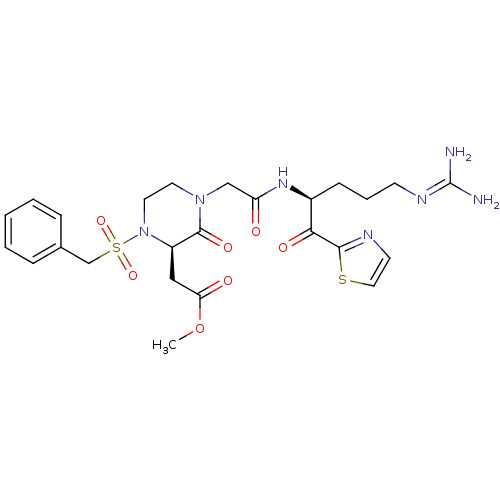
BDBM50228756 ((R)-4-{[(S)-4-Guanidino-1-(thiazole-2-carbonyl)-butylcarbamoyl]-methyl}-3-oxo-1-phenylmethanesulfonyl-piperazin-2-yl)-acetic acid methyl ester::CHEMBL272135::methyl 2-((R)-1-(benzylsulfonyl)-4-(2-((S)-5-guanidino-1-oxo-1-(thiazol-2-yl)pentan-2-ylamino)-2-oxoethyl)-3-oxopiperazin-2-yl)acetate
SMILES [#6]-[#8]-[#6](=O)-[#6]-[#6@H]-1-[#7](-[#6]-[#6]-[#7](-[#6]-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-c2nccs2)-[#6]-1=O)S(=O)(=O)[#6]-c1ccccc1
InChI Key InChIKey=OVBHVFLKMRIMFI-RBUKOAKNSA-N
Data 4 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50228756
Found 4 hits for monomerid = 50228756
Affinity DataIC50: 5.00E+3nMAssay Description:In vitro inhibitory activity against serine protease thrombinMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 8nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of human alpha-thrombinMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 8nMAssay Description:In vitro inhibitory activity against factor XaMore data for this Ligand-Target Pair