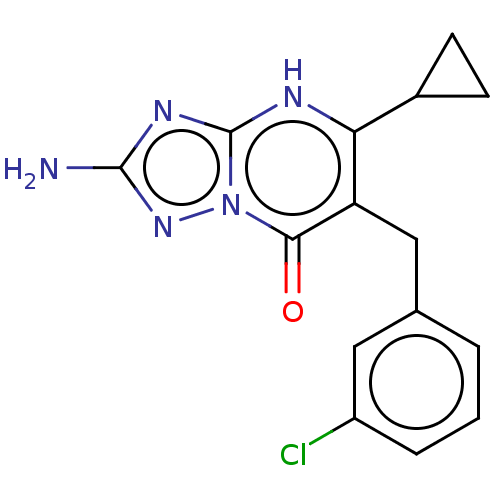
BDBM50506125 CHEMBL4517518
SMILES Nc1nc2[nH]c(C3CC3)c(Cc3cccc(Cl)c3)c(=O)n2n1
InChI Key InChIKey=YLVBQGLIQXQGPH-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50506125
Found 6 hits for monomerid = 50506125
Affinity DataKi: 1.30nMAssay Description:Displacement of [3H]-CCR2-RA-[R] from human CCR2 expressed in human U2OS cells incubated for 2 hrs by scintillation spectrometric methodMore data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:Displacement of [3H]-CCR2-RA-[R] from human CCR2 expressed in human U2OS cells incubated for 2 hrs by scintillation spectrometric methodMore data for this Ligand-Target Pair
Affinity DataIC50: 575nMAssay Description:Antagonist activity at human TEV cleavage site-linked CCR5 expressed in human U2OS cells harboring beta-lactamase reporter gene assessed as inhibitio...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:Antagonist activity at CCR2b in human THP1 cells assessed as inhibition of human MCP1 induced calcium flux by fluo-4 dye based fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Antagonist activity at human TEV cleavage site-linked CCR2b expressed in human U2OS cells harboring beta-lactamase reporter gene assessed as inhibiti...More data for this Ligand-Target Pair
Affinity DataIC50: 575nMAssay Description:Antagonist activity at human TEV cleavage site-linked CCR5 expressed in human U2OS cells harboring beta-lactamase reporter gene assessed as inhibitio...More data for this Ligand-Target Pair