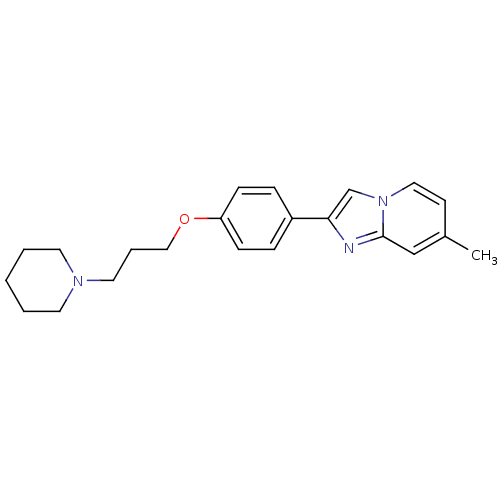
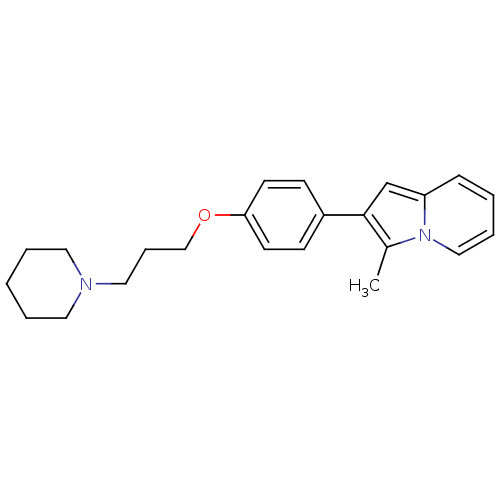
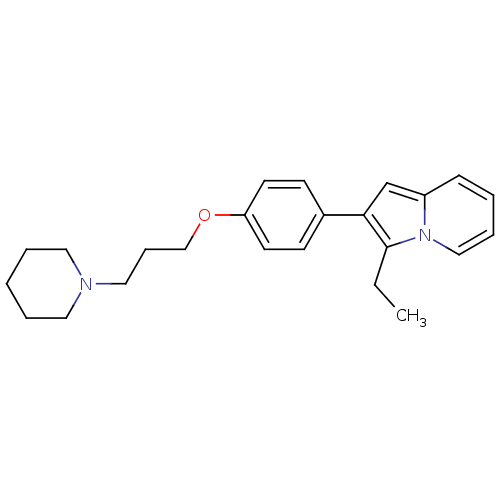
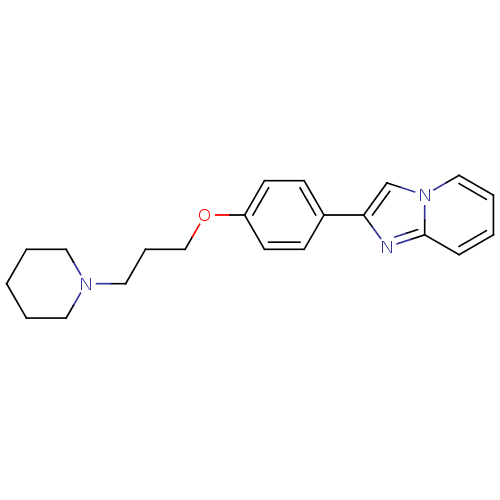
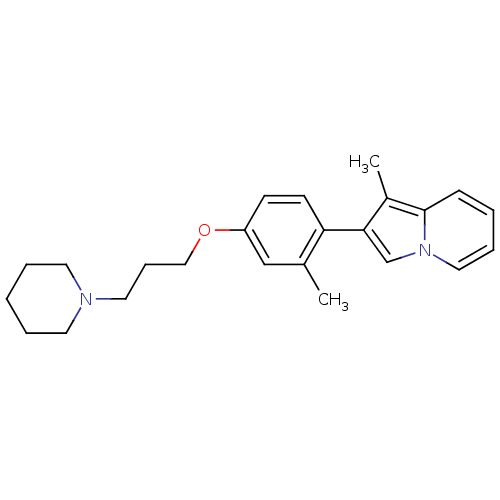
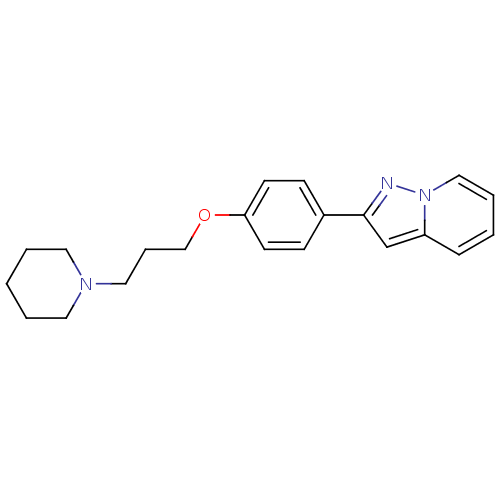
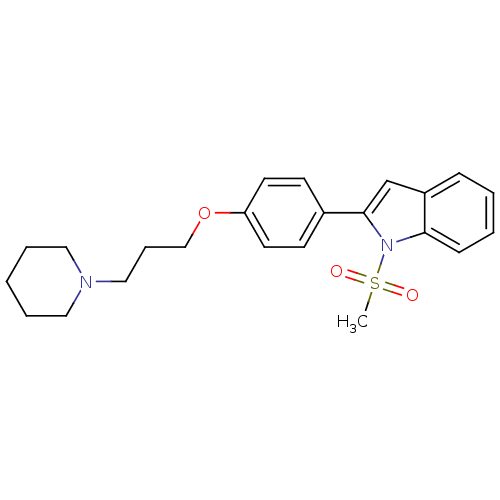
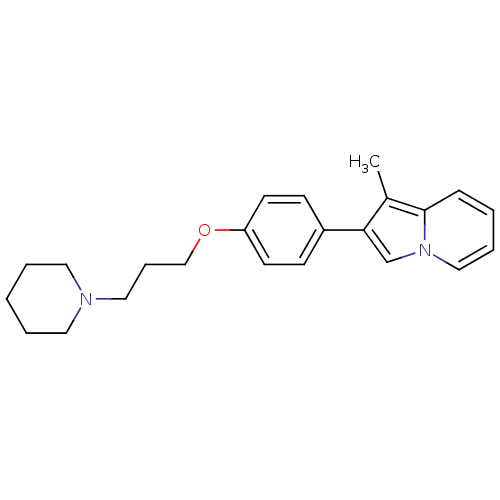
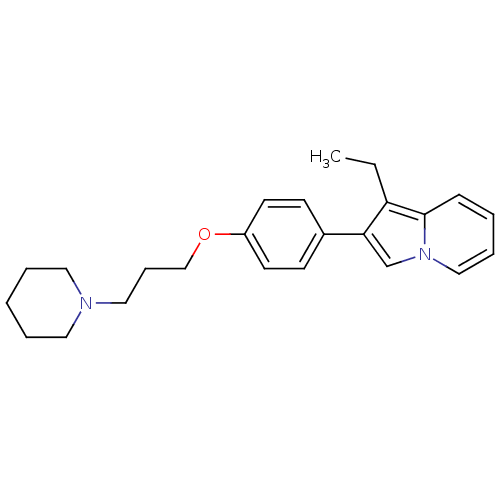
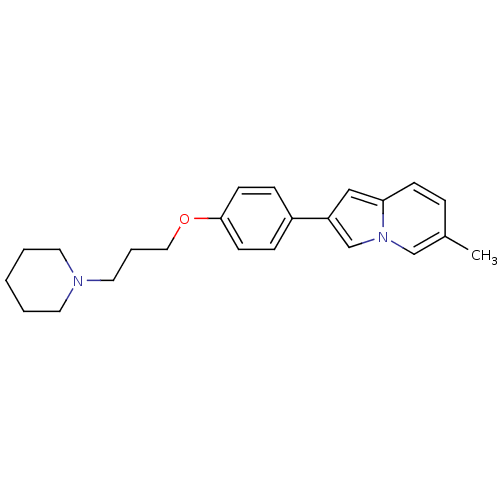
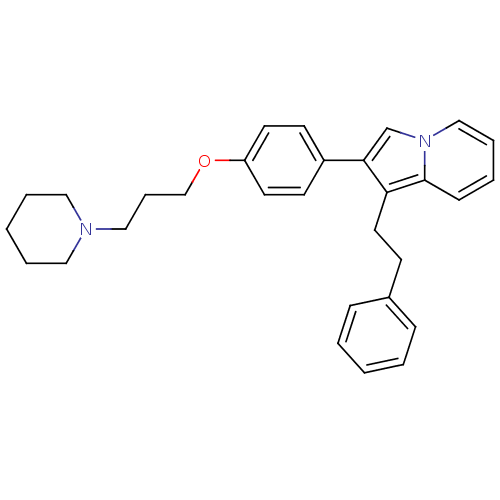
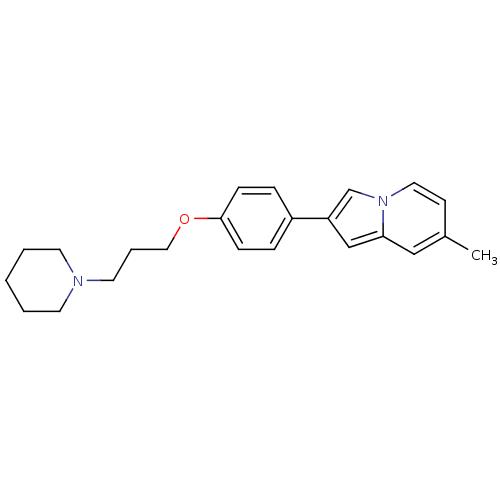
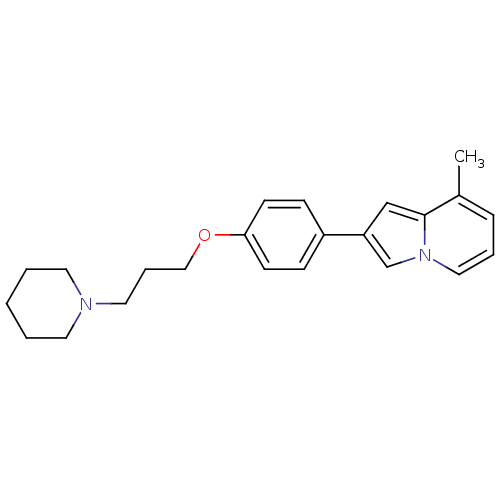
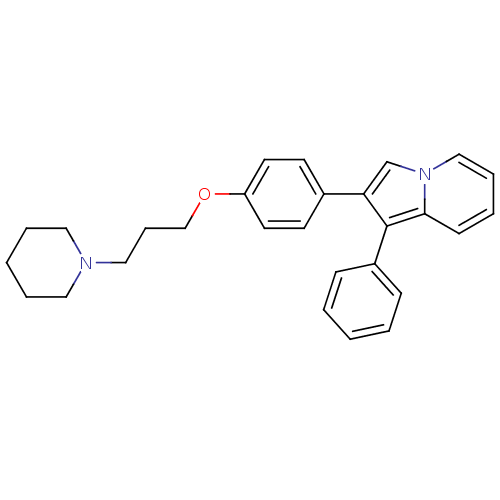
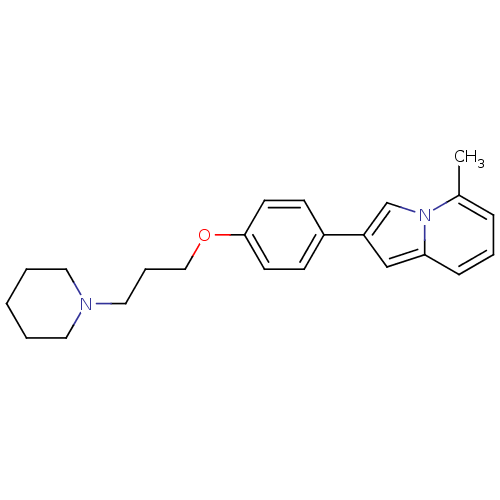
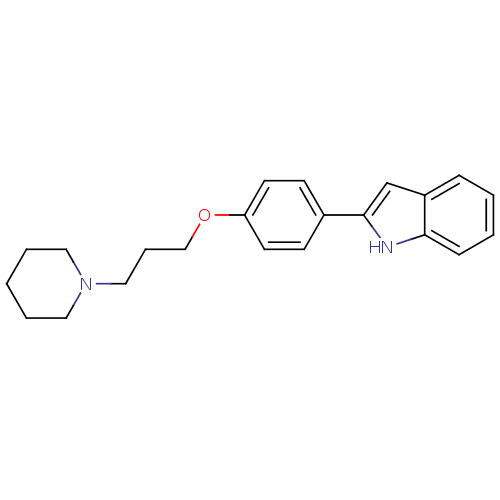
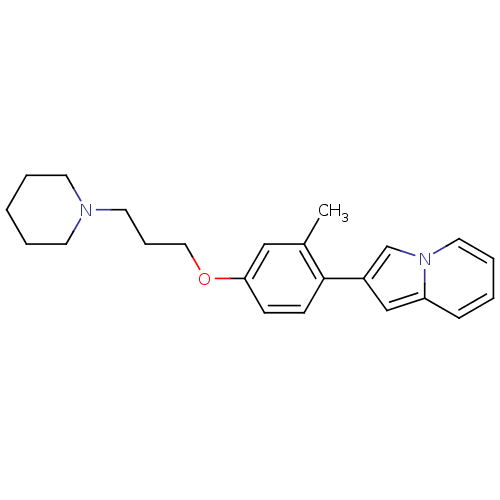
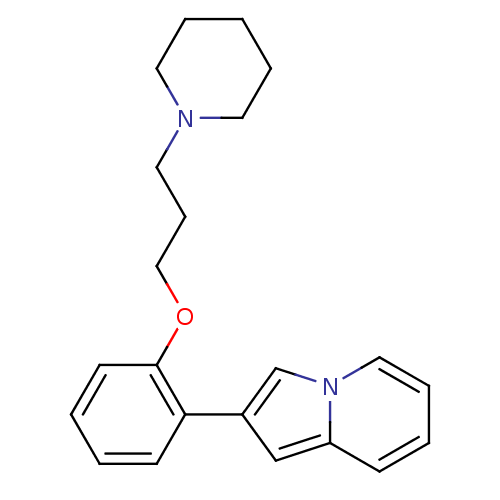
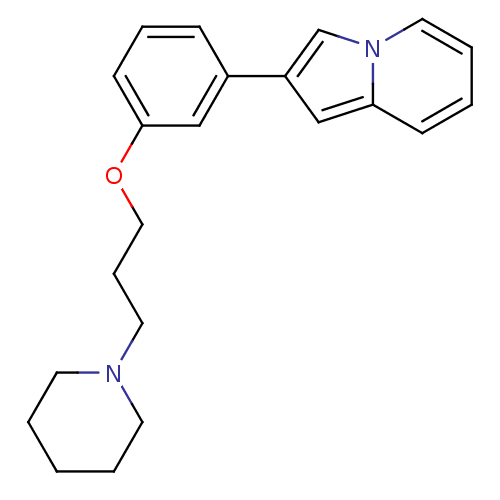
Report error Found 20 Enz. Inhib. hit(s) with all data for entry = 50013221
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 2nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 2nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 5nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 6nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 11nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 11nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 13nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 16nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 16nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 19nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 28nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 37nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 40nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 40nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 40nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 46nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 47nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 152nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 236nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 308nMAssay Description:Binding affinity towards human Histamine H3 receptor using [3H]N-methyl-histamine as radioligandMore data for this Ligand-Target Pair