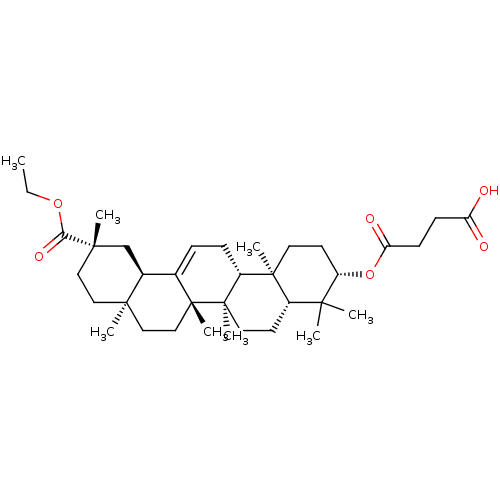
BDBM50154561 CHEMBL3774603
SMILES [H][C@@]12C[C@](C)(CC[C@]1(C)CC[C@]1(C)C2=CC[C@]2([H])[C@@]3(C)CC[C@H](OC(=O)CCC(O)=O)C(C)(C)[C@]3([H])CC[C@@]12C)C(=O)OCC
InChI Key InChIKey=UJCCELSWWRMAPH-AYVZPHPZSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50154561
Found 3 hits for monomerid = 50154561
Affinity DataKi: 42nMAssay Description:Competitive inhibition of CE2 in human liver microsomes using fluorescein diacetate as substrate preincubated for 10 mins followed by substrate addit...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:Inhibition of CE2 in human liver microsomes using fluorescein diacetate as substrate preincubated for 10 mins followed by substrate addition measured...More data for this Ligand-Target Pair
TargetLiver carboxylesterase 1(Homo sapiens (Human))
Dalian Institute Of Chemical Physics
Curated by ChEMBL
Dalian Institute Of Chemical Physics
Curated by ChEMBL
Affinity DataIC50: 2.04E+4nMAssay Description:Inhibition of CE1 in human liver microsomes using 2-(2-Benzoyl-3-methoxyphenyl) benzothiazole as substrate preincubated for 10 mins followed by subst...More data for this Ligand-Target Pair