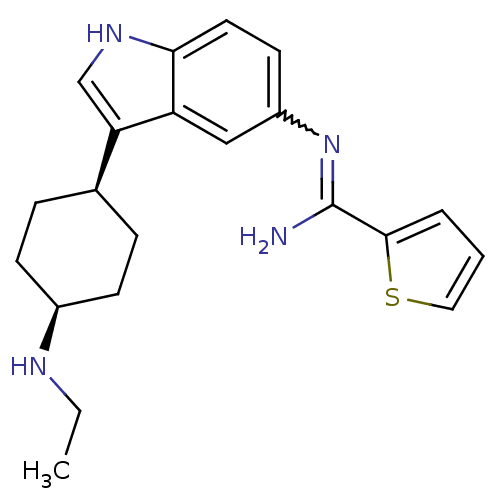
BDBM50384898 CHEMBL2036270
SMILES CCN[C@H]1CC[C@H](CC1)c1c[nH]c2ccc(cc12)N=C(N)c1cccs1
InChI Key InChIKey=WKNJJZDFDCGVSZ-GASCZTMLSA-N
Data 10 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 10 hits for monomerid = 50384898
Found 10 hits for monomerid = 50384898
Affinity DataIC50: 4.93E+5nMAssay Description:Inhibition of human iNOS expressed in baculovirus-infected insect sf9 cells assessed as conversion of [3H]-L-arginine to [3H]-L-citrulline preincubat...More data for this Ligand-Target Pair
Affinity DataIC50: 380nMAssay Description:Displacement of [3H]nisoxetine from human NET expressed in CHO cells after 120 mins by scintillation countingMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Neuraxon
Curated by ChEMBL
Neuraxon
Curated by ChEMBL
Affinity DataIC50: 2.30E+4nMAssay Description:Displacement of [3H]astemizole from human Erg expressed in HEK293 cells after 75 minsMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of CYP1A2More data for this Ligand-Target Pair
Affinity DataIC50: 2.15E+4nMAssay Description:Inhibition of CYP2C9More data for this Ligand-Target Pair
Affinity DataIC50: 2.31E+4nMAssay Description:Inhibition of CYP3A4More data for this Ligand-Target Pair
Affinity DataIC50: 540nMAssay Description:Inhibition of human nNOS expressed in baculovirus-infected insect sf9 cells assessed as conversion of [3H]-L-arginine to [3H]-L-citrulline preincubat...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90E+4nMAssay Description:Inhibition of human eNOS expressed in baculovirus-infected insect sf9 cells assessed as conversion of [3H]-L-arginine to [3H]-L-citrulline preincubat...More data for this Ligand-Target Pair
Affinity DataIC50: 2.34E+4nMAssay Description:Inhibition of CYP2C19More data for this Ligand-Target Pair