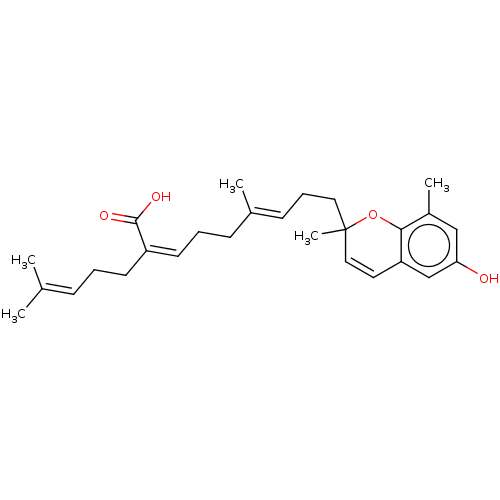
BDBM50267968 CHEMBL4085945
SMILES [#6]\[#6](-[#6])=[#6]/[#6]-[#6]\[#6](=[#6]\[#6]-[#6]\[#6](-[#6])=[#6]\[#6]-[#6]C1([#6])[#8]-c2c(-[#6])cc(-[#8])cc2-[#6]=[#6]1)-[#6](-[#8])=O
InChI Key InChIKey=QKXAGRZCXAYBQX-WDZAAMDOSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50267968
Found 4 hits for monomerid = 50267968
Affinity DataKi: 2.90E+3nMAssay Description:Noncompetitive inhibition of recombinant human BACE1 using Rh-EVNLDAEFK-Quencher as substrate after 60 mins by Dixon plot analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 9.40E+3nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine chloride as substrate incubated for 15 mins by spectrophotometric analysisMore data for this Ligand-Target Pair
TargetAcetylcholinesterase(Electrophorus electricus (Electric eel))
Pukyong National University
Curated by ChEMBL
Pukyong National University
Curated by ChEMBL
Affinity DataIC50: 9.73E+4nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine iodide as substrate incubated for 15 mins by spectrophotometric analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 7.00E+3nMAssay Description:Inhibition of recombinant human BACE1 using Rh-EVNLDAEFK-Quencher as substrate after 60 mins by FRET assayMore data for this Ligand-Target Pair