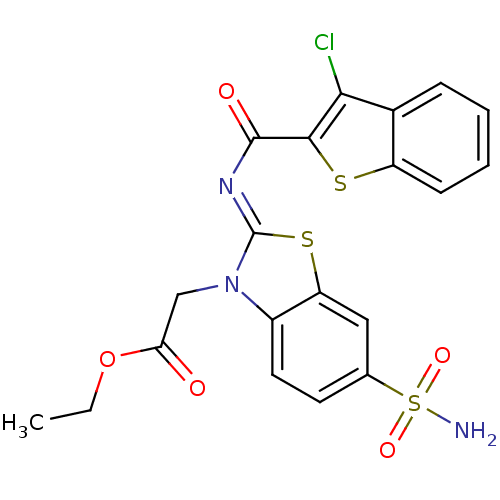
BDBM43091 2-[2-(3-chlorobenzothiophene-2-carbonyl)imino-6-sulfamoyl-1,3-benzothiazol-3-yl]acetic acid ethyl ester::2-[2-[(3-chloro-1-benzothiophen-2-yl)-oxomethyl]imino-6-sulfamoyl-1,3-benzothiazol-3-yl]acetic acid ethyl ester::MLS000697082::SMR000237894::cid_4625720::ethyl 2-[2-(3-chloro-1-benzothiophene-2-carbonyl)imino-6-sulfamoyl-1,3-benzothiazol-3-yl]acetate::ethyl 2-[2-[(3-chloranyl-1-benzothiophen-2-yl)carbonylimino]-6-sulfamoyl-1,3-benzothiazol-3-yl]ethanoate
SMILES CCOC(=O)Cn1c2ccc(cc2s\c1=N/C(=O)c1sc2ccccc2c1Cl)S(N)(=O)=O
InChI Key InChIKey=PFUIZHAZQWAOBU-ATJXCDBQSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 9 hits for monomerid = 43091
Found 9 hits for monomerid = 43091
TargetNuclear receptor coactivator 2(Homo sapiens (Human))
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
Affinity DataEC50: 7.96E+3nMAssay Description:Source (MLSCN Center Name): The Scripps Research Institute Molecular Screening Center Center Affiliation: The Scripps Research Institute (TSRI) Assay...More data for this Ligand-Target Pair
TargetNuclear receptor coactivator 1(Homo sapiens (Human))
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
Affinity DataEC50: 1.13E+4nMAssay Description:Source (MLSCN Center Name): The Scripps Research Institute Molecular Screening Center Center Affiliation: The Scripps Research Institute (TSRI) Assay...More data for this Ligand-Target Pair
TargetNuclear receptor coactivator 3(Homo sapiens (Human))
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
Affinity DataEC50: 1.43E+4nMAssay Description:Source (MLSCN Center Name): The Scripps Research Institute Molecular Screening Center http://molscreen.florida.scripps.edu/ Center Affiliation: The ...More data for this Ligand-Target Pair
TargetSUMO-conjugating enzyme UBC9(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 1.18E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 22(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
TargetCarboxy-terminal domain RNA polymerase II polypeptide A small phosphatase 1(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 2.06E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetLow molecular weight phosphotyrosine protein phosphatase(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
TargetLow molecular weight phosphotyrosine protein phosphatase(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
TargetSUMO-activating enzyme subunit 1(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 4.97E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair