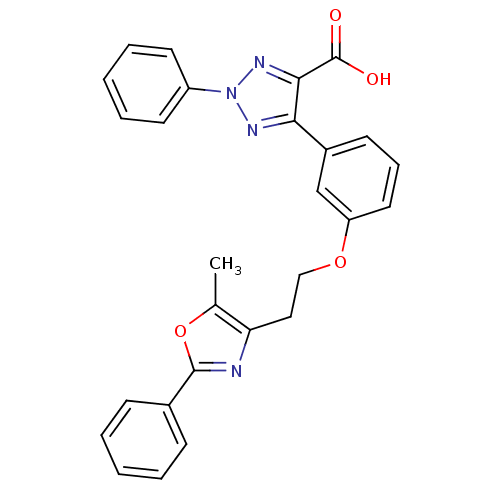
BDBM50248346 5-(3-(2-(5-methyl-2-phenyloxazol-4-yl)ethoxy)phenyl)-2-phenyl-2H-1,2,3-triazole-4-carboxylic acid::CHEMBL474551
SMILES Cc1oc(nc1CCOc1cccc(c1)-c1nn(nc1C(O)=O)-c1ccccc1)-c1ccccc1
InChI Key InChIKey=UBPSAUWQNXEZQY-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50248346
Found 4 hits for monomerid = 50248346
TargetPeroxisome proliferator-activated receptor alpha(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataEC50: 4.02E+3nMAssay Description:Agonist activity at human PPARalpha ligand binding domain expressed in HEK293 cells by Gal4 transactivation assayMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataEC50: 2.19E+3nMAssay Description:Agonist activity at human PPARgamma ligand binding domain expressed in HEK293 cells by Gal4 transactivation assayMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 670nMAssay Description:Binding affinity to PPARgamma (unknown origin) by fluorescence polarization assayMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor alpha(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 1.61E+3nMAssay Description:Binding affinity to PPARalpha (unknown origin) by fluorescence polarization assayMore data for this Ligand-Target Pair