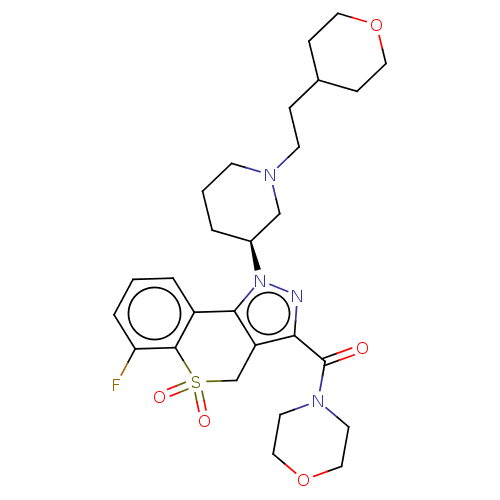
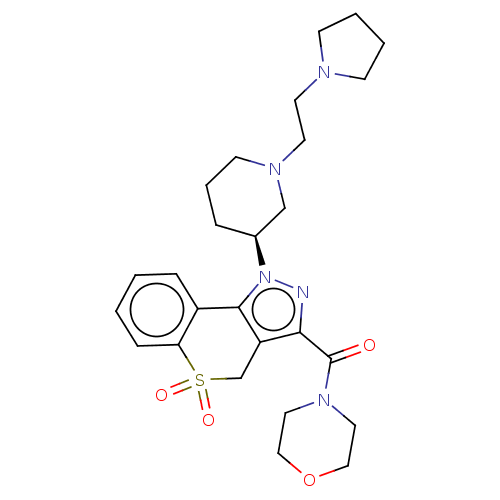
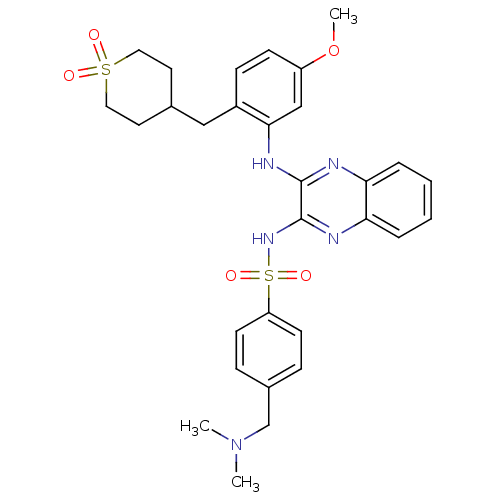
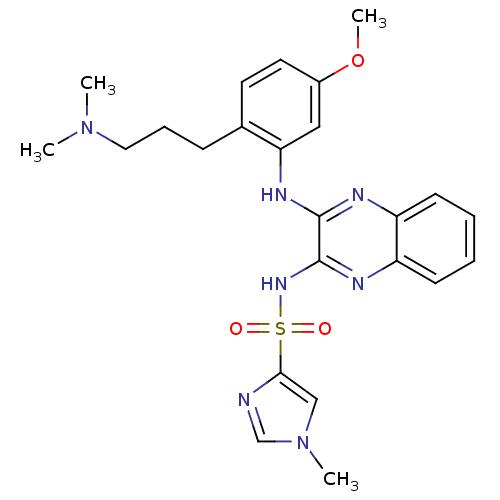
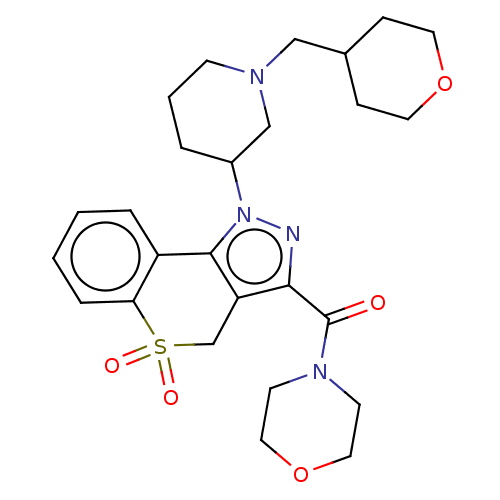
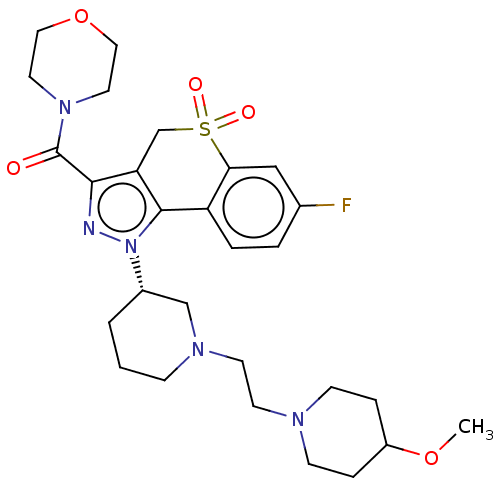
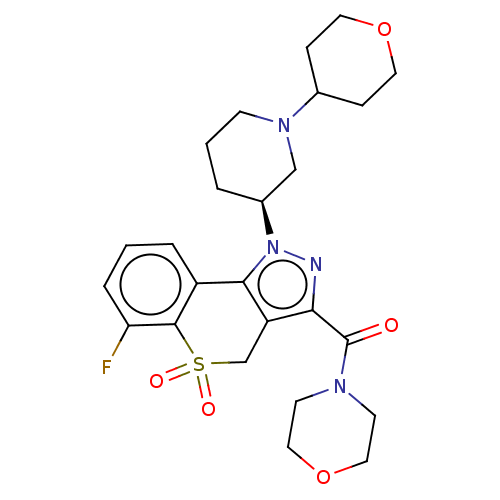
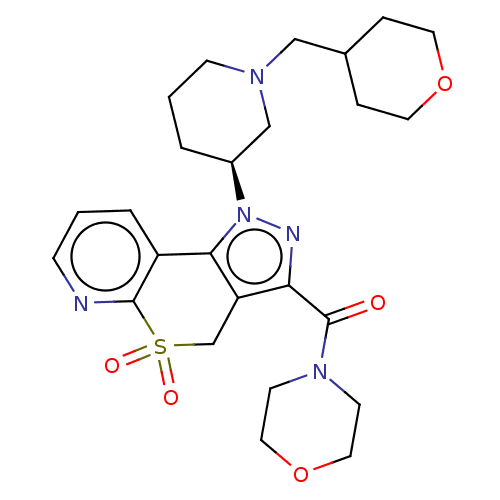
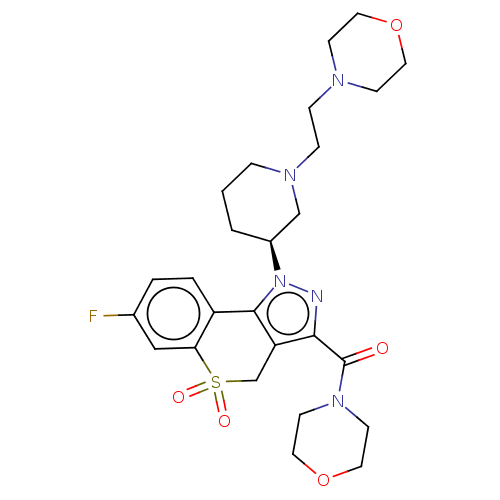
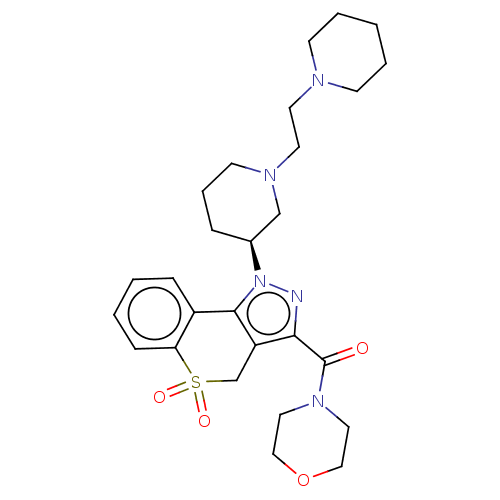
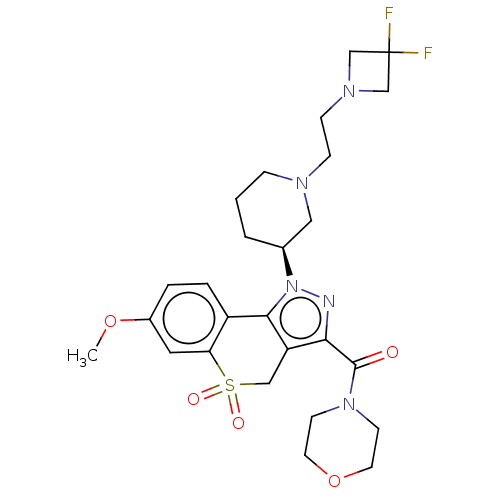
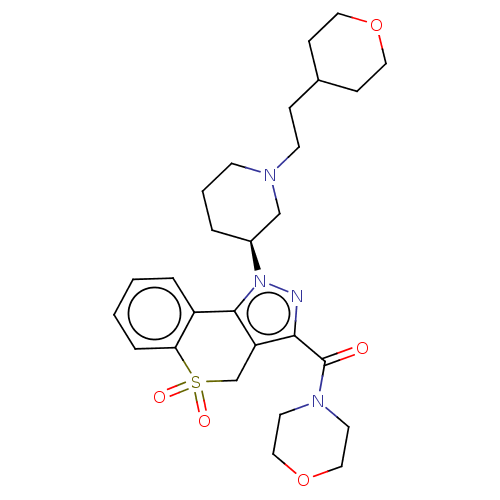
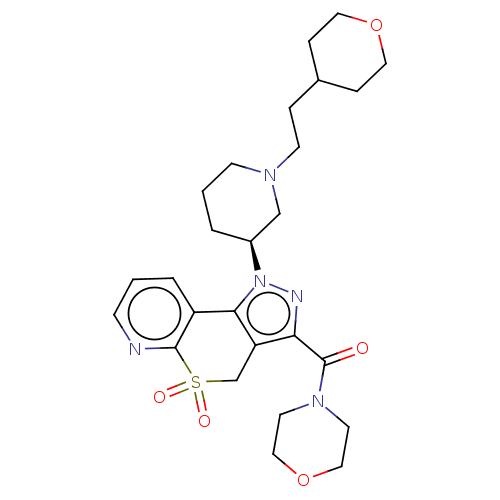
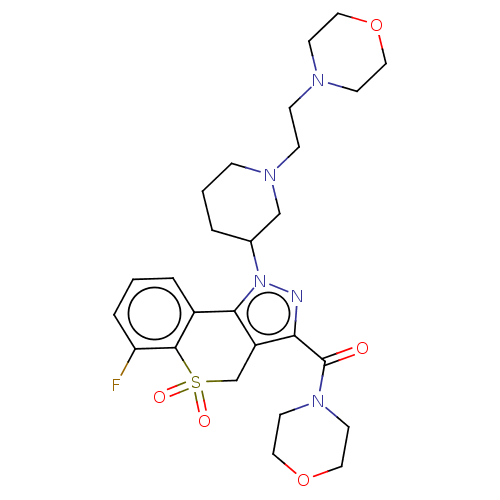
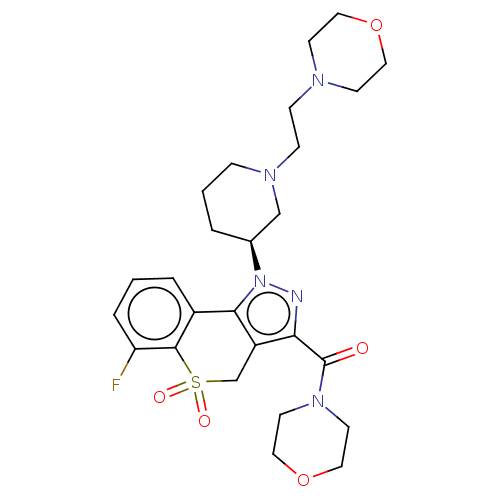
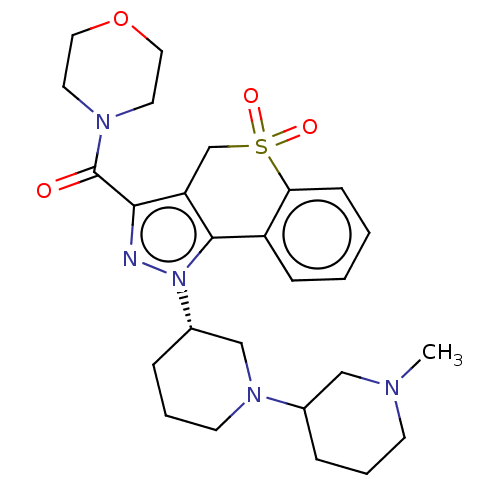
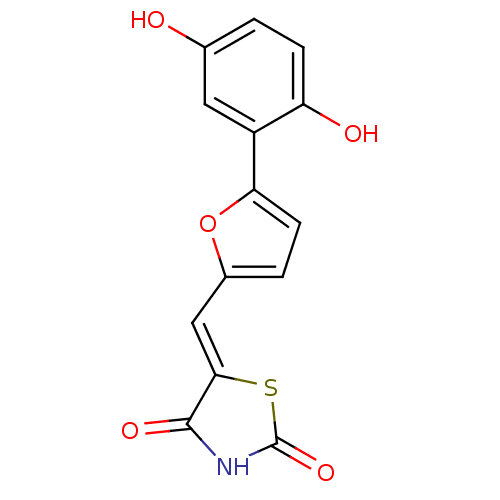
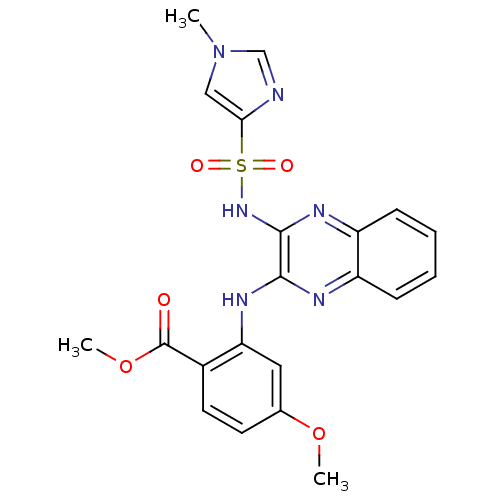
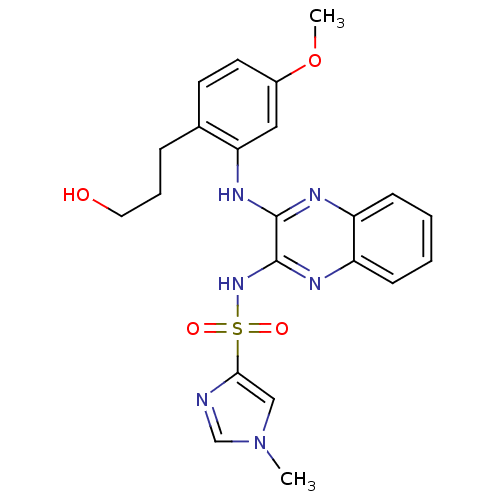
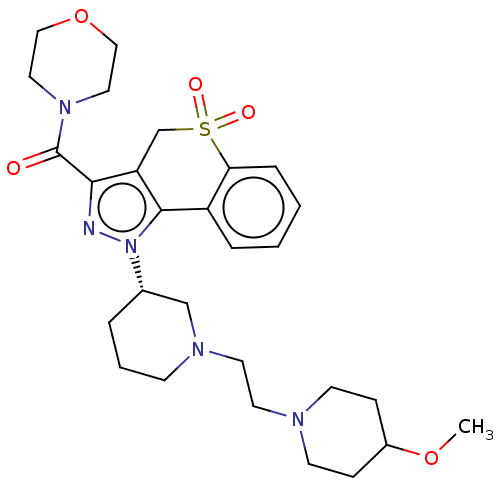
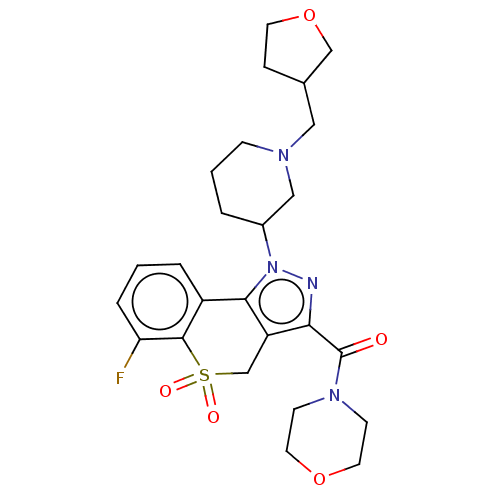
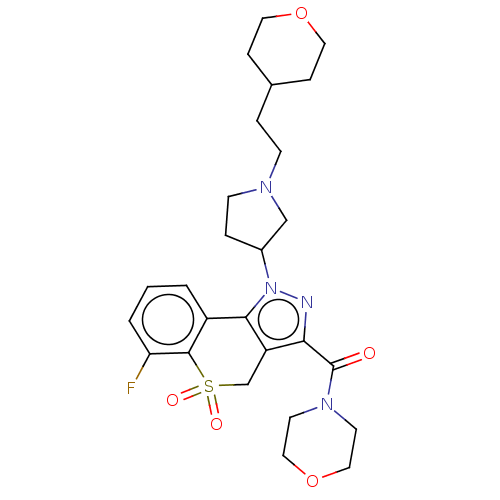
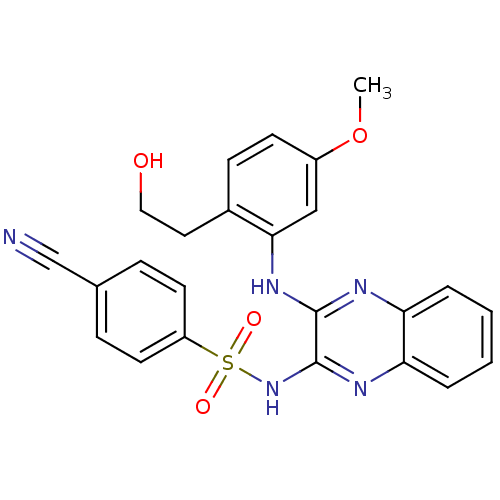
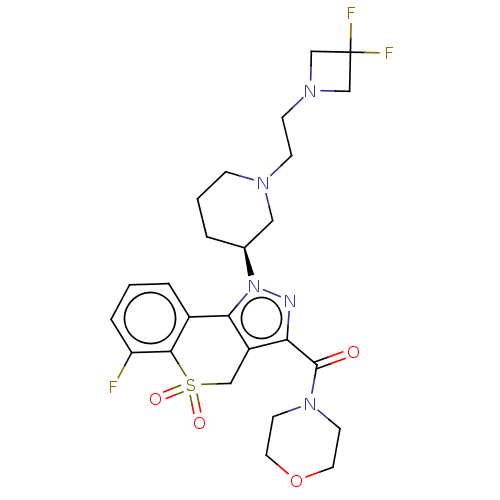
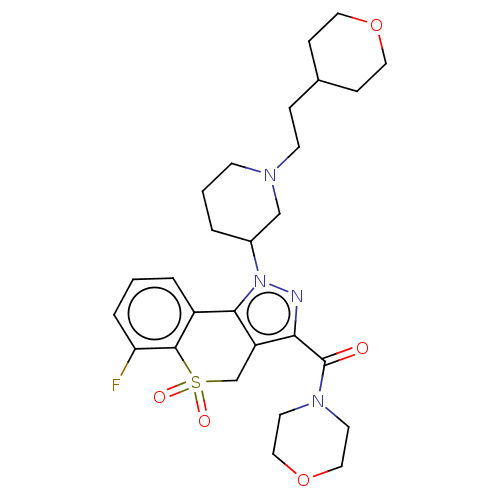
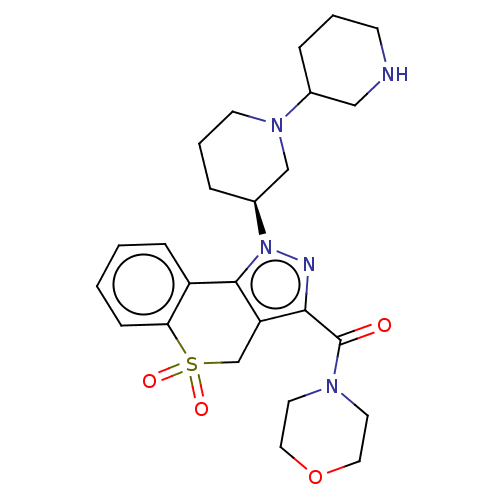
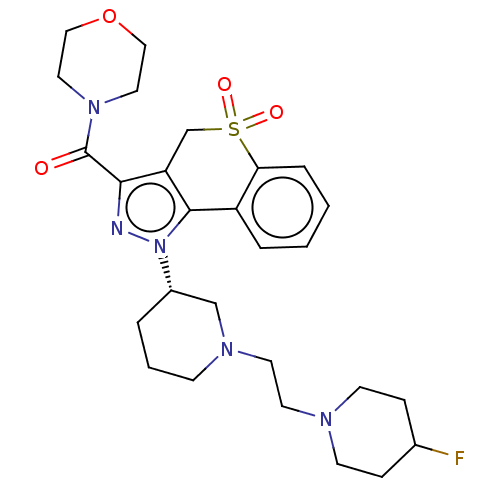
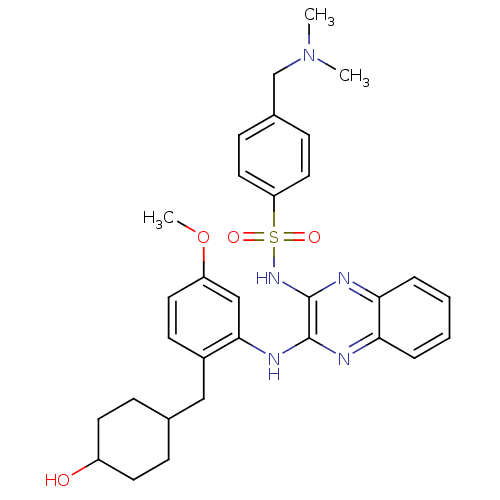
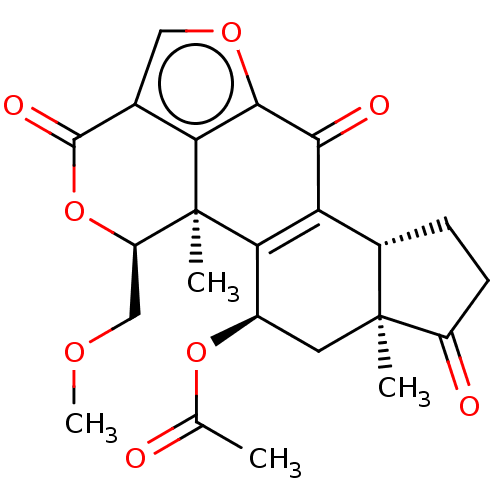
Affinity DataKi: 1nMAssay Description:The potency of inhibition of compound I and compound II on prostaglandin F2α receptor was assessed by analyzing the affinity of these compounds...More data for this Ligand-Target Pair
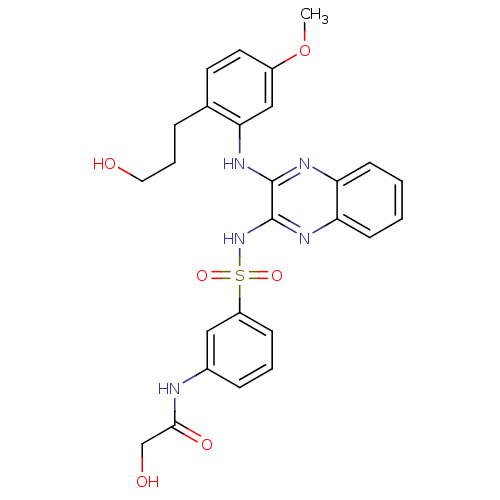
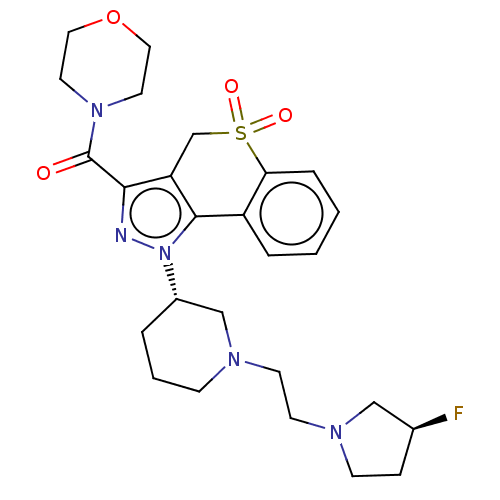
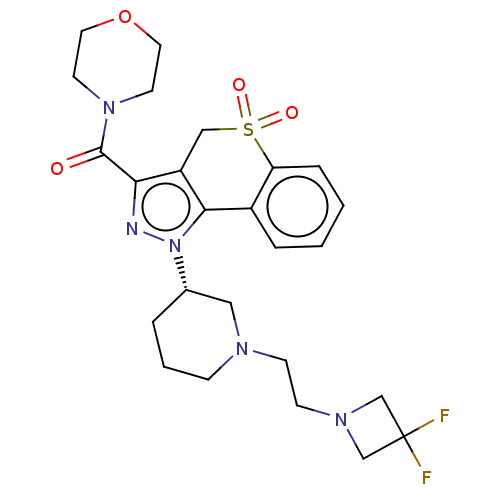
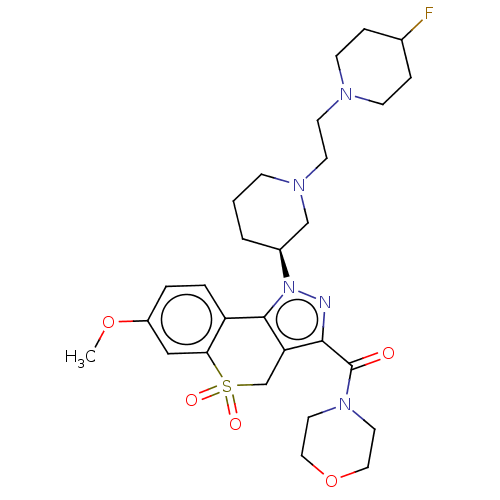
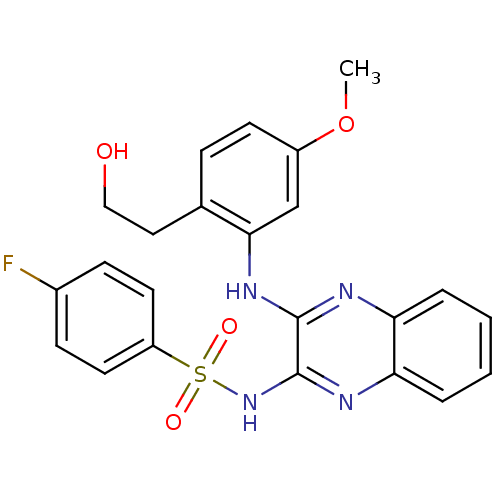
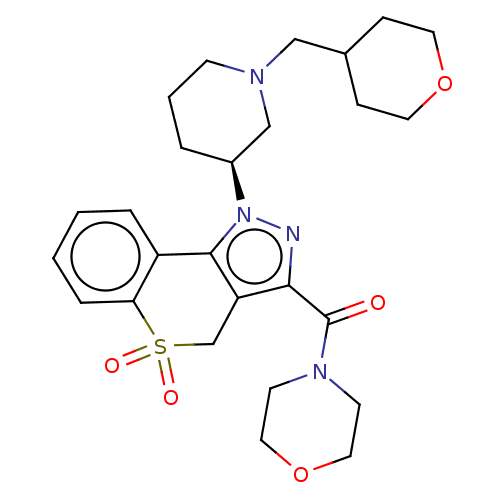
Affinity DataKi: 6nMAssay Description:The potency of inhibition of compound I and compound II on prostaglandin F2α receptor was assessed by analyzing the affinity of these compounds...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
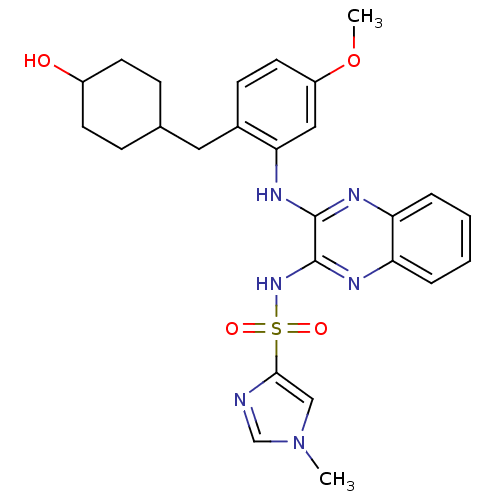
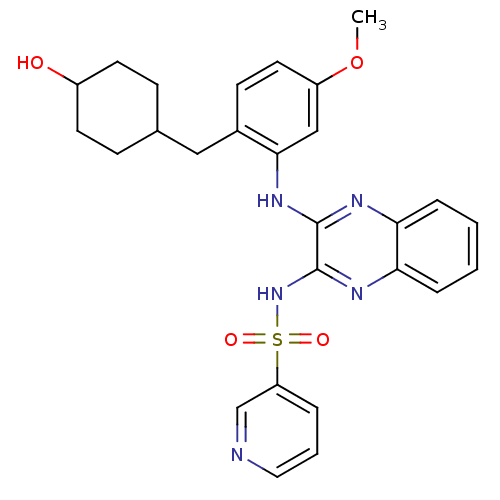
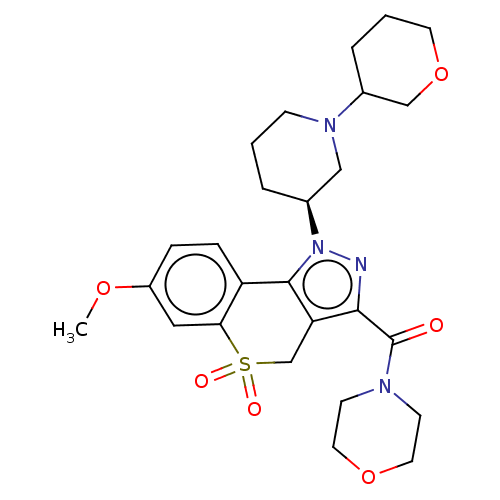
Affinity DataIC50: 1nMAssay Description:Inhibition of human PI3KalphaMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
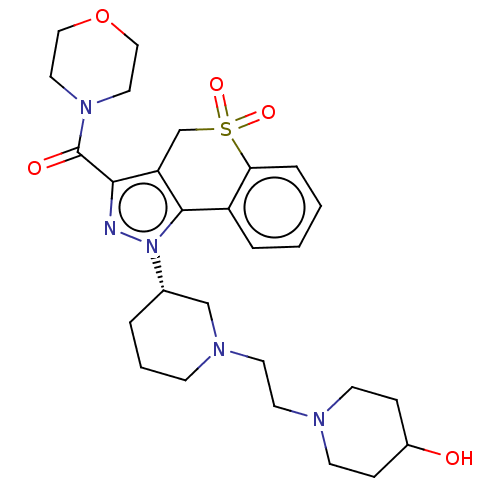
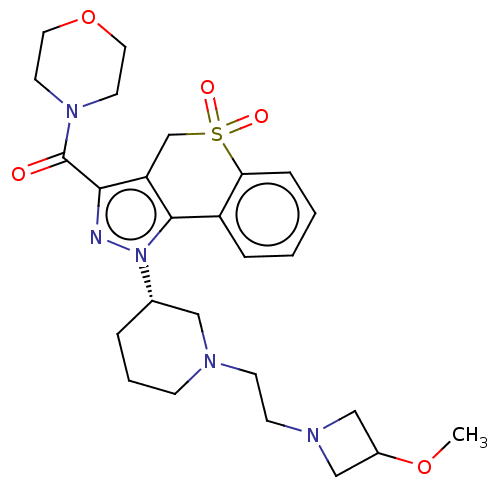
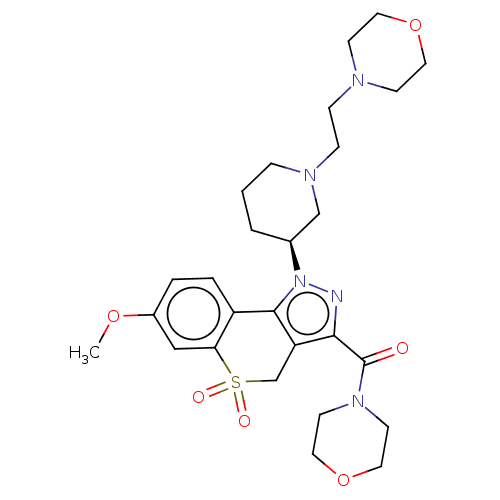
Affinity DataIC50: 5nMAssay Description:Inhibition of human PI3KgammaMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 6nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 7nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 9nMAssay Description:Inhibition of human PI3KdeltaMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of human PI3KbetaMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:The assay combines the scintillation proximity assay technology (SPA, Amersham) with the capacity of neomycin (a polycationic antibiotic) to bind pho...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:The assay combines the scintillation proximity assay technology (SPA, Amersham) with the capacity of neomycin (a polycationic antibiotic) to bind pho...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 11nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 12nMAssay Description:The assay combines the scintillation proximity assay technology (SPA, Amersham) with the capacity of neomycin (a polycationic antibiotic) to bind pho...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 13nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 13nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 13nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 14nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 14nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 15nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 15nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 16nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 16nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 16nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 16nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 17nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 18nMAssay Description:The assay combines the scintillation proximity assay technology (SPA, Amersham) with the capacity of neomycin (a polycationic antibiotic) to bind pho...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 18nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 18nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 19nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 20nMAssay Description:The assay combines the scintillation proximity assay technology (SPA, Amersham) with the capacity of neomycin (a polycationic antibiotic) to bind pho...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 20nMAssay Description:Inhibition of human PI3KgammaMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 20nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 20nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 20nMAssay Description:Inhibition of human PI3KgammaMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 21nMAssay Description:The assay combines the scintillation proximity assay technology (SPA, Amersham) with the capacity of neomycin (a polycationic antibiotic) to bind pho...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 21nMAssay Description:The assay combines the scintillation proximity assay technology (SPA, Amersham) with the capacity of neomycin (a polycationic antibiotic) to bind pho...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 21nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 21nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 21nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 22nMAssay Description:The assay combines the scintillation proximity assay technology (SPA, Amersham) with the capacity of neomycin (a polycationic antibiotic) to bind pho...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 22nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 22nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 23nMAssay Description:The assay combines the scintillation proximity assay technology (SPA, Amersham) with the capacity of neomycin (a polycationic antibiotic) to bind pho...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 24nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 24nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 25nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 25nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 25nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 25nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha/beta/delta/gamma isoform(Homo sapiens (Human))
Merck Serono
US Patent
Merck Serono
US Patent
Affinity DataIC50: 25nMAssay Description:The efficacy of compounds of the invention in inhibiting the PI3K induced-lipid phosphorylation may be tested in the following binding assay. The ass...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Serono Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 26nMAssay Description:The assay combines the scintillation proximity assay technology (SPA, Amersham) with the capacity of neomycin (a polycationic antibiotic) to bind pho...More data for this Ligand-Target Pair

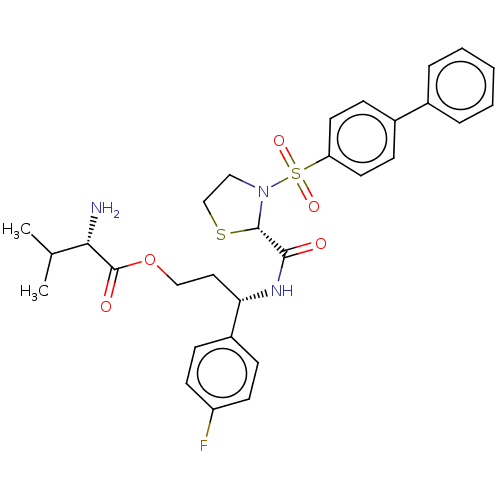
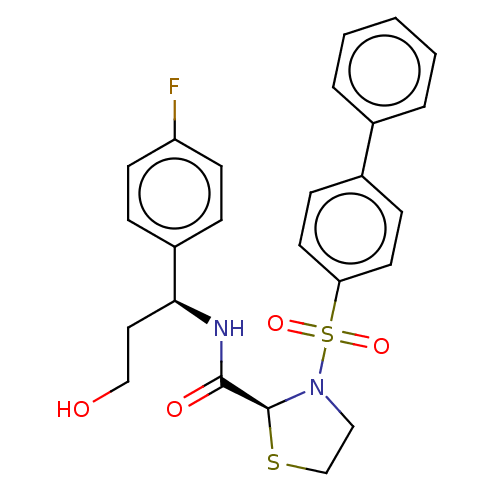
 3D Structure (crystal)
3D Structure (crystal)