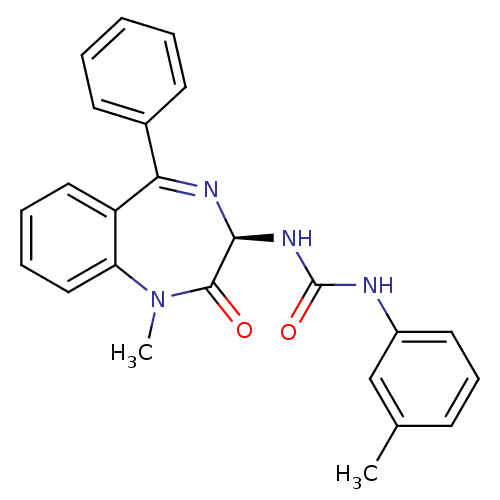
BDBM81962 S-L-365,260
SMILES CN1c2ccccc2C(=N[C@@H](NC(=O)Nc2cccc(C)c2)C1=O)c1ccccc1
InChI Key InChIKey=KDFQABSFVYLGPM-QFIPXVFZSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 81962
Found 4 hits for monomerid = 81962
Affinity DataKi: 0.148nMAssay Description:Inhibition by displacing [3H]CCK-8S against Cholecystokinin type A receptor of rat pancreatic membranesMore data for this Ligand-Target Pair
TargetGastrin/cholecystokinin type B receptor(Homo sapiens (Human))
Glaxo Wellcome Medicines Research Centre
Curated by ChEMBL
Glaxo Wellcome Medicines Research Centre
Curated by ChEMBL
Affinity DataKi: 1.60nMAssay Description:Inhibition by displacing [3H]CCK-8S against human Cholecystokinin type B receptorMore data for this Ligand-Target Pair
TargetGastrin/cholecystokinin type B receptor(Homo sapiens (Human))
Glaxo Wellcome Medicines Research Centre
Curated by ChEMBL
Glaxo Wellcome Medicines Research Centre
Curated by ChEMBL
Affinity DataKi: 3nMAssay Description:Inhibition by displacing [3H]CCK-8S against Cholecystokinin type B receptor of guinea pigMore data for this Ligand-Target Pair
TargetGastrin/cholecystokinin type B receptor(RAT)
Glaxo Wellcome Medicines Research Centre
Curated by ChEMBL
Glaxo Wellcome Medicines Research Centre
Curated by ChEMBL
Affinity DataKi: 269nMAssay Description:Inhibition by displacing [3H]CCK-8S against Cholecystokinin type B receptor of rat pancreatic membranesMore data for this Ligand-Target Pair