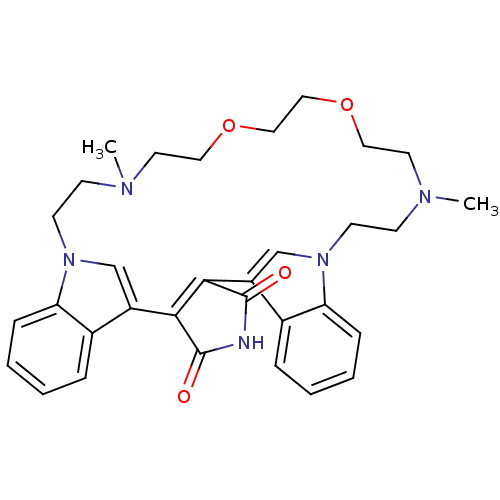
BDBM50132316 17,26-dimethyl-20,23-dioxa-4,14,17,26,29-pentaazahexacyclo[27.6.1.17,14.02,6.08,13.030,35]heptatriaconta-1(36),2(6),7(37),8(13),9,11,30(35),31,33-nonaene-3,5-dione::CHEMBL105620
SMILES CN1CCOCCOCCN(C)CCn2cc(C3=C(C(=O)NC3=O)c3cn(CC1)c1ccccc31)c1ccccc21
InChI Key InChIKey=BEYBHILIDYTJTL-UHFFFAOYSA-N
Data 4 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50132316
Found 4 hits for monomerid = 50132316
TargetProtein kinase C alpha type(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 770nMAssay Description:Inhibition of Protein kinase C alphaMore data for this Ligand-Target Pair
TargetProtein kinase C beta type(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 140nMAssay Description:Inhibition of Protein kinase C beta 2More data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 8nMAssay Description:Inhibition of Glycogen synthase kinase-3 betaMore data for this Ligand-Target Pair
TargetProtein kinase C beta type(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 75nMAssay Description:Inhibition of Protein kinase C beta 2More data for this Ligand-Target Pair