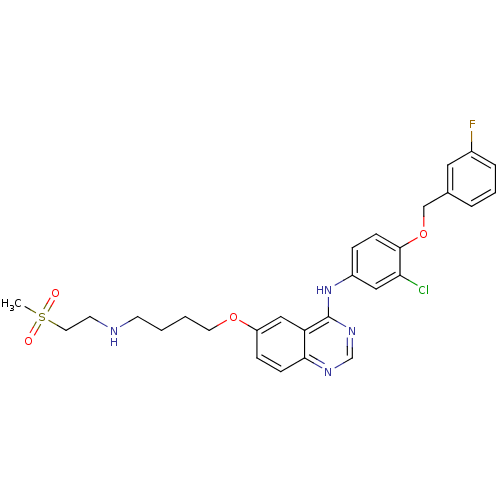
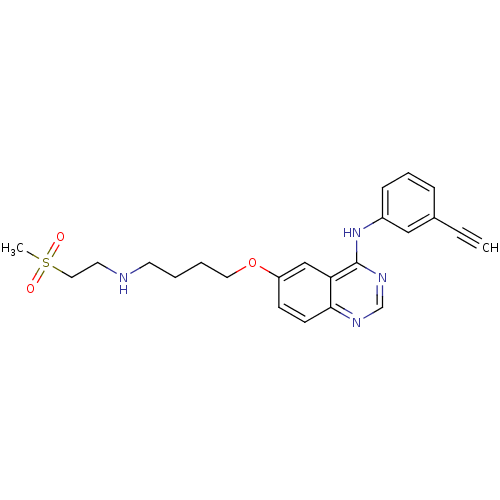
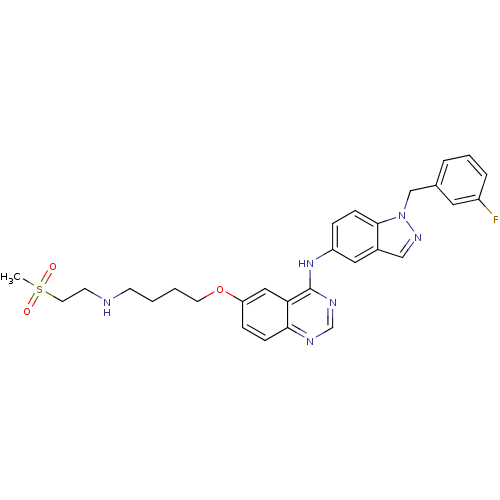
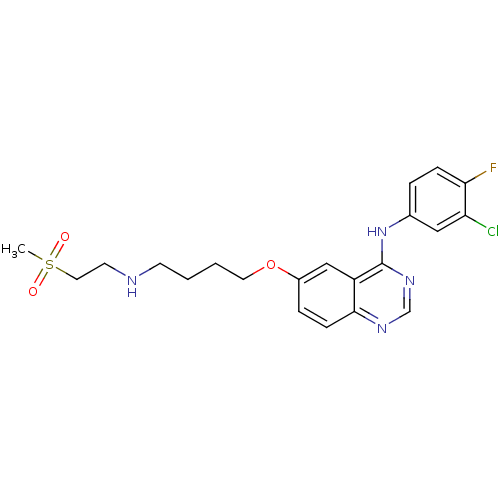
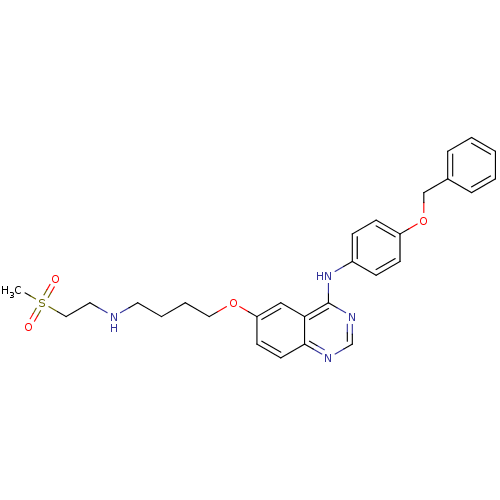
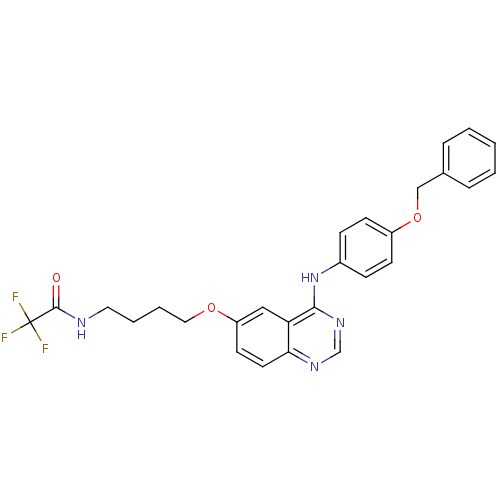
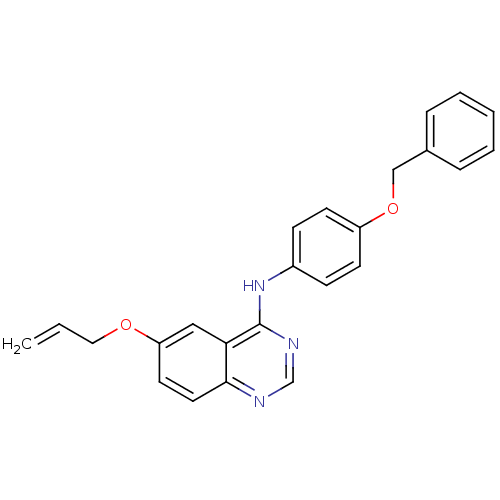
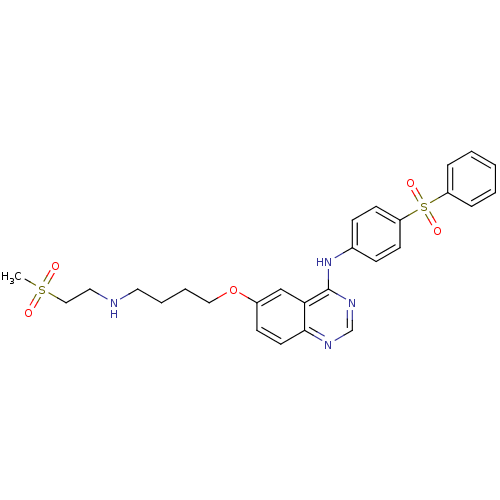
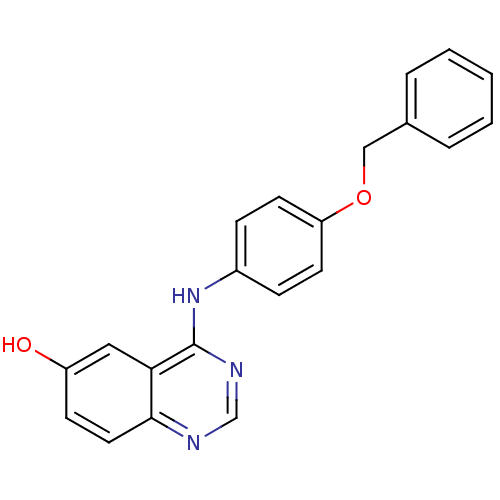
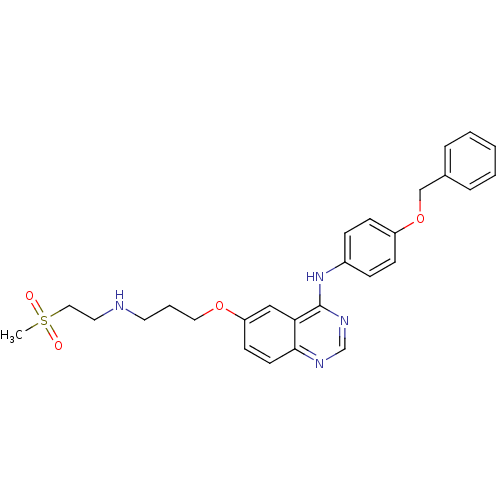
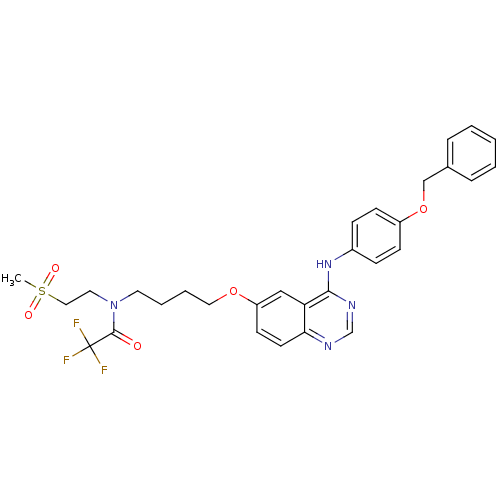
Report error Found 26 Enz. Inhib. hit(s) with all data for entry = 725
Affinity DataIC50: 10nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 11nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 19nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 24nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 27nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 68nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 74nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 74nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 79nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 93nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 95nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 98nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 104nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 117nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 147nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 210nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 234nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 234nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 263nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 263nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 309nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 347nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 830nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 977nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair
Affinity DataIC50: 1.78E+3nMpH: 7.5 T: 2°CAssay Description:Enzymatic reactions were initiated by adding kinase to the reaction mixture containing ATP, [gamma-33P] ATP, peptide substrate and test inhibitor com...More data for this Ligand-Target Pair