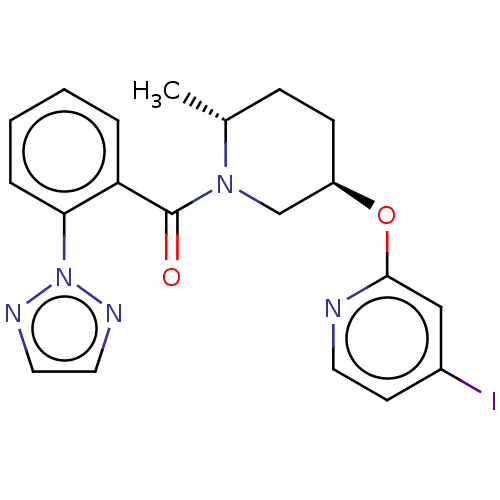
BDBM50084396 CHEMBL3426143
SMILES C[C@@H]1CC[C@H](CN1C(=O)c1ccccc1-n1nccn1)Oc1cc(I)ccn1
InChI Key InChIKey=DGGVZEGNYMJZDB-GDBMZVCRSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50084396
Found 4 hits for monomerid = 50084396
Affinity DataKi: 0.400nMAssay Description:Displacement of [3H]-(S)-N-(2-(1H-pyrrol-1-yl)phenyl)-1-(2-(1-methyl-1H-benzo[d]imidazol-2-ylthio)acetyl)pyrrolidine-2-carboxamide from human OX2R ex...More data for this Ligand-Target Pair
TargetOrexin/Hypocretin receptor type 1(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataKi: 59nMAssay Description:Displacement of [3H]-(S)-N-(biphenyl-2-yl)-1-(2-(1-methyl-1H-benzo[d]imidazol-2-ylthio)acetyl)pyrrolidine-2-carboxamide from human OX1R expressed in ...More data for this Ligand-Target Pair
Affinity DataIC50: 7.20nMAssay Description:Antagonist activity against human OX2R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intercellular Ca2+ mobilization by FLIPR a...More data for this Ligand-Target Pair
TargetOrexin/Hypocretin receptor type 1(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 410nMAssay Description:Antagonist activity against human OX1R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intercellular Ca2+ mobilization by FLIPR a...More data for this Ligand-Target Pair