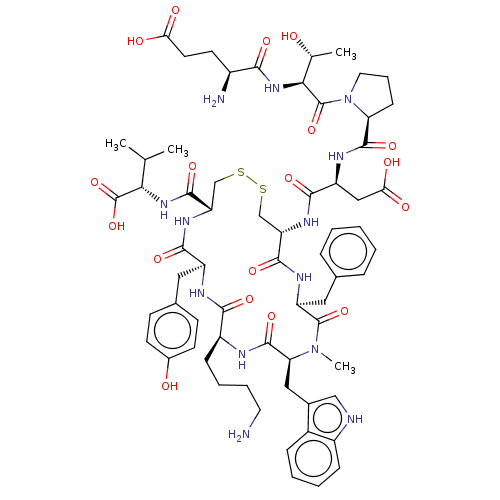
BDBM50517317 CHEMBL4475783
SMILES CC(C)[C@H](NC(=O)[C@@H]1CSSC[C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]2CCCN2C(=O)[C@@H](NC(=O)[C@@H](N)CCC(O)=O)[C@@H](C)O)C(=O)N[C@@H](Cc2ccccc2)C(=O)N(C)[C@@H](Cc2c[nH]c3ccccc23)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc2ccc(O)cc2)C(=O)N1)C(O)=O
InChI Key InChIKey=ACPSPBMVAMLTFE-JWNUYQQJSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50517317
Found 3 hits for monomerid = 50517317
Affinity DataKi: 20nMAssay Description:Displacement of [125I]-URP or human [125I[-urotensin-2 from human urotensin 2 receptor expressed in HEK293 or CHOK1 cells after 2 hrs by gamma-counti...More data for this Ligand-Target Pair
Affinity DataEC50: 5.60nMAssay Description:Agonist activity at urotensin 2 receptor (unknown origin) expressed in HEK293 cells co-expressing Gq-polycistronic BRET biosensor assessed as inducti...More data for this Ligand-Target Pair
Affinity DataEC50: 5.80nMAssay Description:Agonist activity at urotensin 2 receptor in Sprague-Dawley rat thoracic aortic ring assessed as induction of contraction relative to KCl-induced cont...More data for this Ligand-Target Pair