 1(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 2.45E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
Affinity DataIC50: 4.23E+3nMAssay Description:NIH Molecular Libraries Screening Centers Network [MLSCN] Emory Chemical Biology Discovery Center in MLSCN Assay provider: John A. Katzenellenbogen, ...More data for this Ligand-Target Pair