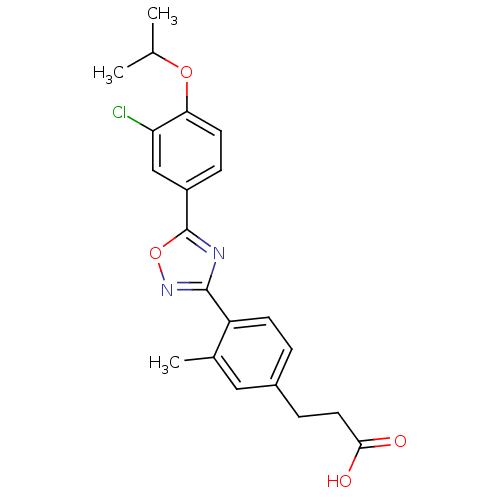
BDBM50186405 3-(4-(5-(3-chloro-4-isopropoxyphenyl)-1,2,4-oxadiazol-3-yl)-3-methylphenyl)propanoic acid::CHEMBL206940
SMILES CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1ccc(CCC(O)=O)cc1C
InChI Key InChIKey=AOXVYFLXNZEUPI-UHFFFAOYSA-N
Data 8 EC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 8 hits for monomerid = 50186405
Found 8 hits for monomerid = 50186405
Affinity DataEC50: 73nMAssay Description:Agonist activity at S1P5 receptor assessed as induction of [35S]GTPgammaS bindingMore data for this Ligand-Target Pair
Affinity DataEC50: 940nMAssay Description:Agonist activity at S1P3 receptor assessed as induction of [35S]GTPgammaS bindingMore data for this Ligand-Target Pair
Affinity DataEC50: 0.150nMAssay Description:Agonist activity at S1P1 receptor assessed as induction of [35S]GTPgammaS bindingMore data for this Ligand-Target Pair
TargetSphingosine 1-phosphate receptor 2(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: >1.00E+4nMAssay Description:Activity at S1P2 receptor expressed in CHO cells measured as S1P-induced [35S]GTPgammaS uptakeMore data for this Ligand-Target Pair
Affinity DataEC50: 1.50E+3nMAssay Description:Agonist activity at S1P3 receptor expressed in CHO cells measured as S1P-induced [35S]GTPgammaS uptakeMore data for this Ligand-Target Pair
TargetSphingosine 1-phosphate receptor 4(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: >1.00E+4nMAssay Description:Activity at S1P4 receptor expressed in CHO cells measured as S1P-induced [35S]GTPgammaS uptakeMore data for this Ligand-Target Pair
Affinity DataEC50: 140nMAssay Description:Agonist activity at S1P5 receptor expressed in CHO cells measured as S1P-induced [35S]GTPgammaS uptakeMore data for this Ligand-Target Pair
Affinity DataEC50: 0.200nMAssay Description:Agonist activity at S1P1 receptor expressed in CHO cells measured as S1P-induced [35S]GTPgammaS uptakeMore data for this Ligand-Target Pair