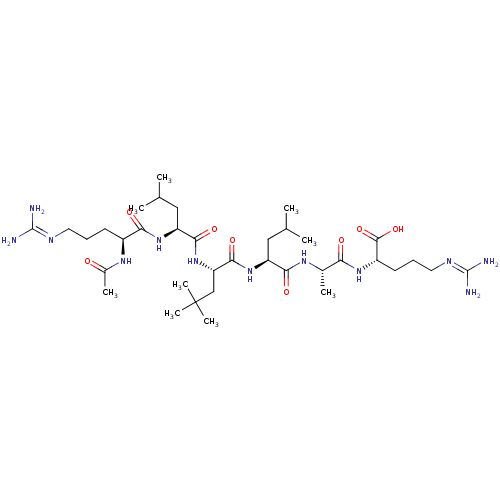
BDBM50314023 (6S,9S,12S,15S,18S,21S)-21-acetamido-1,26-diamino-1,26-diimino-12,18-diisobutyl-9-methyl-15-neopentyl-8,11,14,17,20-pentaoxo-2,7,10,13,16,19,25-heptaazahexacosane-6-carboxylic acid::CHEMBL1089597
SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]C([#6])([#6])[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](-[#8])=O
InChI Key InChIKey=VIJSPAIQWVPKQZ-BLECARSGSA-N
Data 2 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50314023
Found 2 hits for monomerid = 50314023
Affinity DataIC50: 250nMpH: 9.5Assay Description:Inhibition of human complement factor B treated for 5 mins before addition of substrate Ac-SHLGLAR-pNA at pH 9.5 by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMpH: 7.4Assay Description:Inhibition of human C3 convertase assessed as inhibition of C3a and C3b fragment production at pH 7.4 after 30 mins by SDS-PAGEMore data for this Ligand-Target Pair