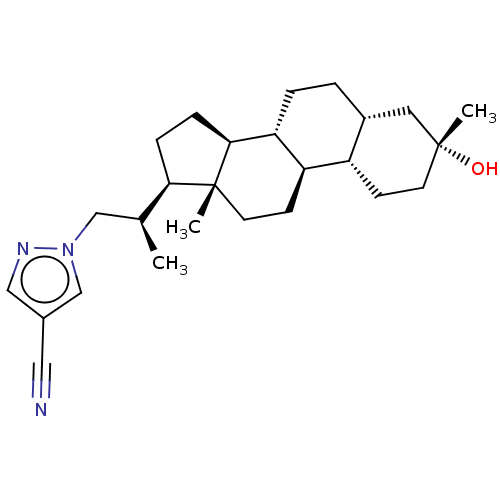
BDBM625663 Synthesis of 1-((R)-2-((3R,5R,8R,9R,10S,13S,14S,17R)-3-hydroxy-3,13-dimethylhexadecahydro-1H-cyclopenta[a]phenanthren-17-yl)propyl)-1H-pyrazole-4-carbonitrile (H7) & 1-((S)-2-((3R,5R,8R,9R,10S,13S,14S,17R)-3-hydroxy-3,13-dimethylhexadecahydro-1H-cyclopenta[a]phenanthren-17-yl)propyl)-1H-pyrazole-4-carbonitrile (H8)::US20230322846, Example 51
SMILES C[C@@H](Cn1cc(cn1)C#N)[C@H]1CC[C@H]2[C@@H]3CC[C@@H]4C[C@](C)(O)CC[C@@H]4[C@H]3CC[C@]12C
InChI Key InChIKey=DXOJYARVVZJJKH-CJNPGIHXSA-N
Data 1 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 1 hit for monomerid = 625663
Found 1 hit for monomerid = 625663
Affinity DataIC50: <100nMAssay Description:Briefly, cortices are rapidly removed following decapitation of carbon dioxide-anesthetized Sprague-Dawley rats (200-250 g). The cortices are homogen...More data for this Ligand-Target Pair