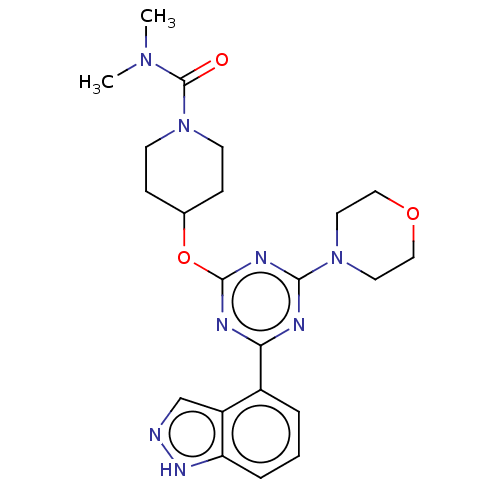
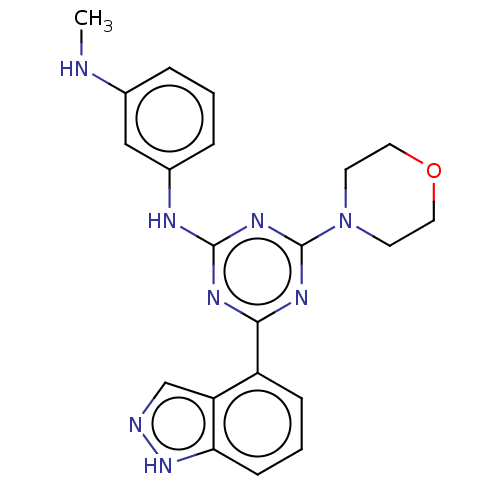
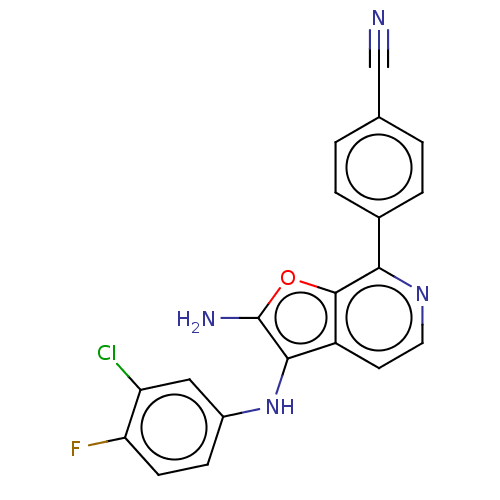
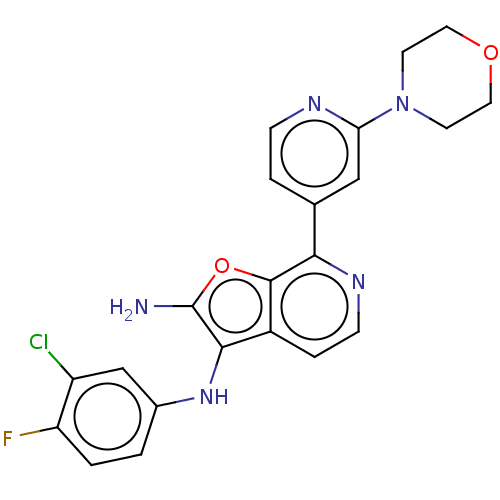
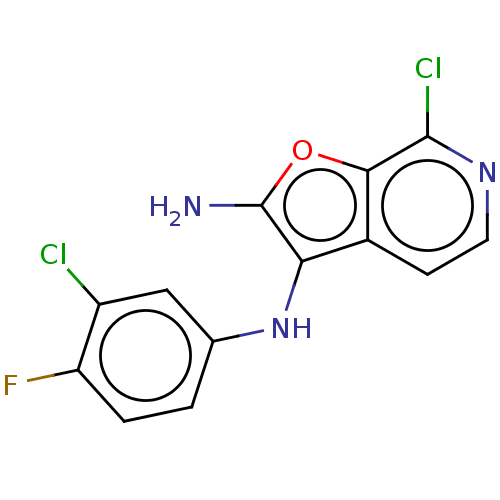
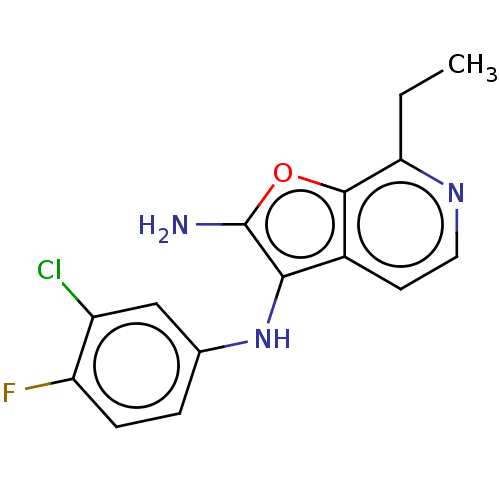
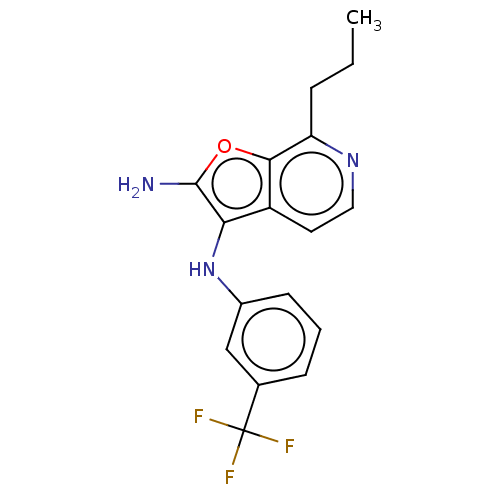
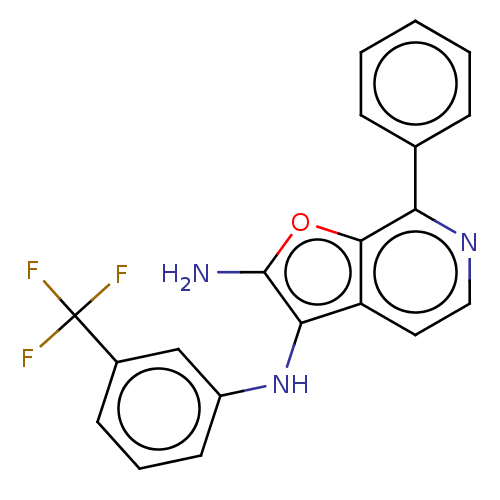
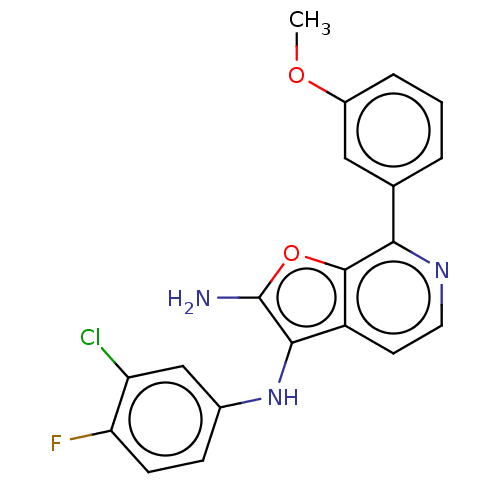
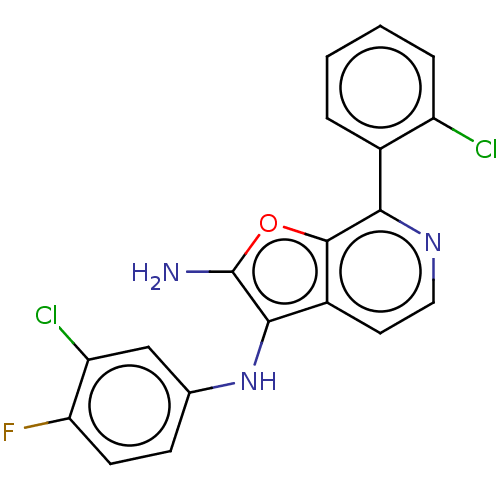
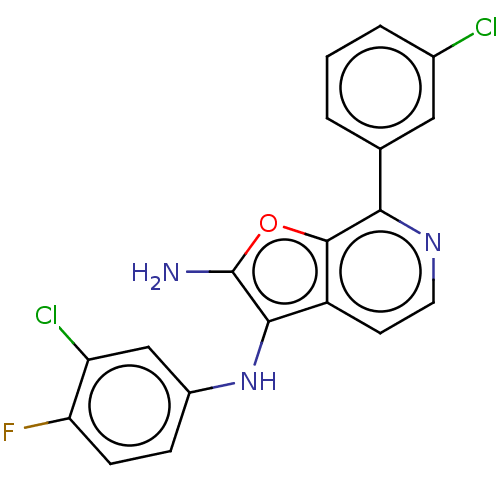
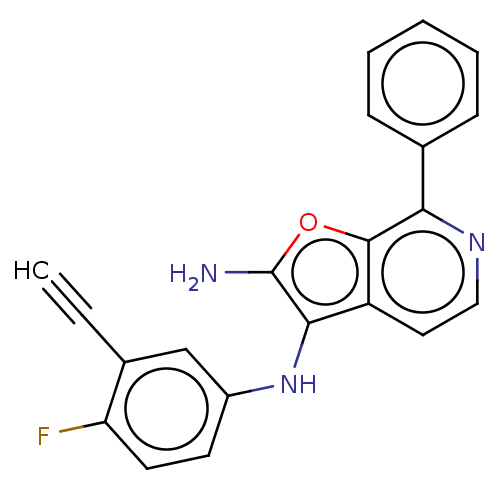
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Sphaera Pharma
Curated by ChEMBL
Sphaera Pharma
Curated by ChEMBL
Affinity DataIC50: 60nMAssay Description:Inhibition of human recombinant PI3K-alpha using phosphatidylinositol biphosphate as substrate preincubated for 15 mins followed by substrate additio...More data for this Ligand-Target Pair
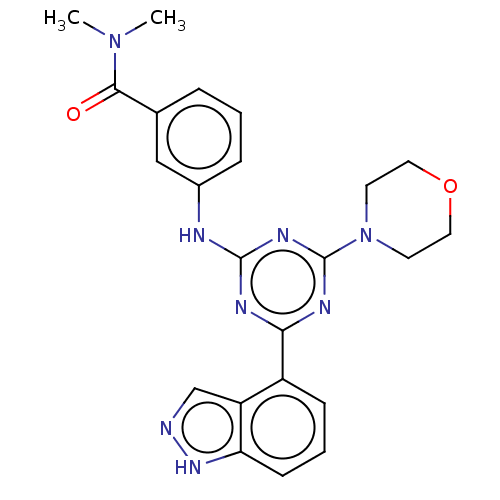
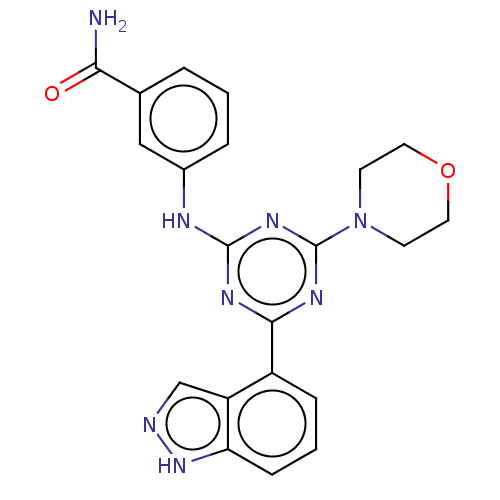
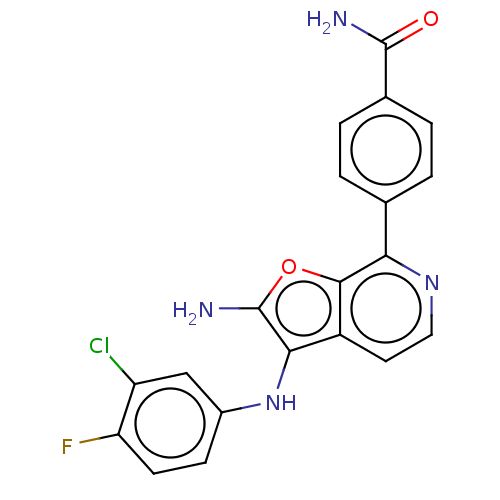
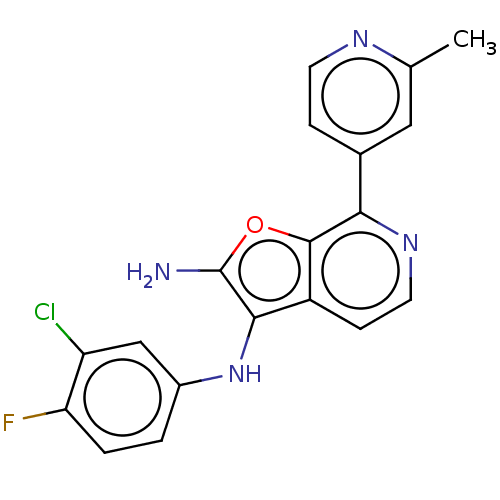
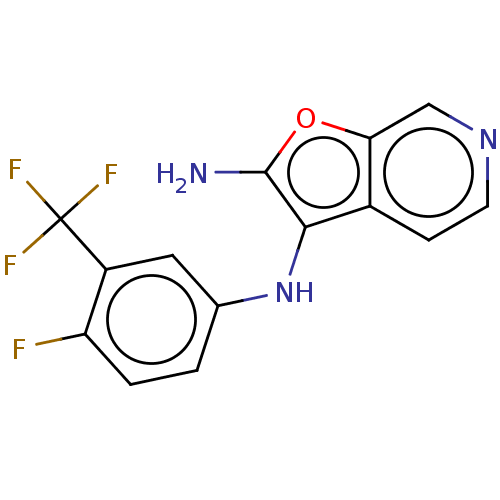
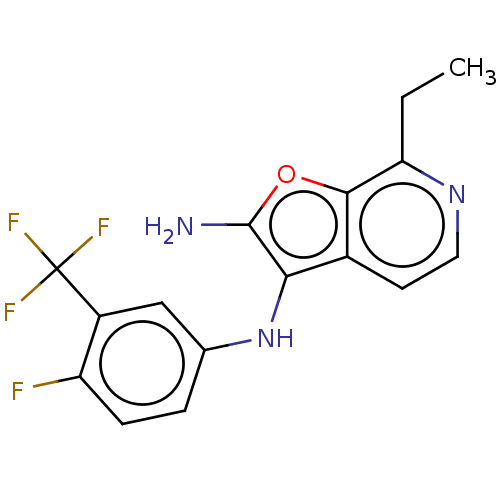
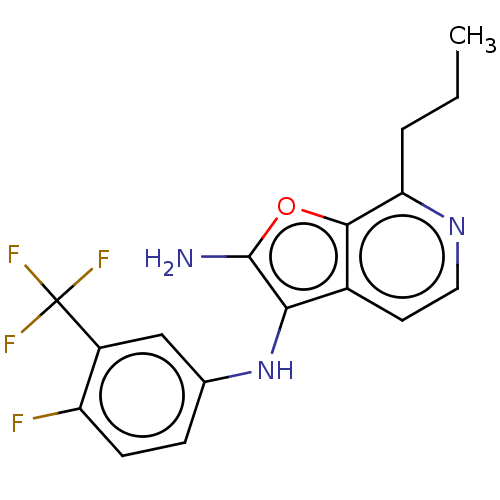
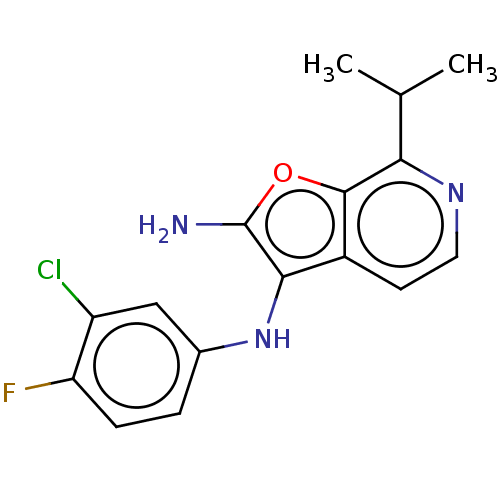
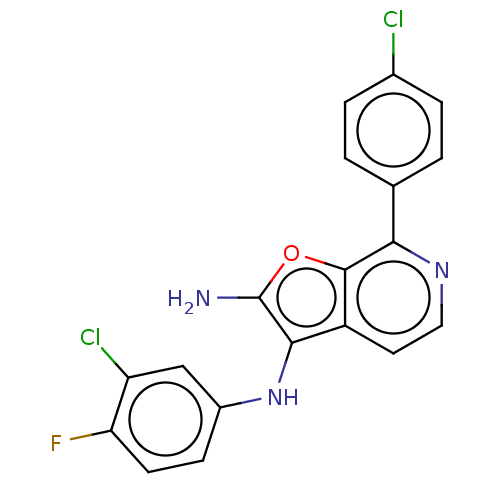
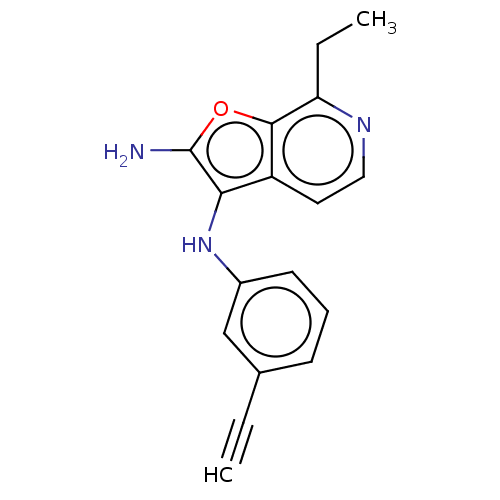
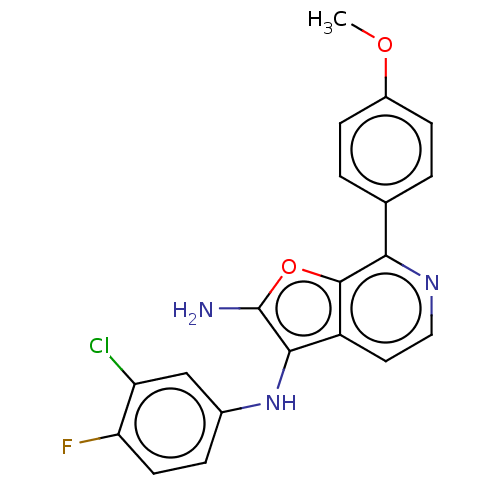
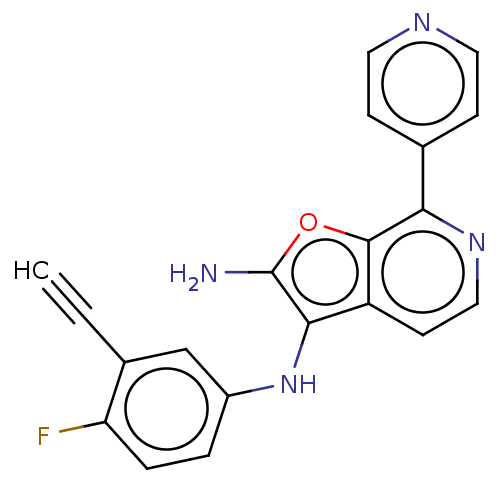
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Sphaera Pharma
Curated by ChEMBL
Sphaera Pharma
Curated by ChEMBL
Affinity DataIC50: 60nMAssay Description:Inhibition of human recombinant PI3K-alpha using phosphatidylinositol biphosphate as substrate preincubated for 15 mins followed by substrate additio...More data for this Ligand-Target Pair
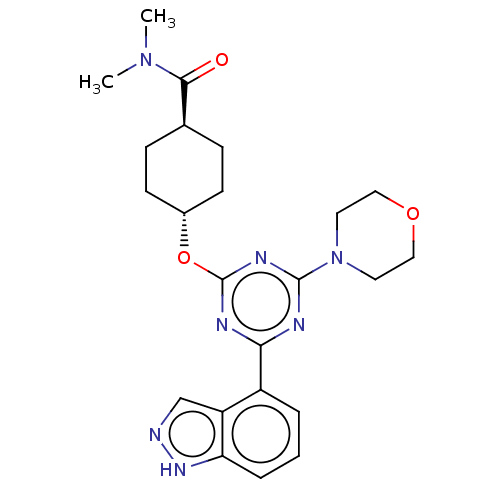
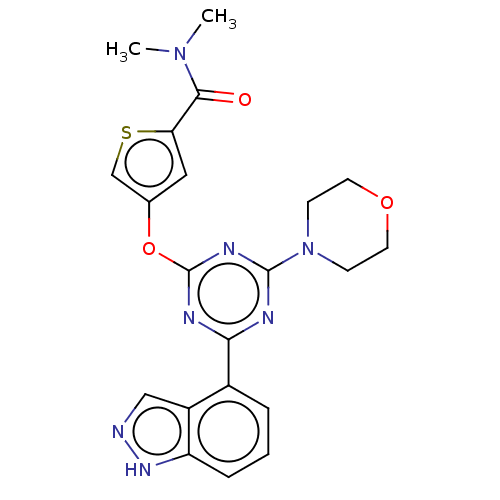
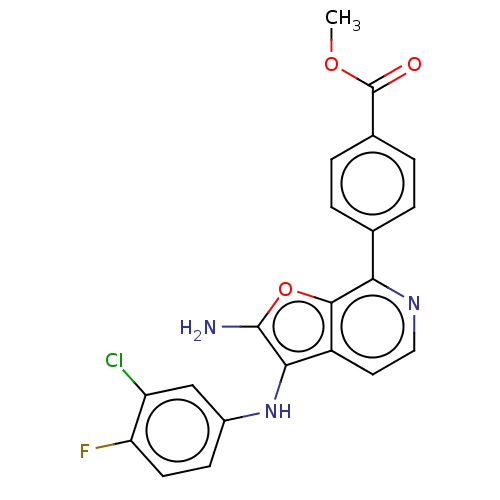
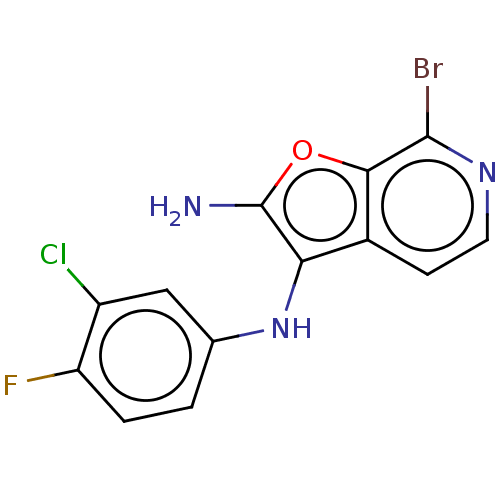
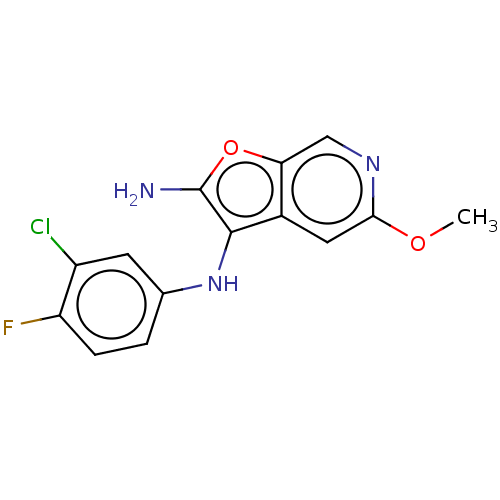
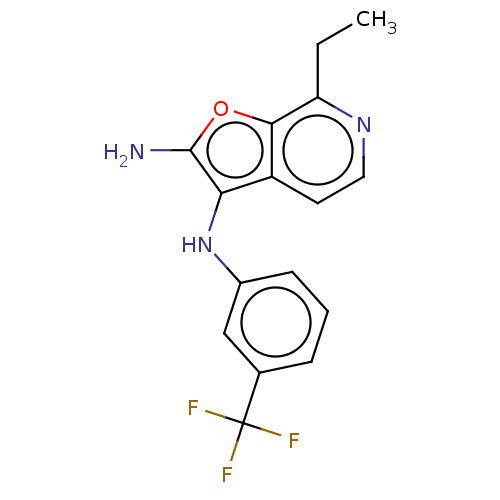
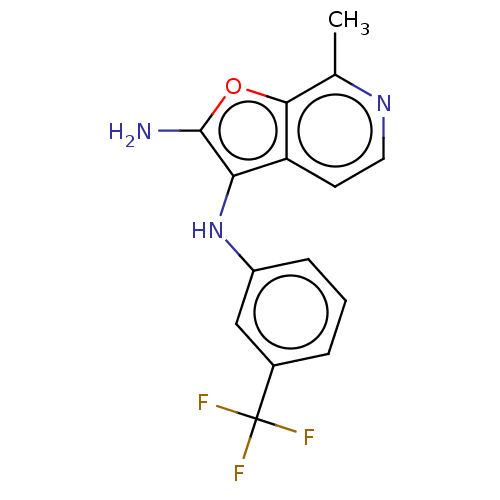
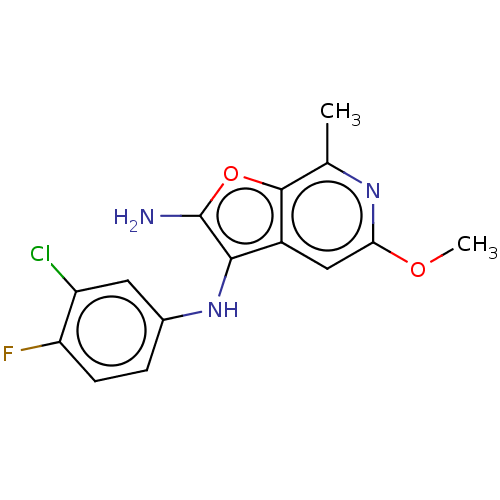
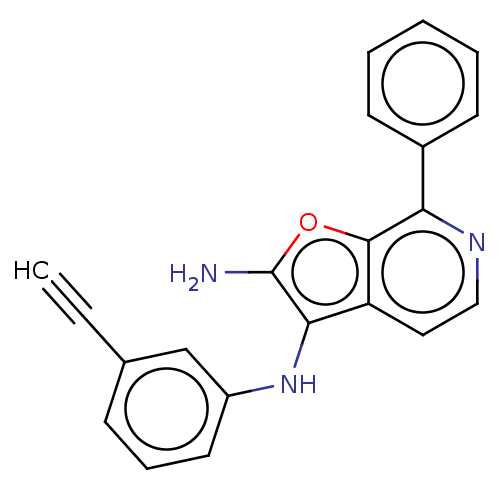
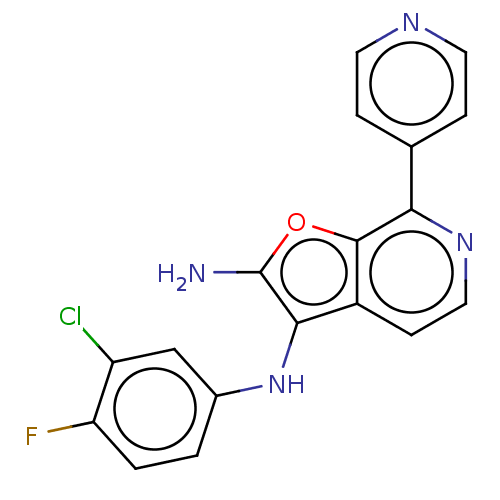
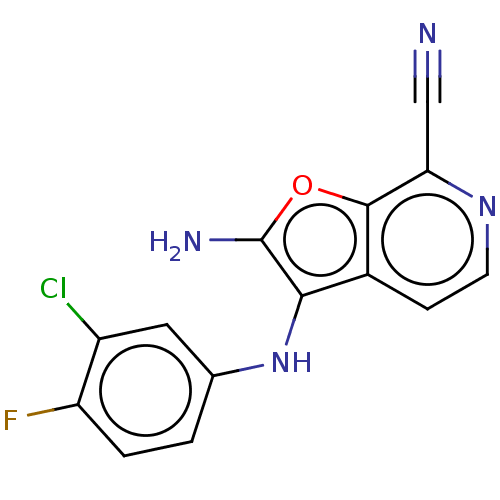
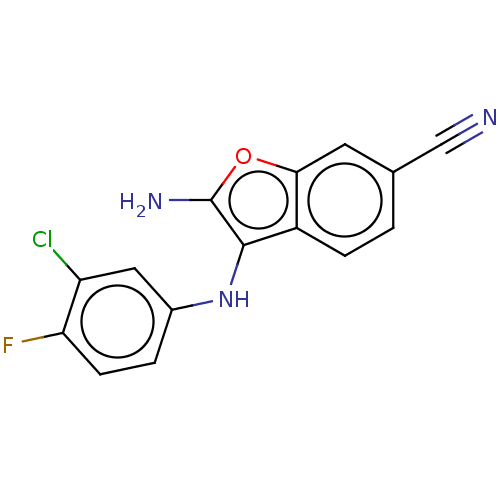
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Sphaera Pharma
Curated by ChEMBL
Sphaera Pharma
Curated by ChEMBL
Affinity DataIC50: 60nMAssay Description:Inhibition of human recombinant PI3K-alpha using phosphatidylinositol biphosphate as substrate preincubated for 15 mins followed by substrate additio...More data for this Ligand-Target Pair
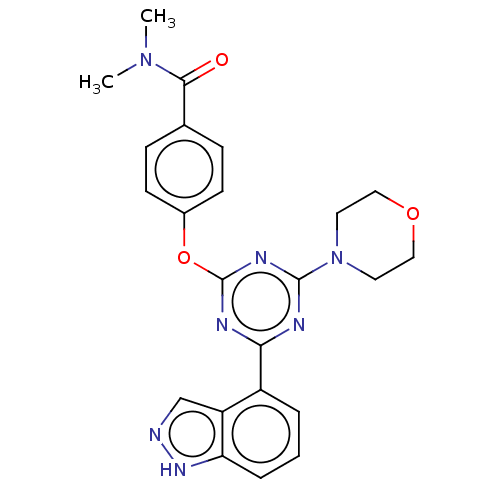
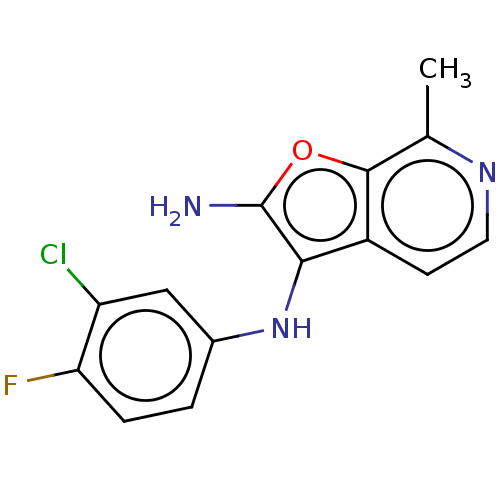
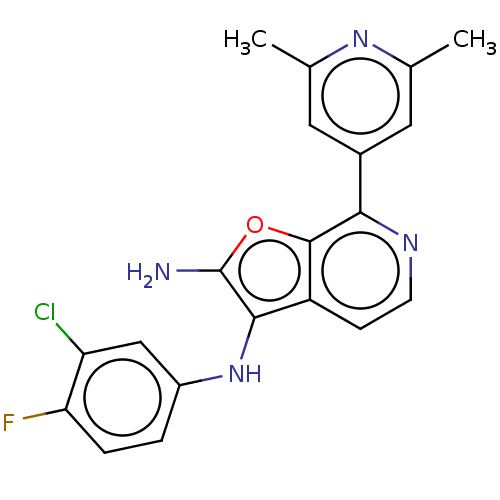
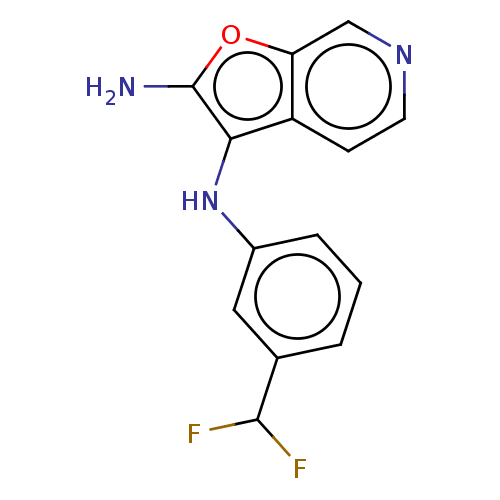
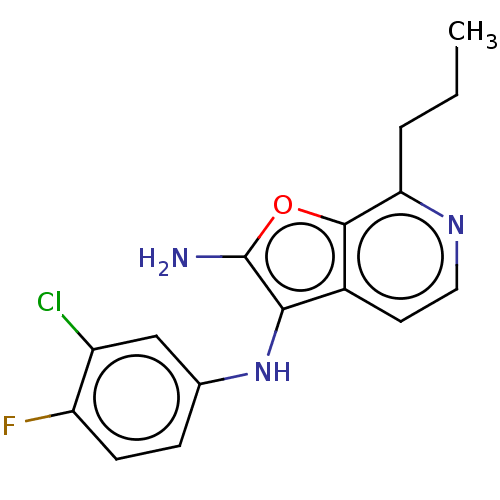
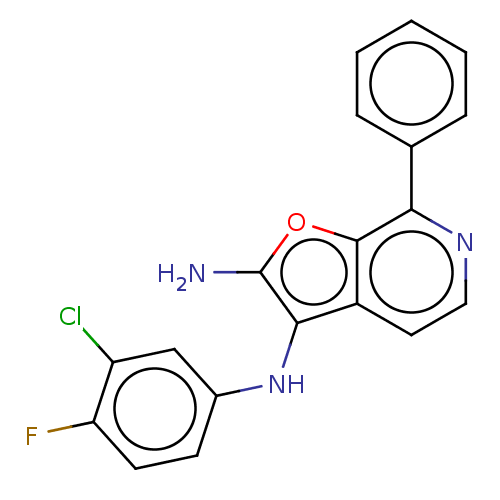
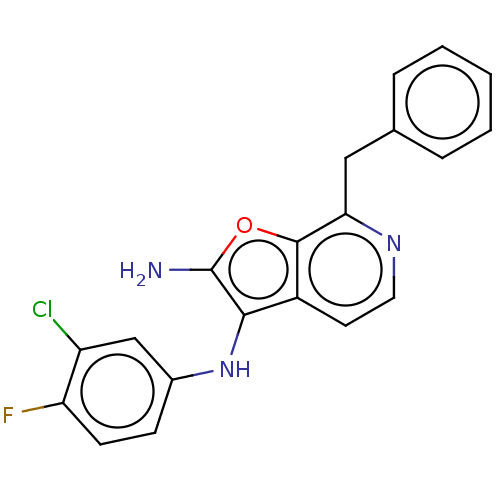
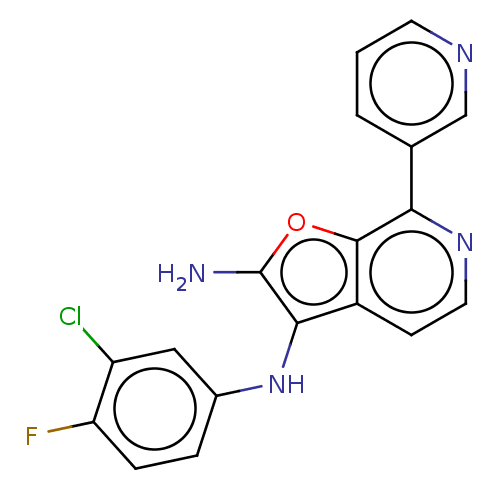
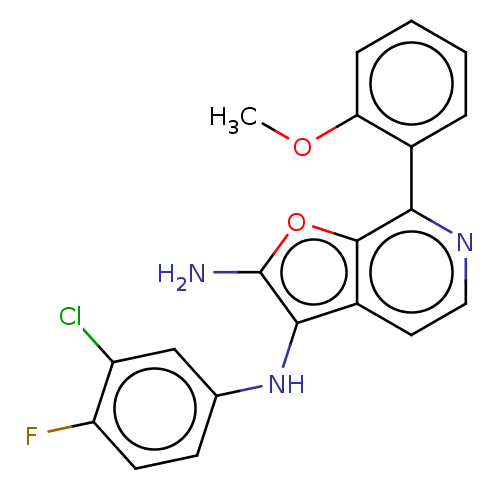
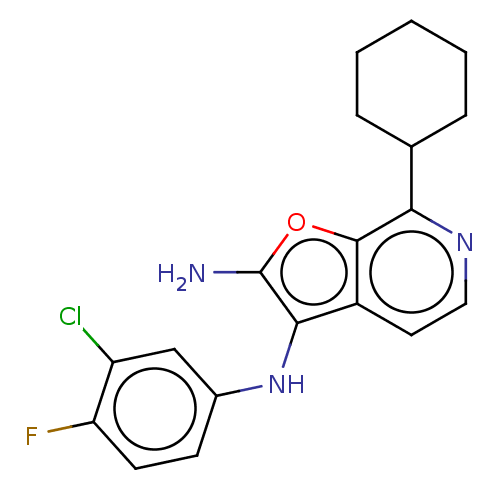
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Sphaera Pharma
Curated by ChEMBL
Sphaera Pharma
Curated by ChEMBL
Affinity DataIC50: 60nMAssay Description:Inhibition of human recombinant PI3K-alpha using phosphatidylinositol biphosphate as substrate preincubated for 15 mins followed by substrate additio...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Sphaera Pharma
Curated by ChEMBL
Sphaera Pharma
Curated by ChEMBL
Affinity DataIC50: 80nMAssay Description:Inhibition of human recombinant PI3K-alpha using phosphatidylinositol biphosphate as substrate preincubated for 15 mins followed by substrate additio...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Sphaera Pharma
Curated by ChEMBL
Sphaera Pharma
Curated by ChEMBL
Affinity DataIC50: 80nMAssay Description:Inhibition of human recombinant PI3K-alpha using phosphatidylinositol biphosphate as substrate preincubated for 15 mins followed by substrate additio...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Sphaera Pharma
Curated by ChEMBL
Sphaera Pharma
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibition of human recombinant PI3K-alpha using phosphatidylinositol biphosphate as substrate preincubated for 15 mins followed by substrate additio...More data for this Ligand-Target Pair
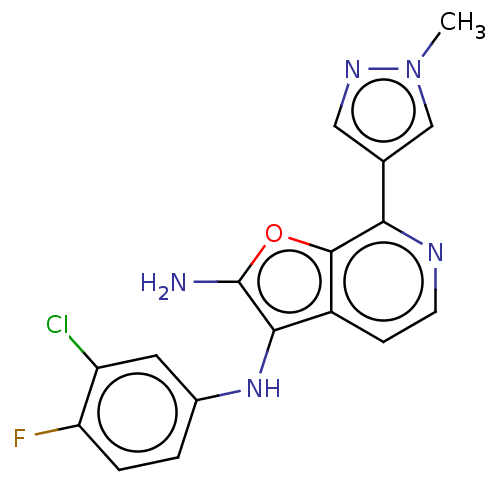
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: <200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair