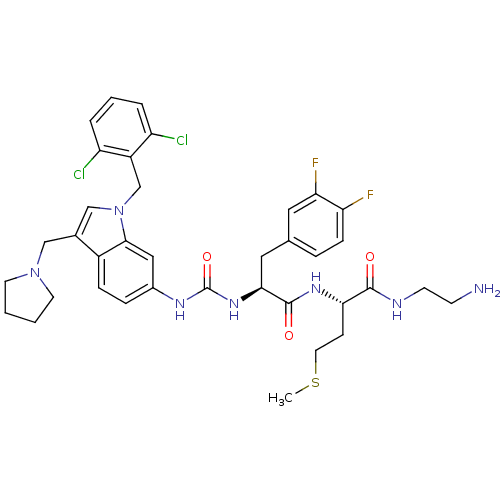
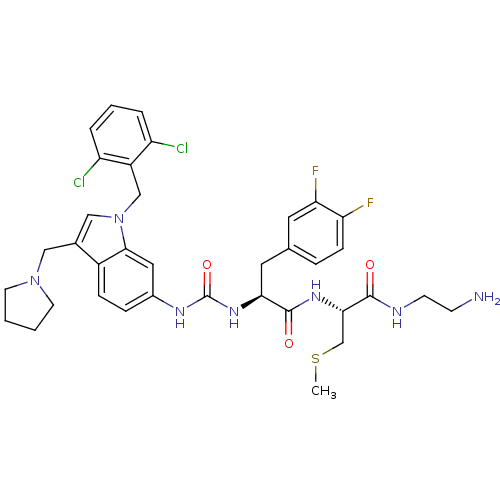
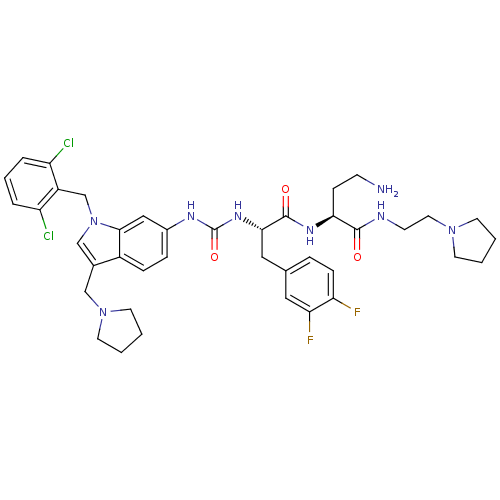
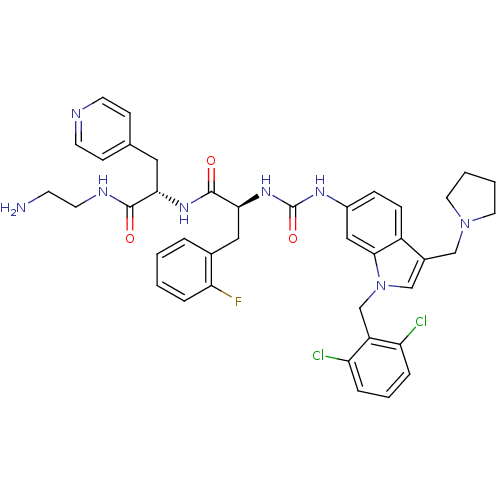
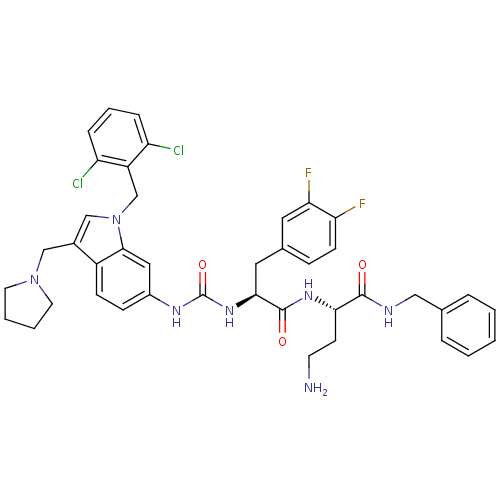
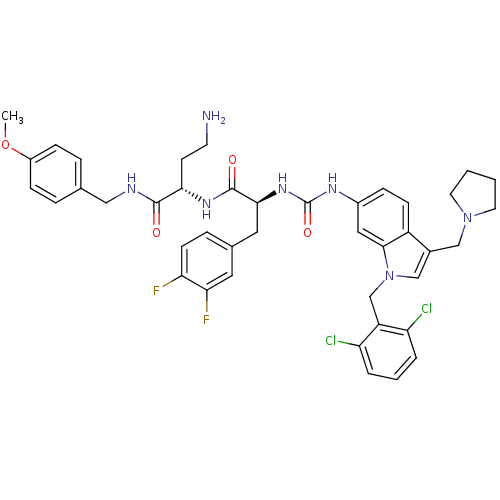
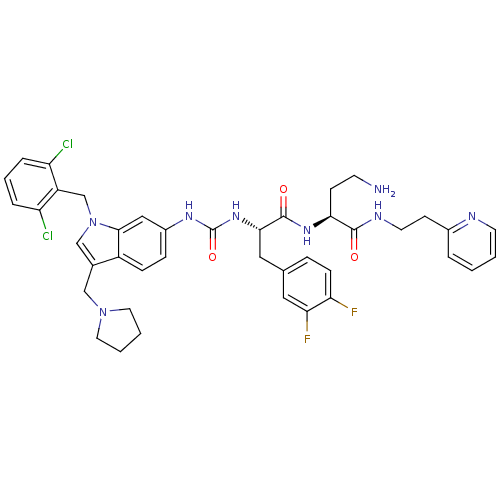
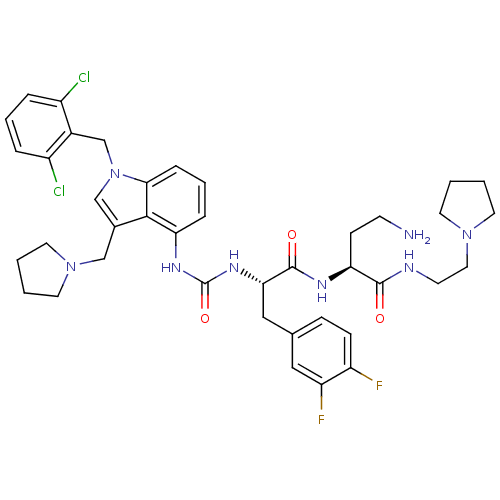
Report error Found 18 Enz. Inhib. hit(s) with all data for entry = 50013344
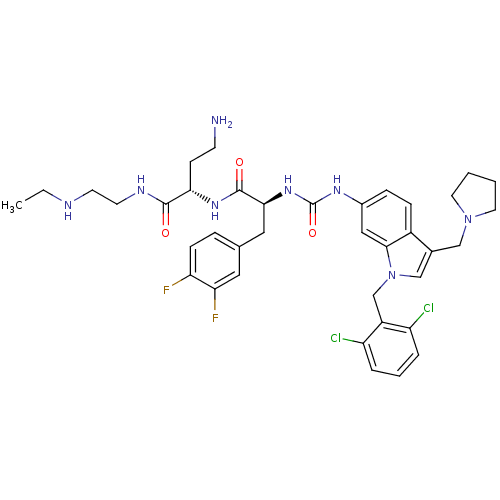
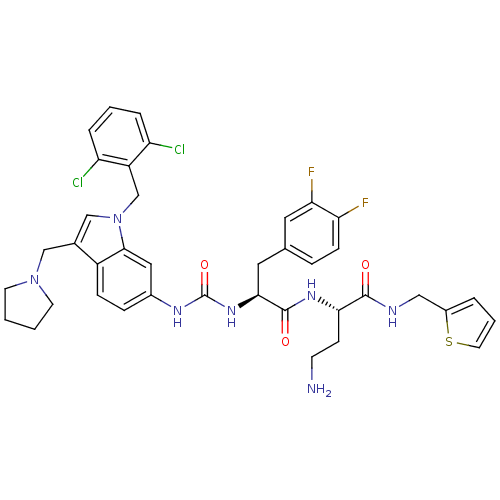
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
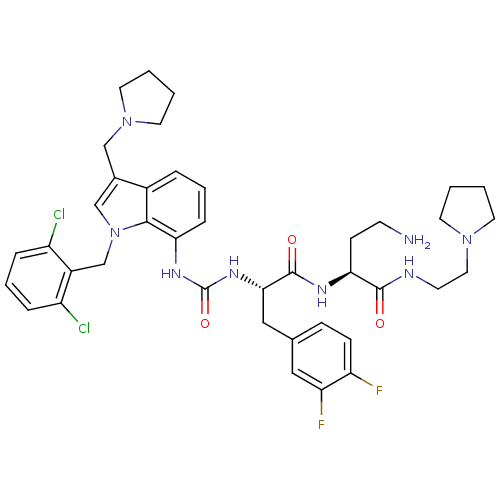
Affinity DataIC50: 25nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
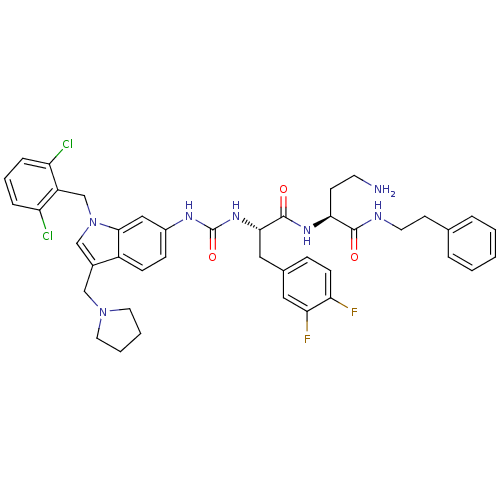
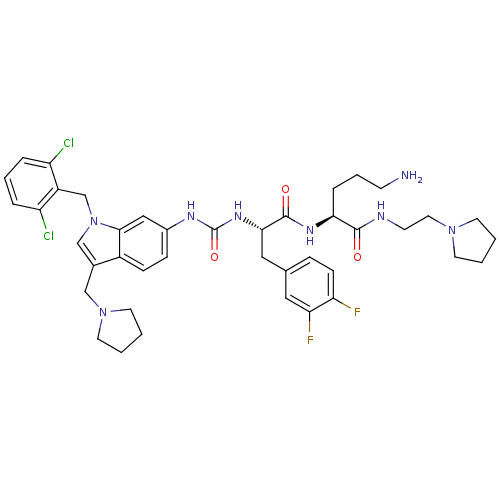
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 35nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
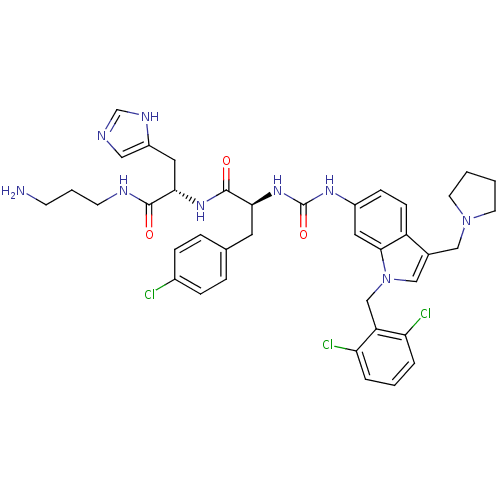
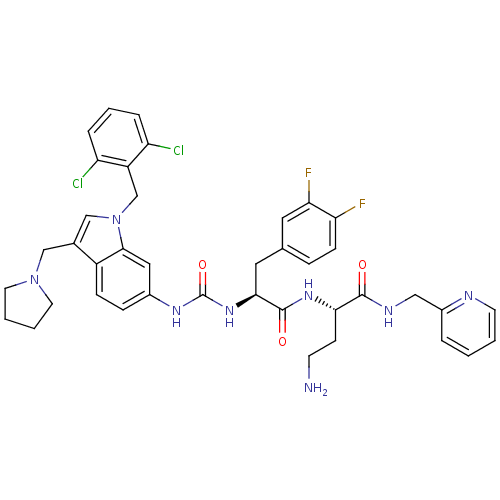
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 60nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
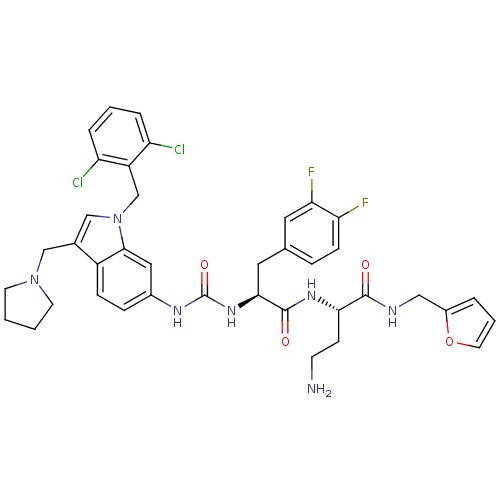
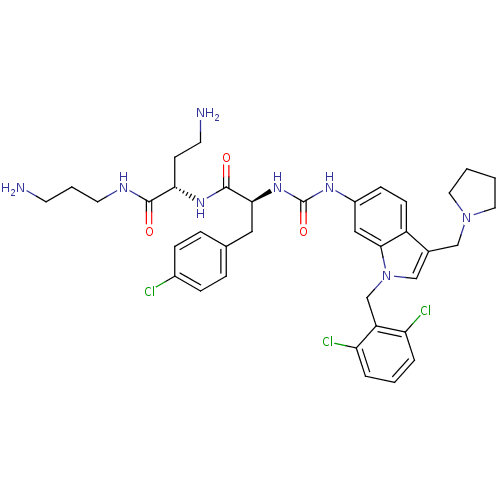
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 70nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 70nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 90nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 120nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 130nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 160nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 230nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 310nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 330nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 430nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 440nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 480nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 500nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 2.00E+3nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair
TargetProteinase-activated receptor 1(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 2.20E+4nMAssay Description:Ability to inhibit the binding of [3H]S-(p-F-Phe)-homoarginine-L-homoarginine-KY-NH2 to thrombin receptor on the membranes of CHRF-288-11 cellsMore data for this Ligand-Target Pair