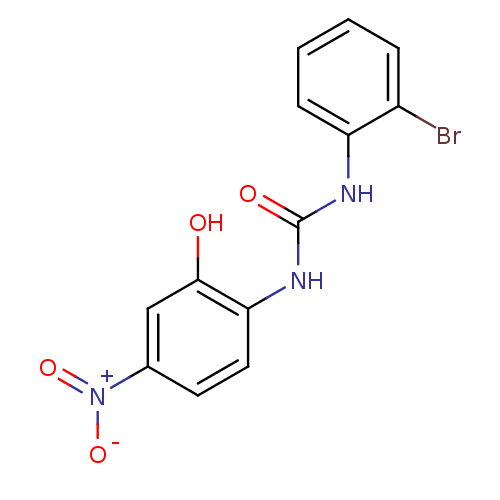
BDBM50203012 1-(2-bromophenyl)-3-(2-hydroxy-4-nitrophenyl)urea::CHEMBL239767::SB-225002
SMILES Oc1cc(ccc1NC(=O)Nc1ccccc1Br)[N+]([O-])=O
InChI Key InChIKey=MQBZVUNNWUIPMK-UHFFFAOYSA-N
Data 7 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 7 hits for monomerid = 50203012
Found 7 hits for monomerid = 50203012
Affinity DataIC50: 40nMAssay Description:Antagonist activity at human CXCR2 expressed in HEK293 cells assessed as inhibition of CXCL8-induced intracellular Ca2+ release by fluorescence based...More data for this Ligand-Target Pair
Affinity DataIC50: 30nMAssay Description:Antagonist activity at CXCR2 in human PMNs assessed as inhibition of CXCL1-induced intracellular Ca2+ release by fluorescence based calcium flux assa...More data for this Ligand-Target Pair
Affinity DataIC50: 22nMAssay Description:Displacement of [125I]IL8 from human recombinant CXCR2 expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 198nMAssay Description:Antagonist activity against CXCR2 (unknown origin) by calcium flux assayMore data for this Ligand-Target Pair
TargetC-X-C chemokine receptor type 1(Homo sapiens (Human))
National Heart And Lung Institute
Curated by ChEMBL
National Heart And Lung Institute
Curated by ChEMBL
Affinity DataIC50: >3.00E+3nMAssay Description:Binding affinity to CXCR1More data for this Ligand-Target Pair
TargetC-X-C chemokine receptor type 1(Homo sapiens (Human))
National Heart And Lung Institute
Curated by ChEMBL
National Heart And Lung Institute
Curated by ChEMBL
Affinity DataIC50: 22nMAssay Description:Antagonist activity at CXCR1 assessed as inhibition of CXCL8 binding by cell based assayMore data for this Ligand-Target Pair
Affinity DataIC50: 22nMAssay Description:Binding affinity to CXCR2More data for this Ligand-Target Pair