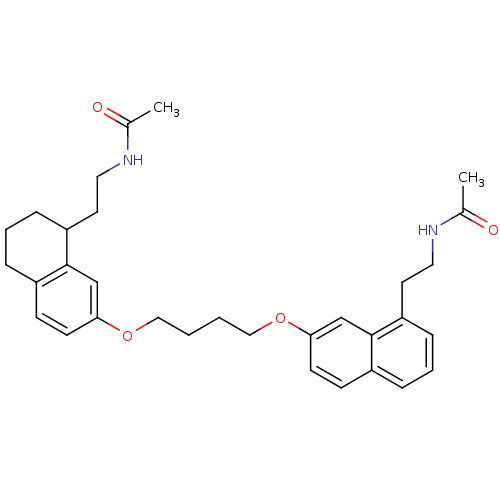
BDBM50316693 CHEMBL1095831::N-[2-(7-{4-[8-(2-Acetylaminoethyl)-5,6,7,8-tetrahydronaphthalen-2-yloxy]butoxy}naphthalen-1-yl)ethyl]acetamide
SMILES CC(=O)NCCC1CCCc2ccc(OCCCCOc3ccc4cccc(CCNC(C)=O)c4c3)cc12
InChI Key InChIKey=DNGCHGMDDOPAPX-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50316693
Found 4 hits for monomerid = 50316693
Affinity DataKi: 0.260nMAssay Description:Displacement of 2-[125I]iodomelatonin from human MT1 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 6.80nMAssay Description:Displacement of 2-[125I]iodomelatonin from human MT2 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Agonist activity at human human MT2 receptor expressed in CHO cells by [35S]GTPgamma binding assayMore data for this Ligand-Target Pair
Affinity DataEC50: 7.80nMAssay Description:Agonist activity at human human MT1 receptor expressed in CHO cells by [35S]GTPgamma binding assayMore data for this Ligand-Target Pair