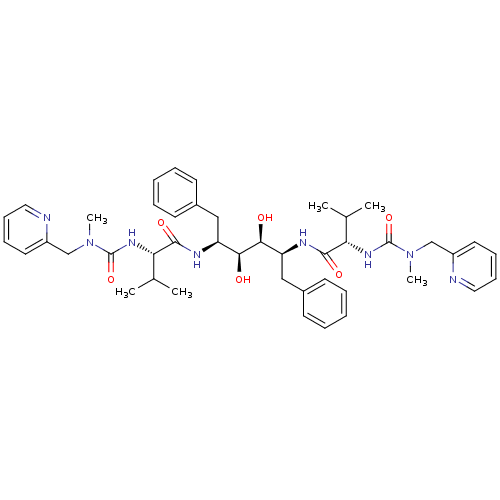
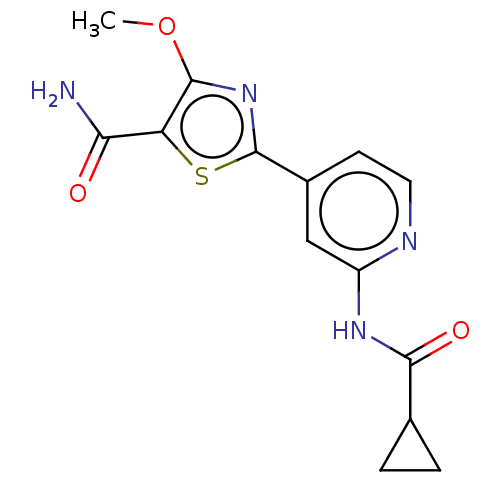
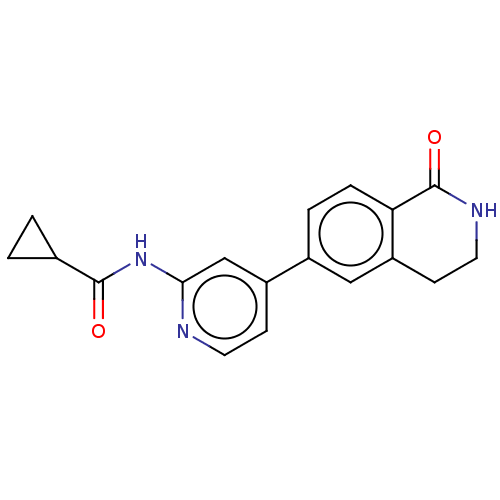
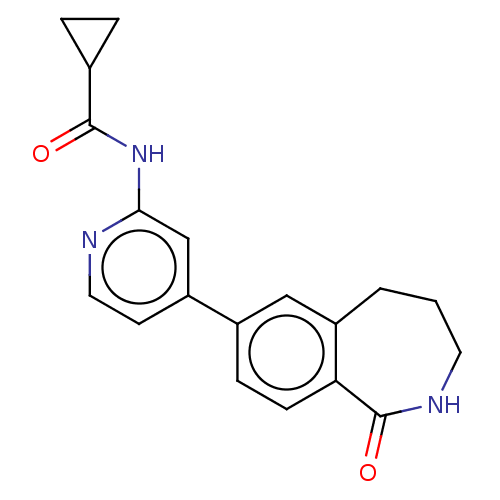
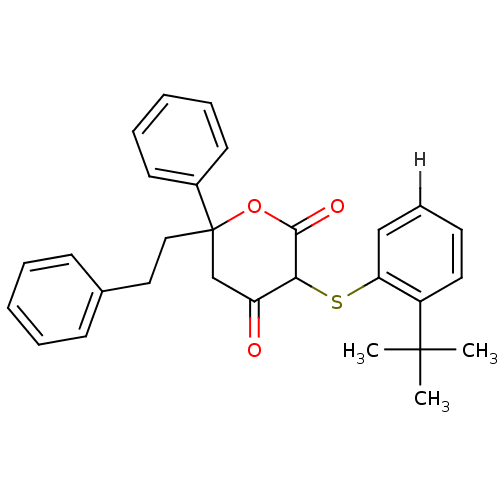
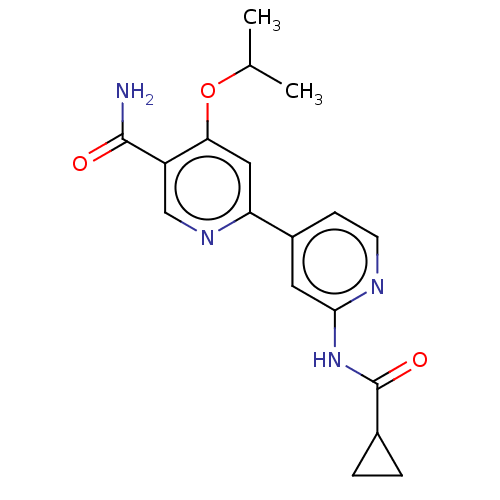
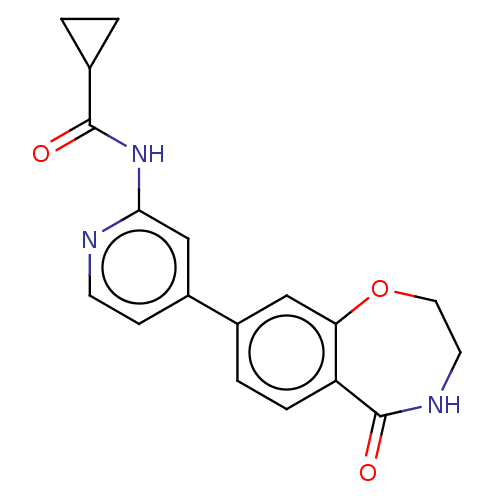
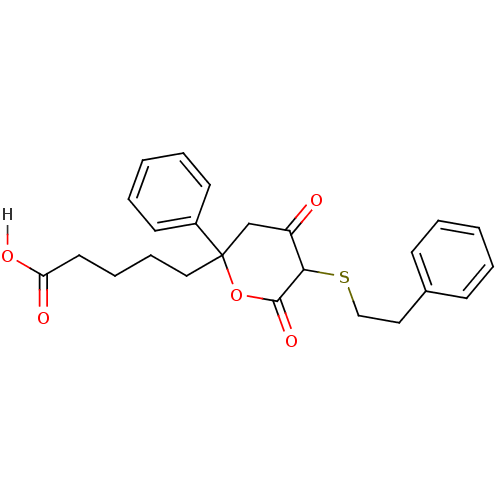
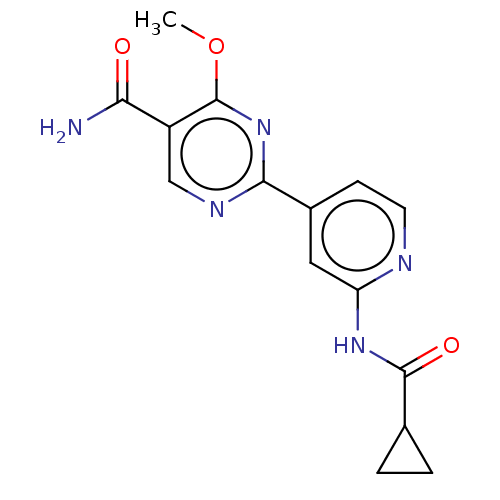
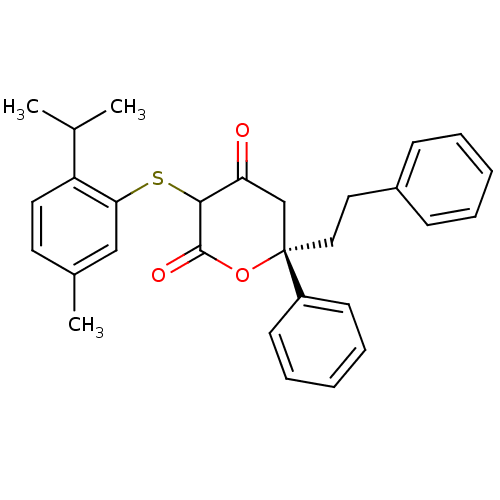
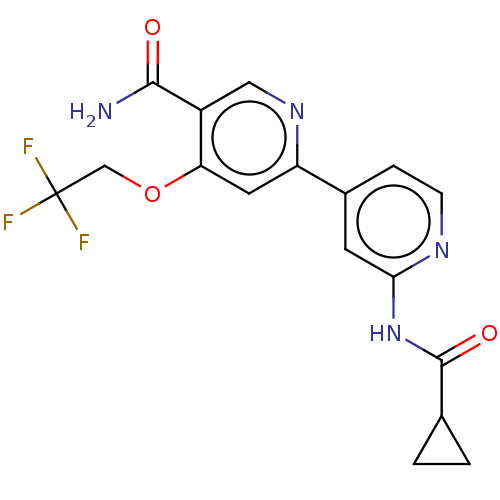
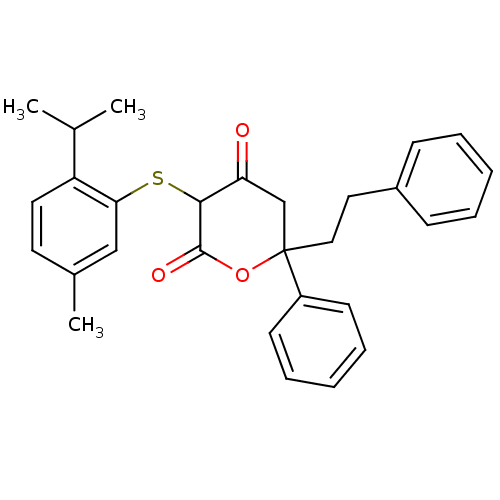
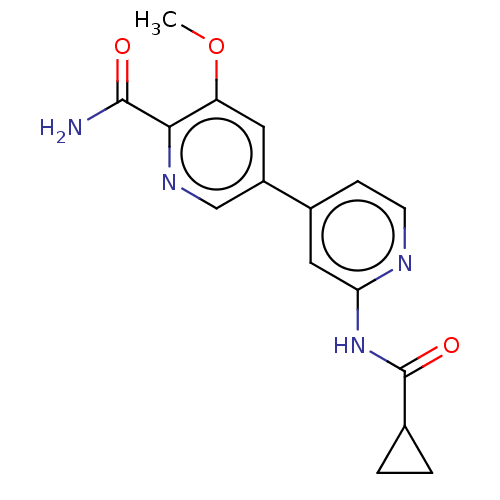
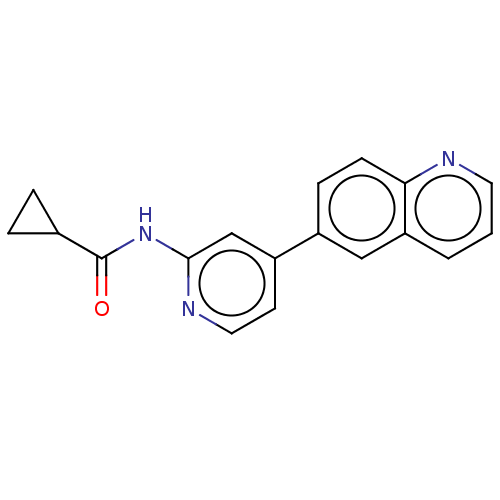
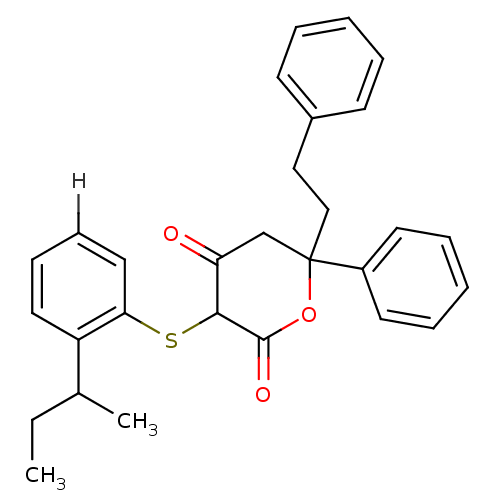
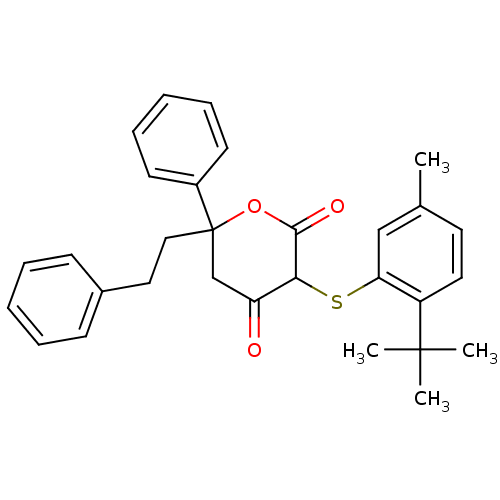
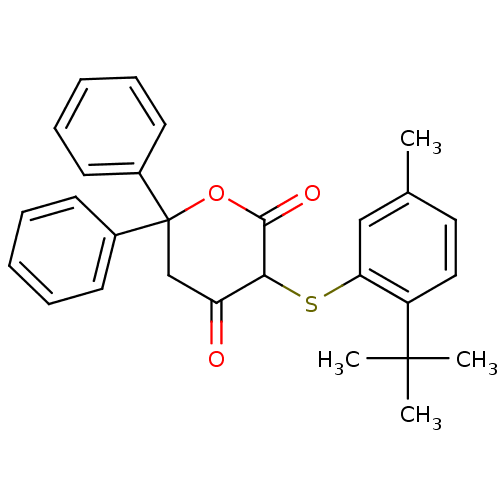
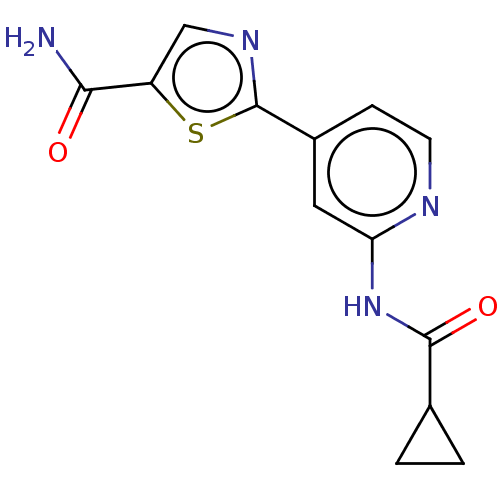
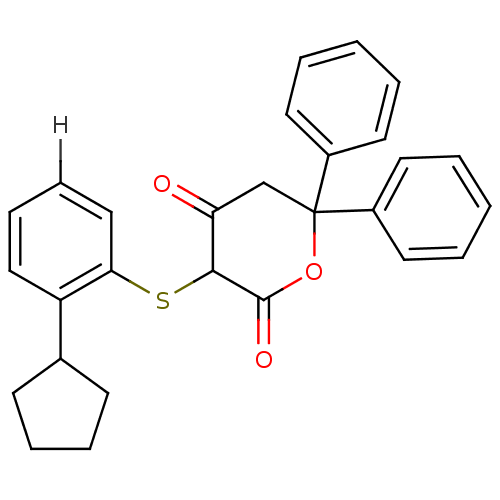
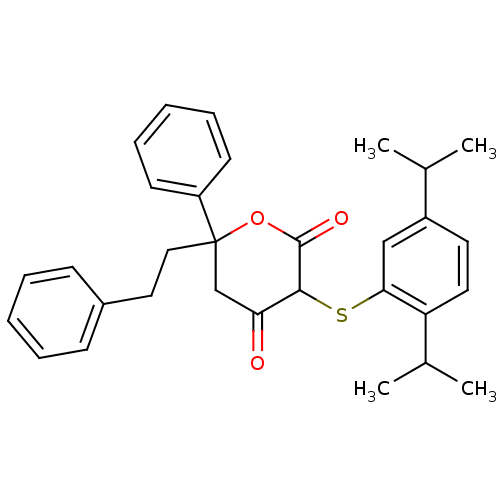
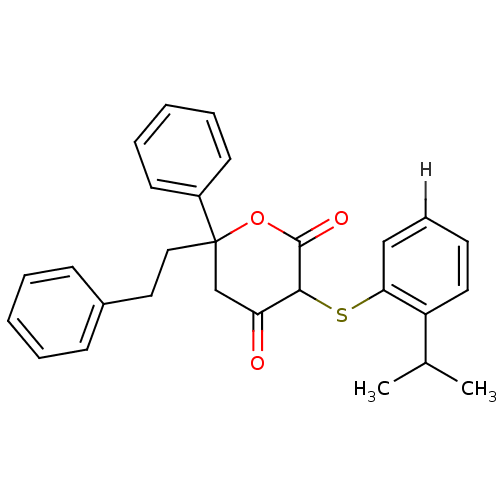
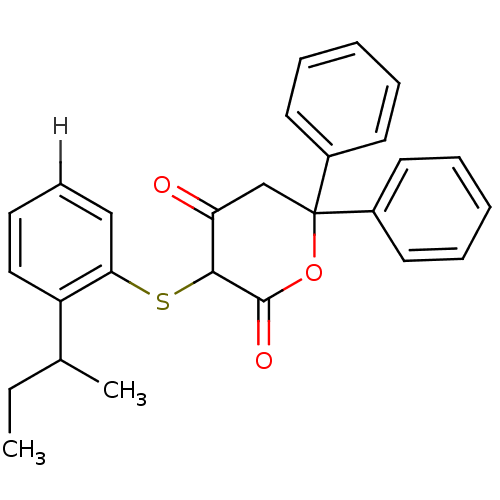
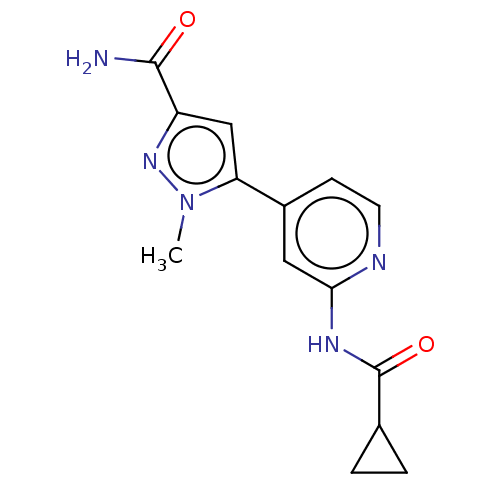
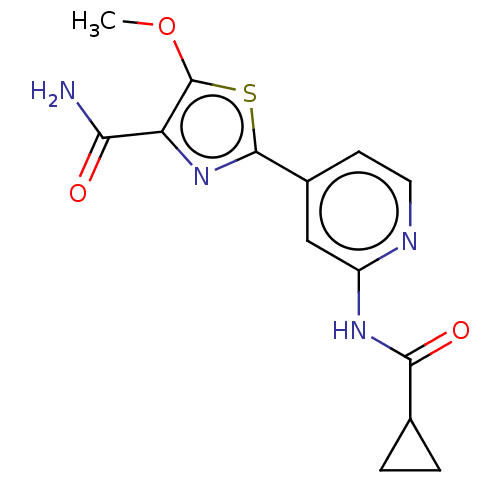
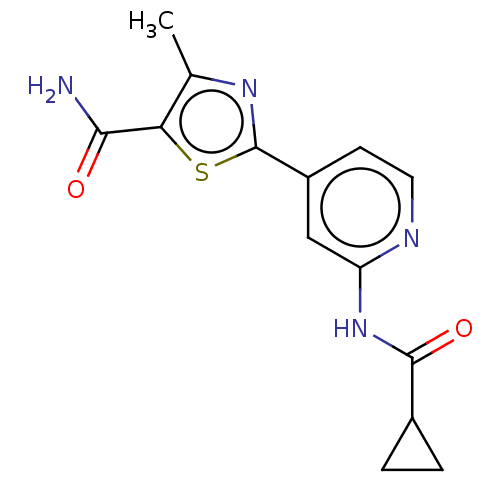
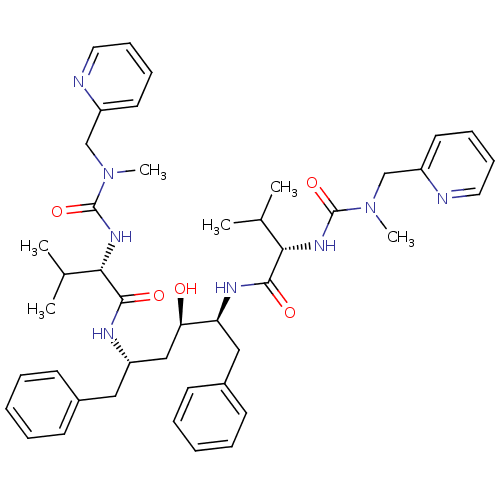
Affinity DataKi: 0.00400nM ΔG°: -66.1kJ/molepH: 4.7 T: 2°CAssay Description:HIV-1 protease activity was measured by a continuous fluorometric assay using the internally quenched fluorogenic substrate DABCYL-GABA-Ser-Gln-Tyr-P...More data for this Ligand-Target Pair
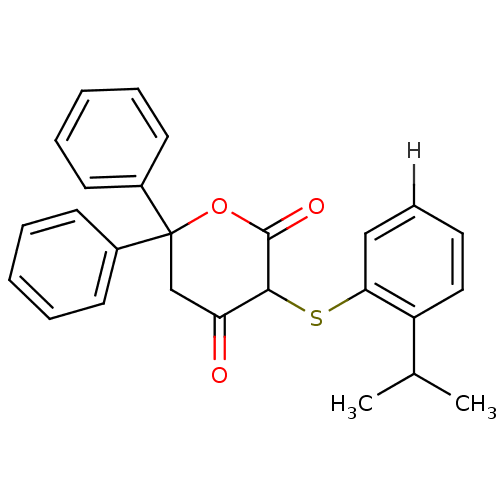
Affinity DataKi: 0.0110nM ΔG°: -63.6kJ/molepH: 4.7 T: 2°CAssay Description:HIV-1 protease activity was measured by a continuous fluorometric assay using the internally quenched fluorogenic substrate DABCYL-GABA-Ser-Gln-Tyr-P...More data for this Ligand-Target Pair
Affinity DataKi: 0.0120nM ΔG°: -63.4kJ/molepH: 4.7 T: 2°CAssay Description:HIV-1 protease activity was measured by a continuous fluorometric assay using the internally quenched fluorogenic substrate DABCYL-GABA-Ser-Gln-Tyr-P...More data for this Ligand-Target Pair
Affinity DataKi: 0.112nM ΔG°: -57.7kJ/molepH: 4.7 T: 2°CAssay Description:HIV-1 protease activity was measured by a continuous fluorometric assay using the internally quenched fluorogenic substrate DABCYL-GABA-Ser-Gln-Tyr-P...More data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
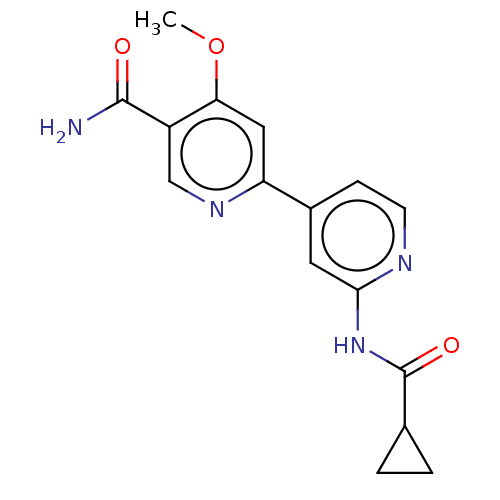
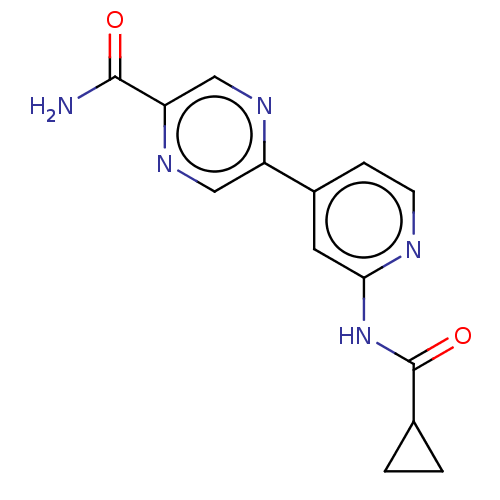
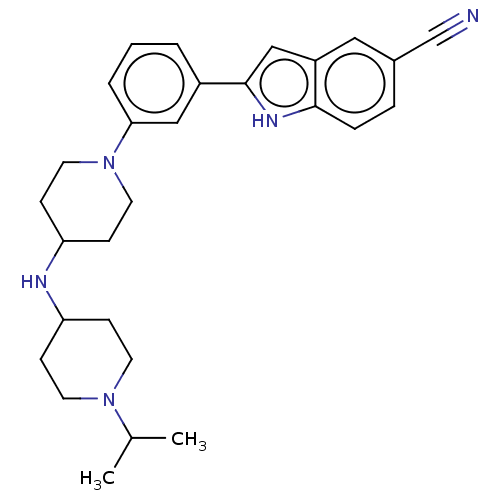
Affinity DataIC50: 0.290nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 0.370nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 0.450nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 0.830nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 1.10nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 2.40nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 2.40nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
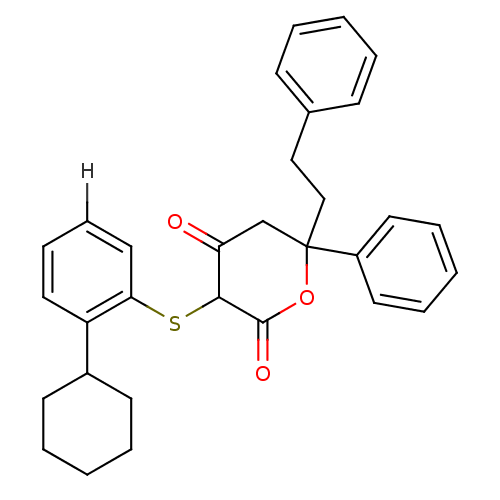
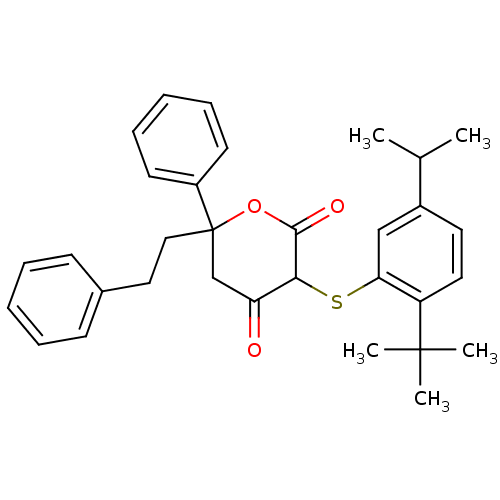
Affinity DataIC50: 3nMAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 4.40nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 4.70nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5nMpH: 4.7 T: 2°CAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 5.90nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 6.80nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 7nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 7.5nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 13nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair
TargetTransitional endoplasmic reticulum ATPase(Homo sapiens (Human))
University of Pittsburgh
US Patent
University of Pittsburgh
US Patent
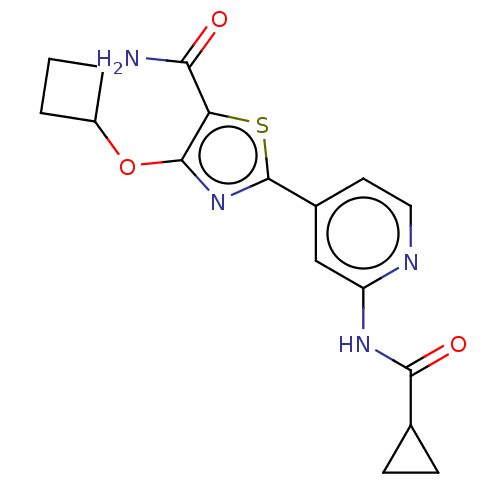
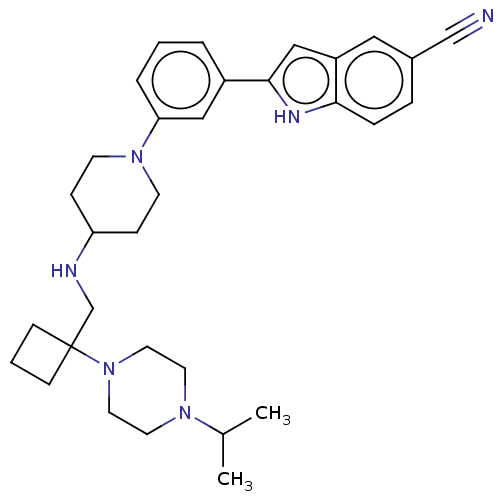
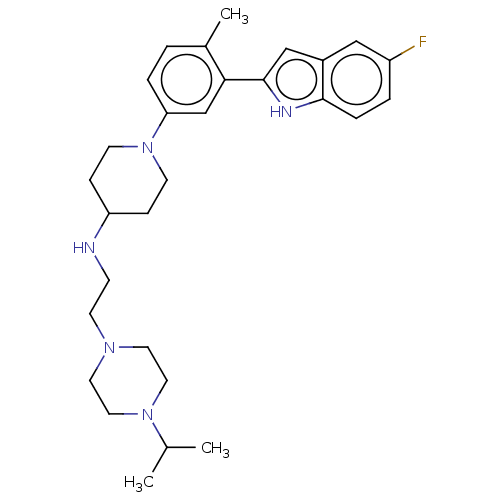
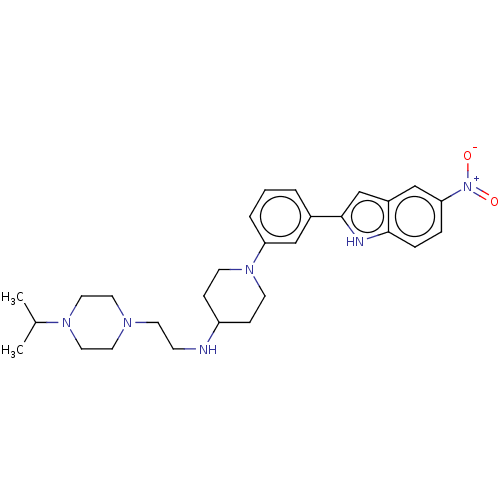
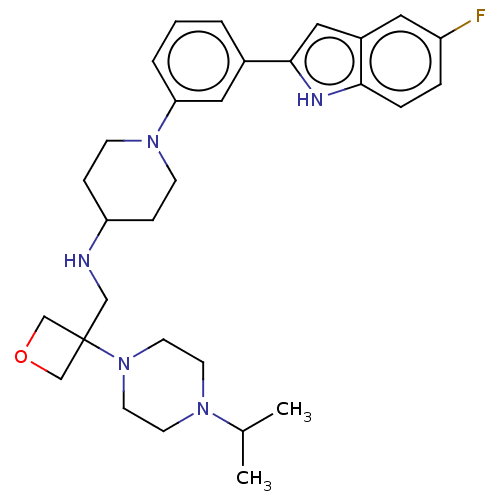
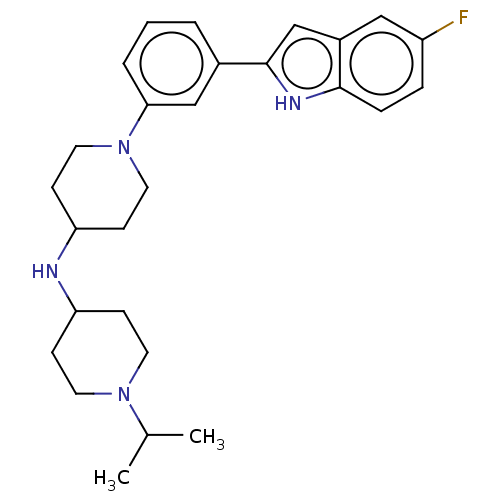
Affinity DataIC50: 15nMAssay Description:To optimize p97 inhibitors, the C-5 trifluoromethylated trifluoromethylated indole 12 was generated as a promising lead structure. In the ADP-Glo ass...More data for this Ligand-Target Pair
TargetTransitional endoplasmic reticulum ATPase(Homo sapiens (Human))
University of Pittsburgh
US Patent
University of Pittsburgh
US Patent
Affinity DataIC50: 15nMAssay Description:To optimize p97 inhibitors, the C-5 trifluoromethylated trifluoromethylated indole 12 was generated as a promising lead structure. In the ADP-Glo ass...More data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 22nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 23nMAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 24nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
TargetTransitional endoplasmic reticulum ATPase(Homo sapiens (Human))
University of Pittsburgh
US Patent
University of Pittsburgh
US Patent
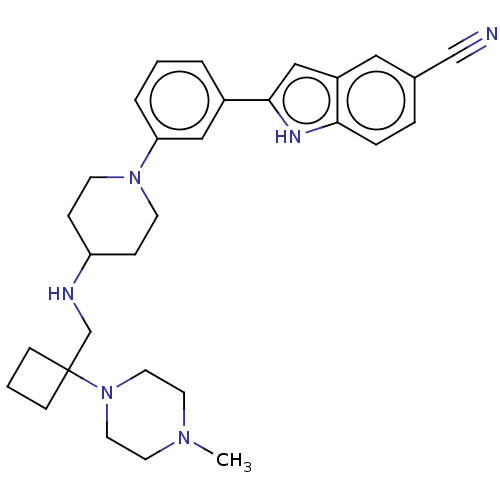
Affinity DataIC50: 25nMAssay Description:To optimize p97 inhibitors, the C-5 trifluoromethylated trifluoromethylated indole 12 was generated as a promising lead structure. In the ADP-Glo ass...More data for this Ligand-Target Pair
TargetTransitional endoplasmic reticulum ATPase(Homo sapiens (Human))
University of Pittsburgh
US Patent
University of Pittsburgh
US Patent
Affinity DataIC50: 26nMAssay Description:To optimize p97 inhibitors, the C-5 trifluoromethylated trifluoromethylated indole 12 was generated as a promising lead structure. In the ADP-Glo ass...More data for this Ligand-Target Pair
TargetTransitional endoplasmic reticulum ATPase(Homo sapiens (Human))
University of Pittsburgh
US Patent
University of Pittsburgh
US Patent
Affinity DataIC50: 26nMAssay Description:To optimize p97 inhibitors, the C-5 trifluoromethylated trifluoromethylated indole 12 was generated as a promising lead structure. In the ADP-Glo ass...More data for this Ligand-Target Pair
TargetTransitional endoplasmic reticulum ATPase(Homo sapiens (Human))
University of Pittsburgh
US Patent
University of Pittsburgh
US Patent
Affinity DataIC50: 27nMAssay Description:To optimize p97 inhibitors, the C-5 trifluoromethylated trifluoromethylated indole 12 was generated as a promising lead structure. In the ADP-Glo ass...More data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 30nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 30nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 32nMAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair
TargetTransitional endoplasmic reticulum ATPase(Homo sapiens (Human))
University of Pittsburgh
US Patent
University of Pittsburgh
US Patent
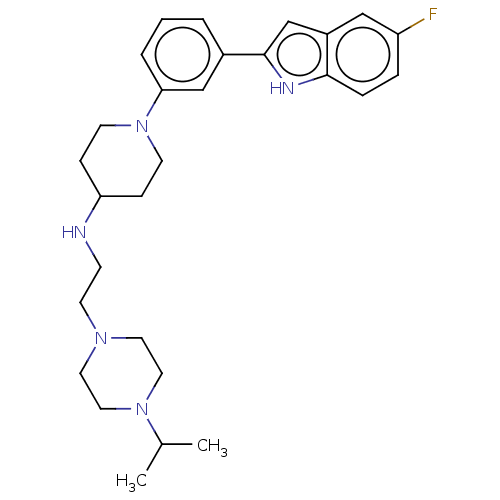
Affinity DataIC50: 36nMAssay Description:To optimize p97 inhibitors, the C-5 trifluoromethylated trifluoromethylated indole 12 was generated as a promising lead structure. In the ADP-Glo ass...More data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 37nMAssay Description:Inhibition of GSK3beta (unknown origin) activity by competitive binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 38nMAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair
TargetTransitional endoplasmic reticulum ATPase(Homo sapiens (Human))
University of Pittsburgh
US Patent
University of Pittsburgh
US Patent
Affinity DataIC50: 41nMAssay Description:To optimize p97 inhibitors, the C-5 trifluoromethylated trifluoromethylated indole 12 was generated as a promising lead structure. In the ADP-Glo ass...More data for this Ligand-Target Pair
Affinity DataIC50: 47nMAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair
TargetTransitional endoplasmic reticulum ATPase(Homo sapiens (Human))
University of Pittsburgh
US Patent
University of Pittsburgh
US Patent
Affinity DataIC50: 47nMAssay Description:To optimize p97 inhibitors, the C-5 trifluoromethylated trifluoromethylated indole 12 was generated as a promising lead structure. In the ADP-Glo ass...More data for this Ligand-Target Pair
TargetTransitional endoplasmic reticulum ATPase(Homo sapiens (Human))
University of Pittsburgh
US Patent
University of Pittsburgh
US Patent
Affinity DataIC50: 49nMAssay Description:To optimize p97 inhibitors, the C-5 trifluoromethylated trifluoromethylated indole 12 was generated as a promising lead structure. In the ADP-Glo ass...More data for this Ligand-Target Pair
Affinity DataIC50: 49nMAssay Description:For determination of IC50 values, HIV-1 protease was added to assay buffer containing inhibitor and the substrate (H-His-Lys-Ala-Arg-Val-Leu- (p-NO2)...More data for this Ligand-Target Pair

 3D Structure (crystal)
3D Structure (crystal)