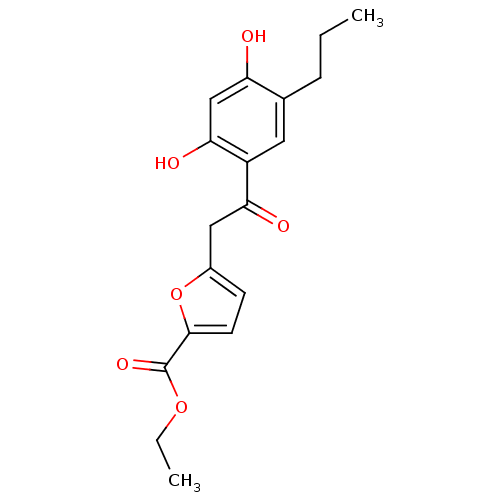
BDBM39605 5-[2-(2,4-dihydroxy-5-propyl-phenyl)-2-keto-ethyl]furan-2-carboxylic acid ethyl ester::5-[2-(2,4-dihydroxy-5-propylphenyl)-2-oxoethyl]-2-furancarboxylic acid ethyl ester::MLS000097854::SMR000059974::cid_765919::ethyl 5-[2-(2,4-dihydroxy-5-propylphenyl)-2-oxoethyl]furan-2-carboxylate::ethyl 5-[2-[2,4-bis(oxidanyl)-5-propyl-phenyl]-2-oxidanylidene-ethyl]furan-2-carboxylate
SMILES CCCc1cc(C(=O)Cc2ccc(o2)C(=O)OCC)c(O)cc1O
InChI Key InChIKey=QTTFMEHZDLTCDR-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 39605
Found 3 hits for monomerid = 39605
TargetAlkaline phosphatase, germ cell type(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 9.99E+5nMAssay Description:Sanford-Burnham Center for Chemical Genomics (SBCCG) Sanford-Burnham Medical Research Institute (SBMRI, San Diego, CA) NIH Molecular Libraries Screen...More data for this Ligand-Target Pair
Affinity DataKd: 2.00E+3nMAssay Description:Binding affinity to 15N-labeled human HSP90alpha isoform 1 N-terminal ATP binding domain (9 to 236 residues) expressed in Escherichia coli BL21(DE3) ...More data for this Ligand-Target Pair
TargetAlkaline phosphatase, germ cell type(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataEC50: 9.99E+5nMAssay Description:Sanford-Burnham Center for Chemical Genomics (SBCCG) Sanford-Burnham Medical Research Institute (SBMRI, San Diego, CA) NIH Molecular Libraries Screen...More data for this Ligand-Target Pair