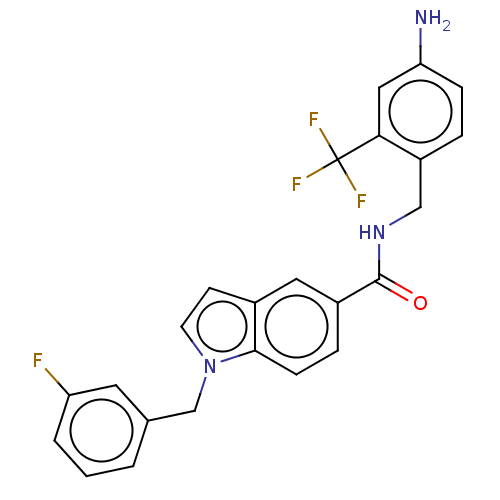
BDBM50586943 CHEMBL5087861
SMILES Nc1ccc(CNC(=O)c2ccc3n(Cc4cccc(F)c4)ccc3c2)c(c1)C(F)(F)F
InChI Key InChIKey=PVBXRSNIUUJOGQ-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50586943
Found 2 hits for monomerid = 50586943
TargetPeroxisome proliferator-activated receptor gamma(Homo sapiens (Human))
Goethe-University
Curated by ChEMBL
Goethe-University
Curated by ChEMBL
Affinity DataEC50: 1.68E+3nMAssay Description:Partial agonist activity at GAL4-tagged human PPARgamma LBD expressed in HEK293T cells incubated for 12 to 14 hrs by dual-Glo luciferase assayMore data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:Inhibition of recombinant human full length sEH assessed as reduction in 6-methoxy-2-naphthaldehyde formation using PHOME as substrate preincubated f...More data for this Ligand-Target Pair