BDBM50448621 CHEMBL3127521
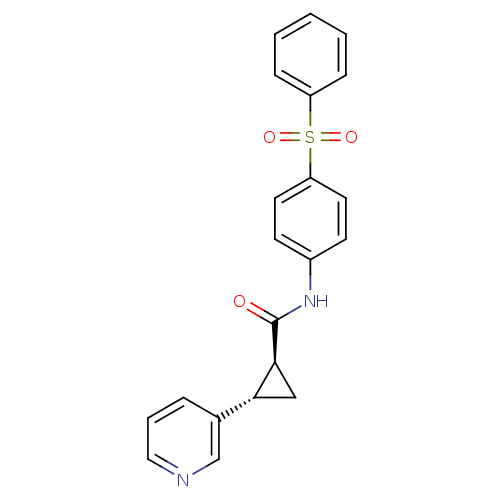
SMILES O=C(Nc1ccc(cc1)S(=O)(=O)c1ccccc1)[C@H]1C[C@@H]1c1cccnc1
InChI Key InChIKey=YERMBKYNILMCNC-UXHICEINSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50448621
Found 5 hits for monomerid = 50448621
Affinity DataIC50: 720nMAssay Description:Reversible inhibition of CYP2C9 in human liver microsomes using (S)- warfarin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Reversible inhibition of CYP2C19 in human liver microsomes using (S)-mephenytoin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Reversible inhibition of CYP1A2 in human liver microsomes using phenacetin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Reversible inhibition of CYP2D6 in human liver microsomes using dextromethorphan as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 3.90nMAssay Description:Inhibition of C-terminal His-tagged human full-length NAMPT expressed in Escherichia coli Rosetta DE3 using nicotinamide as substrate preincubated fo...More data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)