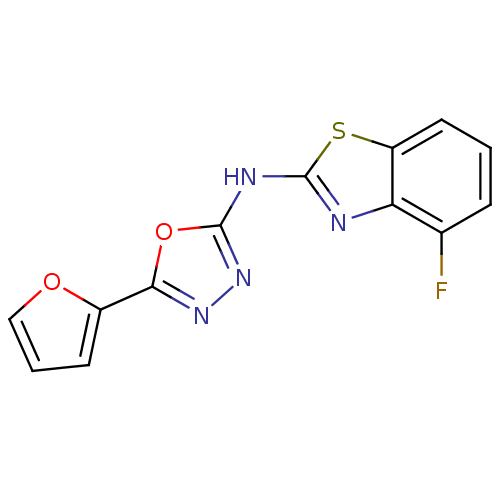
BDBM56999 (4-fluoro-1,3-benzothiazol-2-yl)-[5-(2-furyl)-1,3,4-oxadiazol-2-yl]amine::4-fluoro-N-[5-(2-furyl)-1,3,4-oxadiazol-2-yl]-1,3-benzothiazol-2-amine::MLS000045464::N-(4-fluoranyl-1,3-benzothiazol-2-yl)-5-(furan-2-yl)-1,3,4-oxadiazol-2-amine::N-(4-fluoro-1,3-benzothiazol-2-yl)-5-(2-furanyl)-1,3,4-oxadiazol-2-amine::N-(4-fluoro-1,3-benzothiazol-2-yl)-5-(furan-2-yl)-1,3,4-oxadiazol-2-amine::SMR000027223::cid_3245517
SMILES Fc1cccc2sc(Nc3nnc(o3)-c3ccco3)nc12
InChI Key InChIKey=GAMMRBPIJMZJKC-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 56999
Found 3 hits for monomerid = 56999
Affinity DataEC50: 1.50E+5nMAssay Description:Keywords: Group A streptococcus, GAS, streptokinase, expression, virulence, inhibition, dose response, EC50 Assay Overview: The goal of this assa...More data for this Ligand-Target Pair
TargetC-C chemokine receptor type 6(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 3.24E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute(SBMRI, San Diego, CA...More data for this Ligand-Target Pair
TargetBeta-galactosidase(Escherichia coli)
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: >6.66E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute(SBMRI, San Diego, C...More data for this Ligand-Target Pair