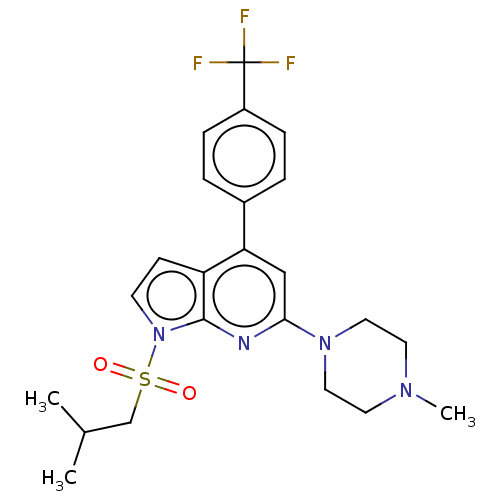
BDBM50166852 CHEMBL3797717
SMILES CC(C)CS(=O)(=O)n1ccc2c(cc(nc12)N1CCN(C)CC1)-c1ccc(cc1)C(F)(F)F
InChI Key InChIKey=RYBRXOLQVVUVSC-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50166852
Found 5 hits for monomerid = 50166852
Affinity DataKi: 4nMAssay Description:Displacement of [3H]LSD from human 5HT6 receptor in HEK293 cell membrane incubated for 1 hr by scintillation counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 367nMAssay Description:Displacement of [3H]NMSP from human dopamine D2S receptor expressed in CHO cell membrane incubated for 1 hr by scintillation counting methodMore data for this Ligand-Target Pair
Affinity DataIC50: 3.30E+3nMAssay Description:Inhibition of human recombinant CYP2D6 using AMMC as substrate incubated for 45 mins by fluorescence analysis in presence of NADPHMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Selvita
Curated by ChEMBL
Selvita
Curated by ChEMBL
Affinity DataIC50: 2.10E+3nMAssay Description:Inhibition of human ERG1 expressed in CHO cells by whole cell patch clamp electrophysiology methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.10E+3nMAssay Description:Inhibition of human recombinant CYP3A4 using 7-benzyloxy-trifluoromethylcoumarin as substrate incubated for 30 mins by fluorescence analysis in prese...More data for this Ligand-Target Pair