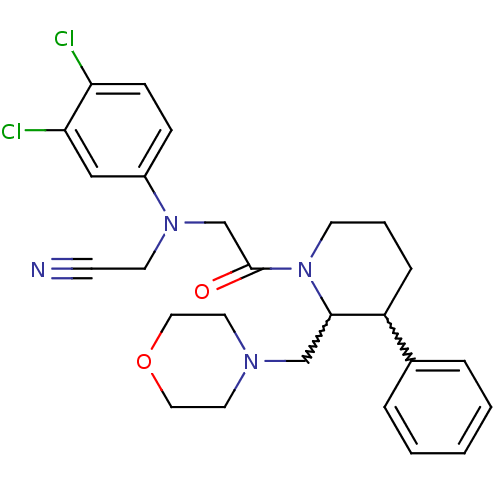
BDBM50377223 CHEMBL258251
SMILES Clc1ccc(cc1Cl)N(CC#N)CC(=O)N1CCCC(C1CN1CCOCC1)c1ccccc1
InChI Key InChIKey=BFPVIFSFUVSFRI-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50377223
Found 4 hits for monomerid = 50377223
Affinity DataKi: 6nMAssay Description:Displacement of [125I]hU-2 from human recombinant Urotensin 2 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataEC50: 5.00E+3nMAssay Description:Agonist activity at kappa opioid receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 3.70E+3nMAssay Description:Inhibition of CYP2D6More data for this Ligand-Target Pair