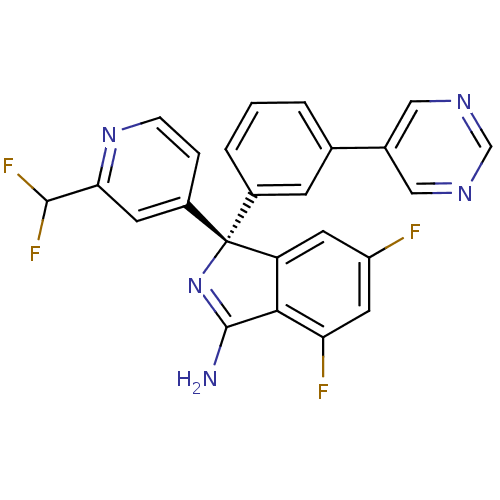
BDBM50398263 CHEMBL2177914
SMILES NC1=N[C@@](c2cc(F)cc(F)c12)(c1cccc(c1)-c1cncnc1)c1ccnc(c1)C(F)F
InChI Key InChIKey=XCGKBIOLPVAHCG-DEOSSOPVSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50398263
Found 5 hits for monomerid = 50398263
Affinity DataKi: 770nMAssay Description:Inhibition of human recombinant BACE2 using (europium)CEVNLDAEFK(Qsy7) as substrate incubated for 10 mins prior to substrate addition measured after ...More data for this Ligand-Target Pair
Affinity DataIC50: 8.60nMAssay Description:Inhibition of BACE1 in human SH-SY5Y cells assessed as inhibition of sAPPbeta release after 16 hrs by immunoassayMore data for this Ligand-Target Pair
Affinity DataIC50: 53nMAssay Description:Inhibition of BACE1-mediated amyloid beta 40 release in C57/BL6 mouse primary cortical neurons after overnight incubation by ELISAMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: 1.90E+4nMAssay Description:Inhibition of human ERG expressed in CHO cells by IonWorks assayMore data for this Ligand-Target Pair
Affinity DataIC50: 36nMAssay Description:Inhibition of human recombinant BACE1 using (europium)CEVNLDAEFK(Qsy7) as substrate incubated for 10 mins prior to substrate addition measured after ...More data for this Ligand-Target Pair