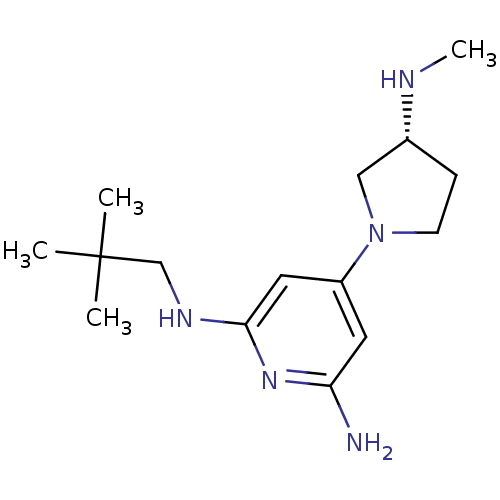
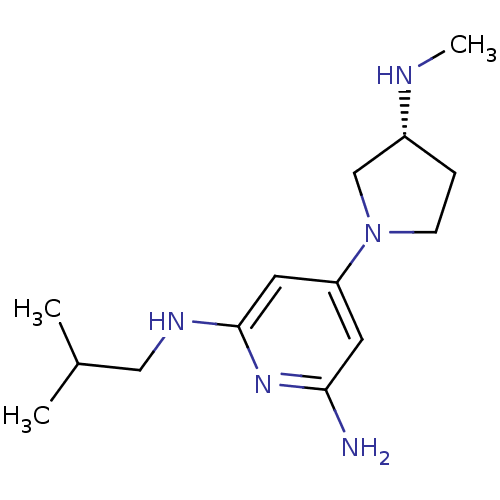
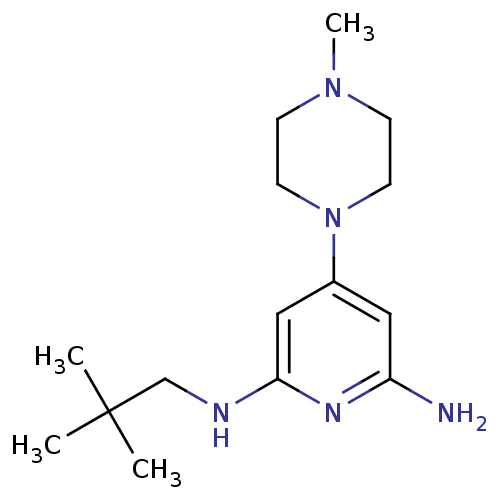
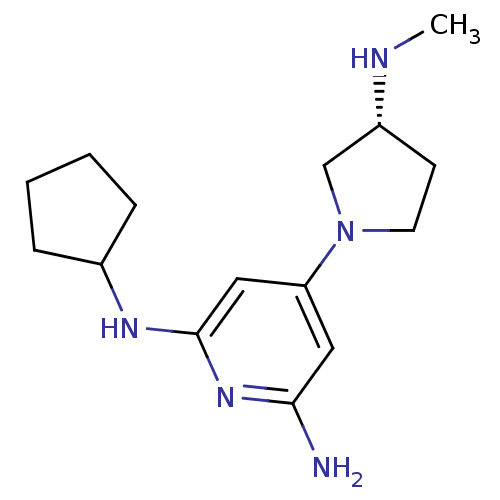
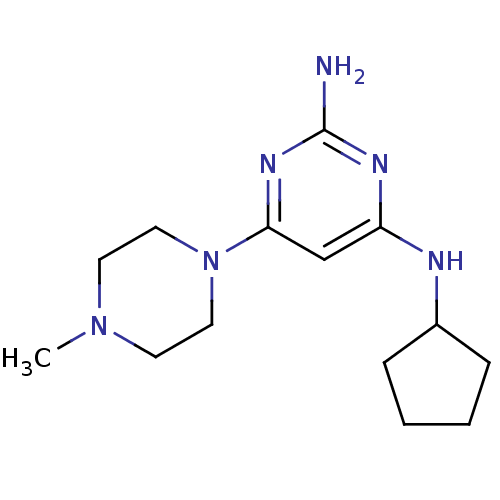
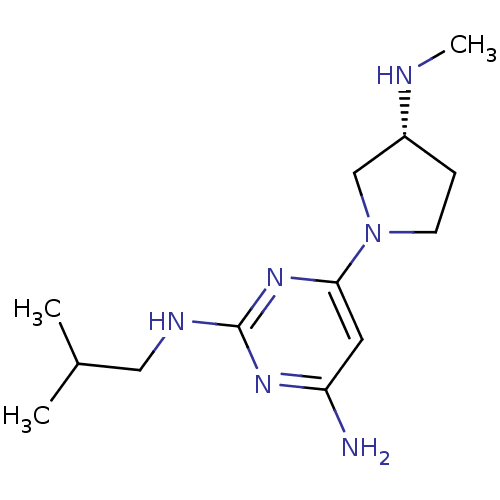
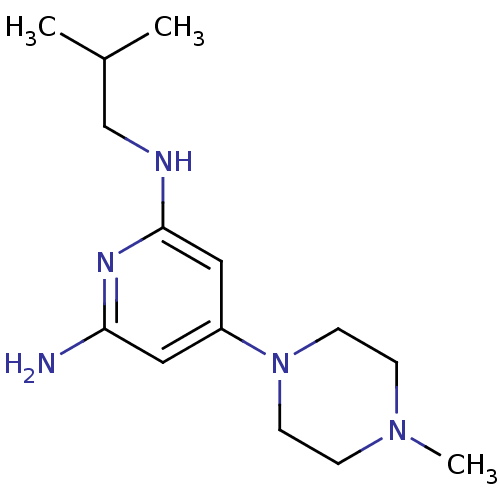
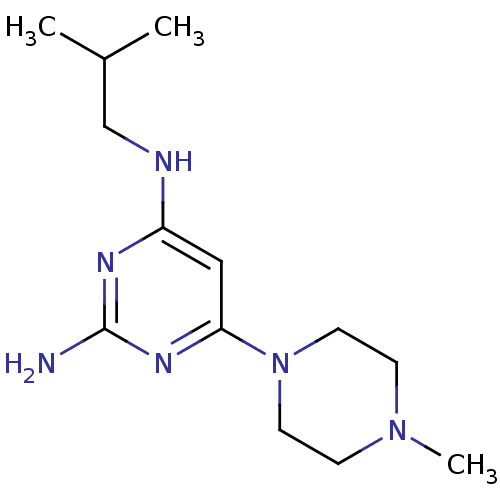
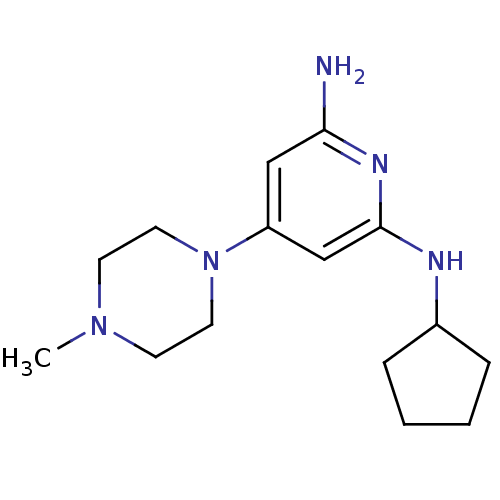
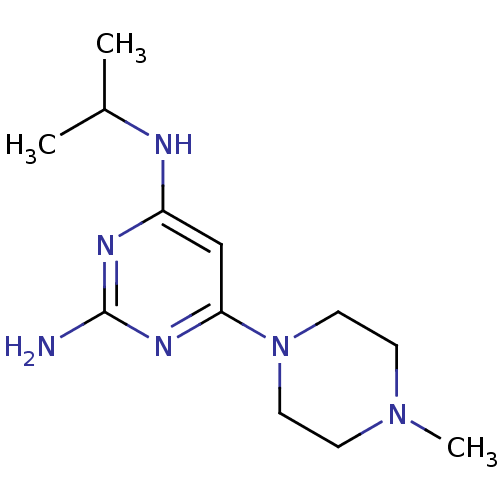
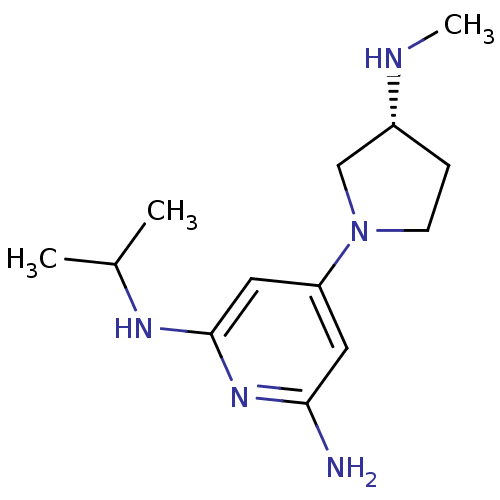
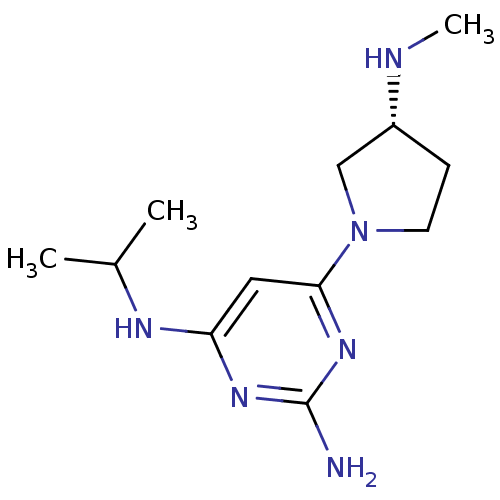
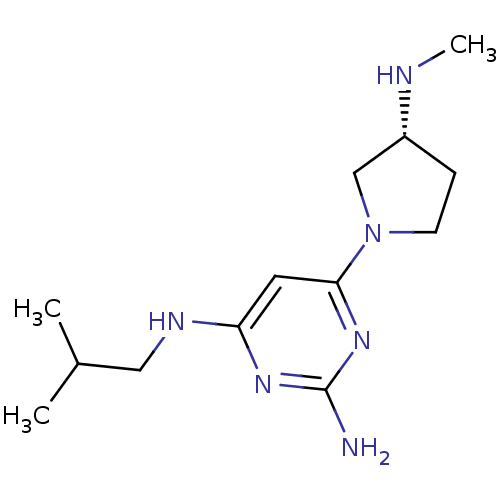
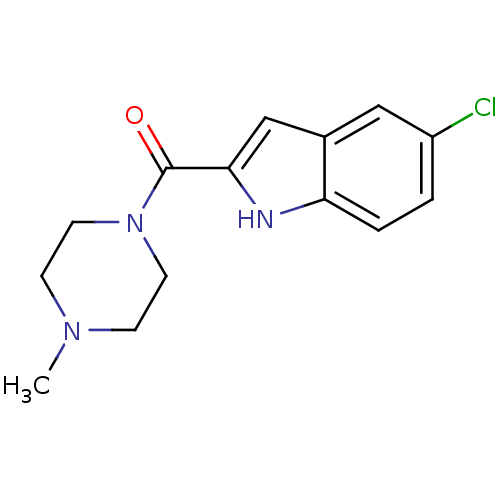
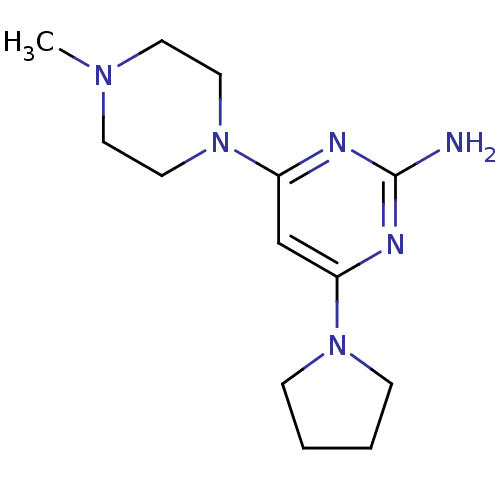
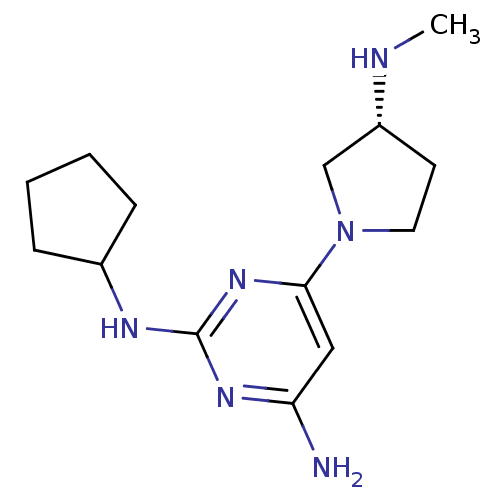
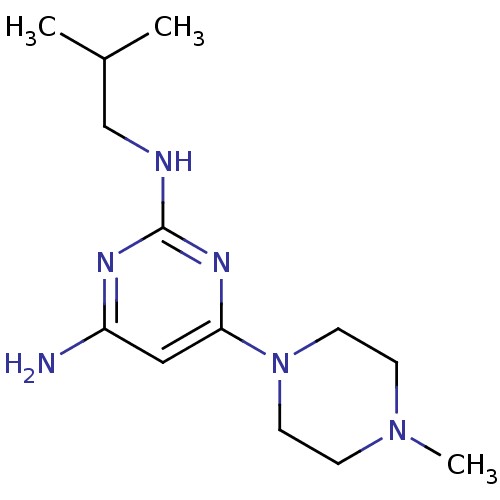
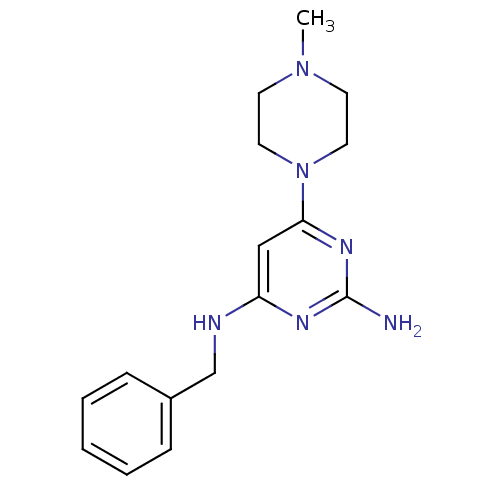
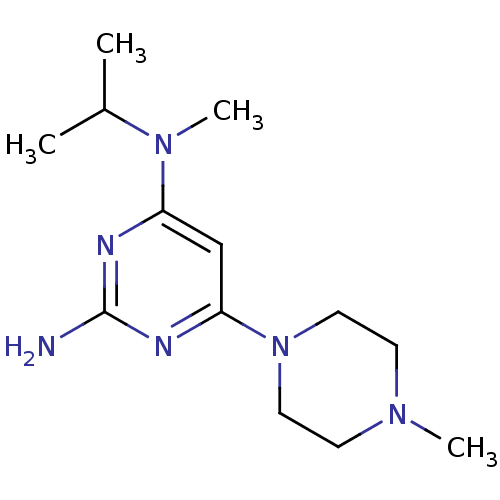
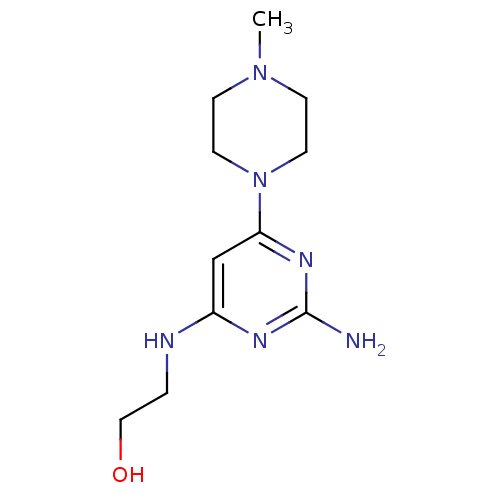
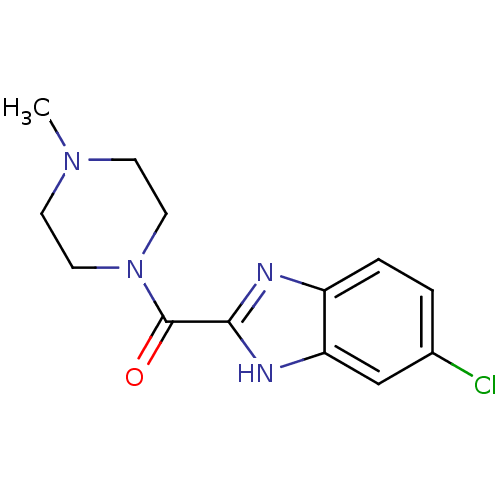
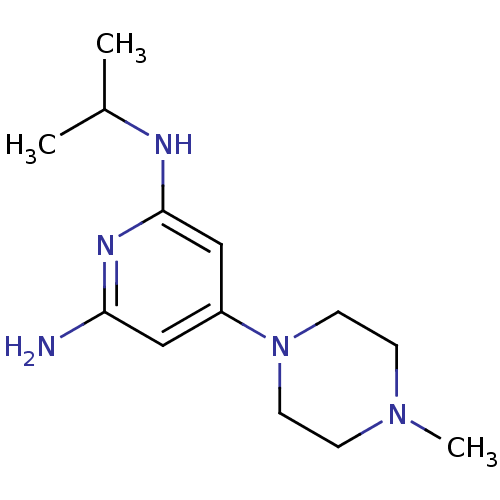
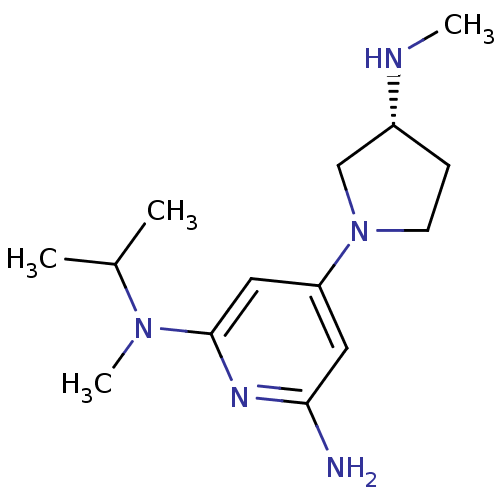
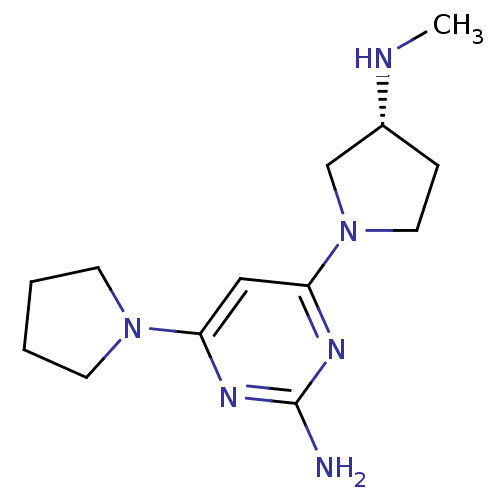
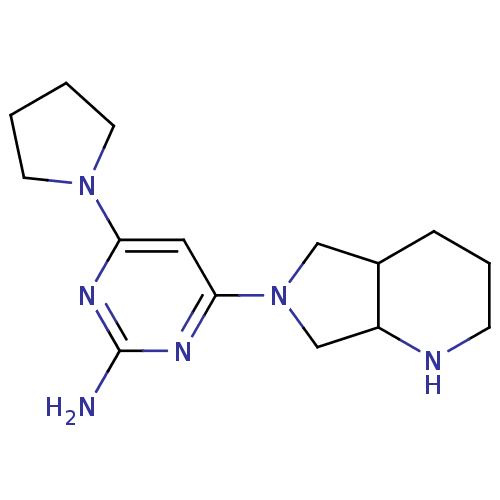
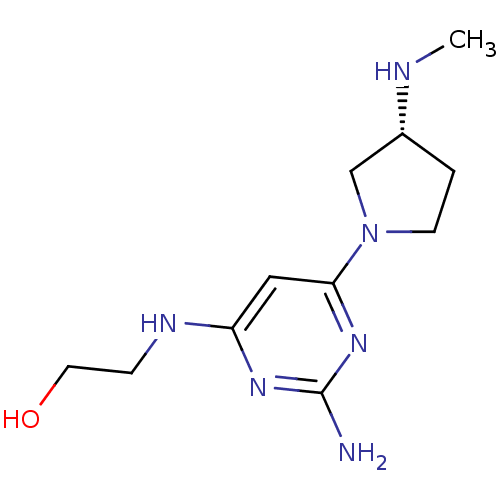
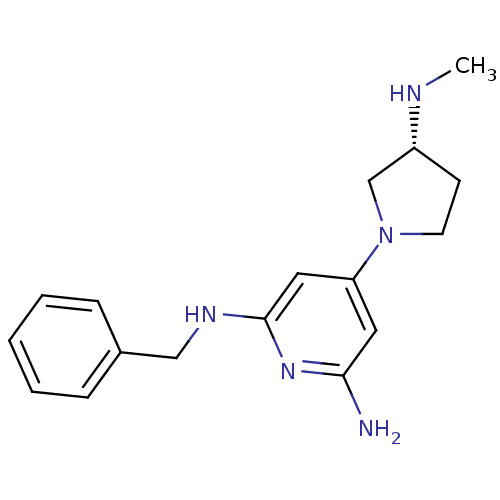
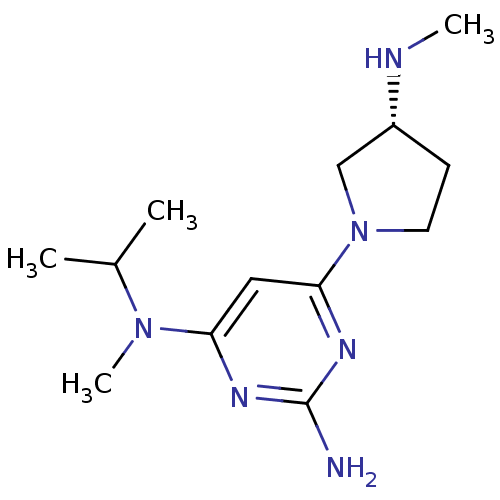
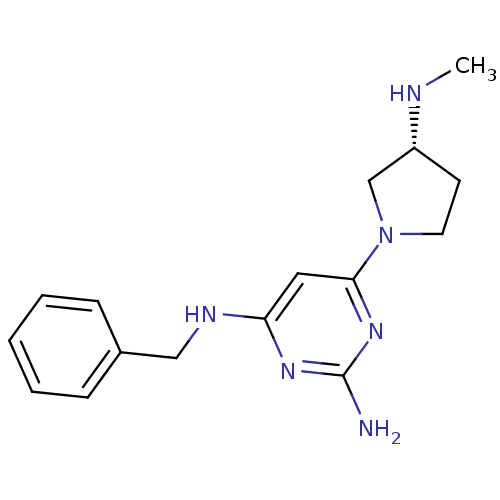
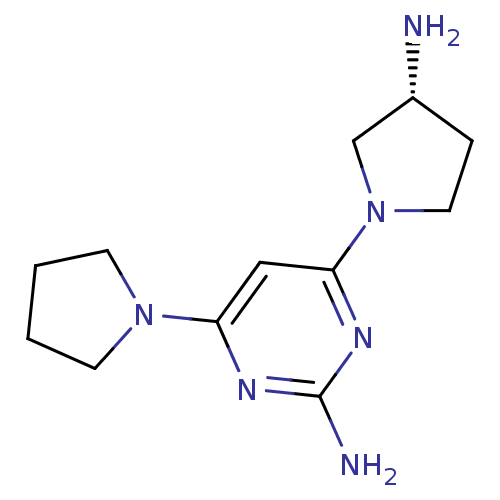
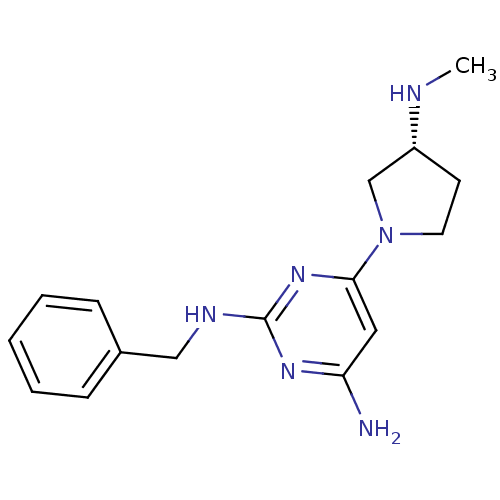
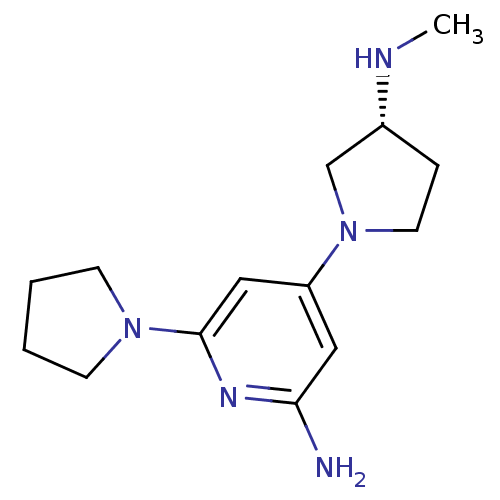
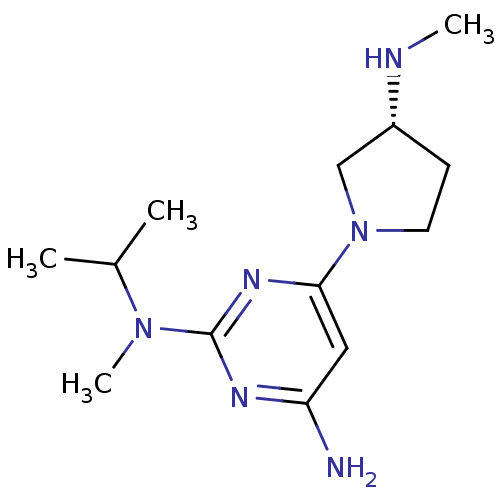
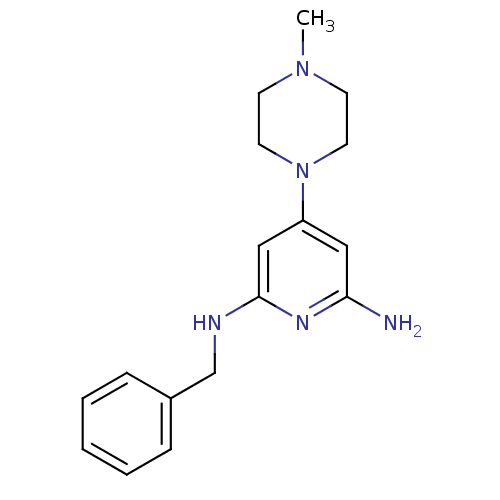
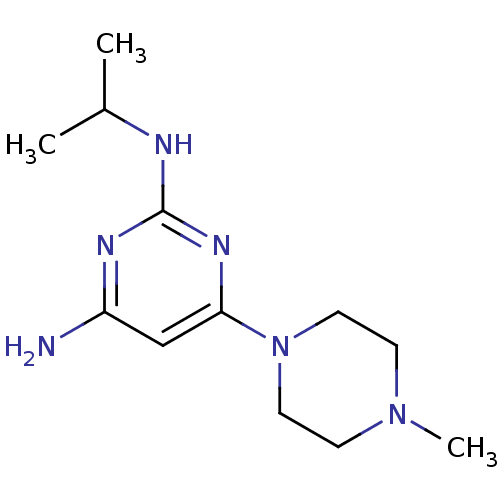
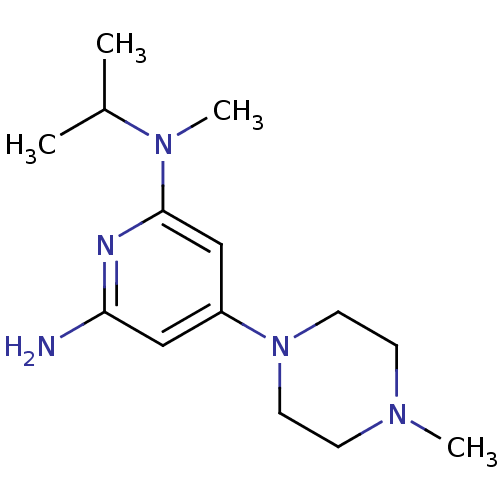
Report error Found 71 Enz. Inhib. hit(s) with all data for entry = 50033298
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
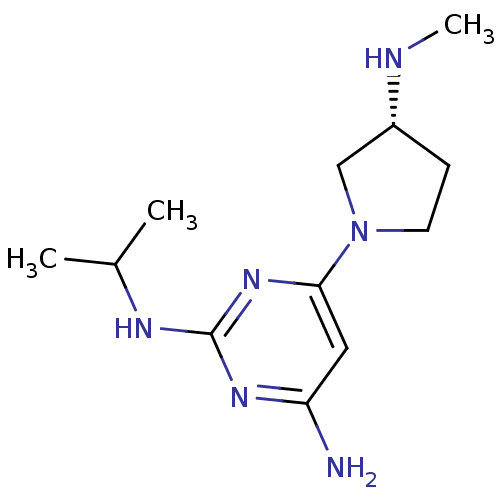
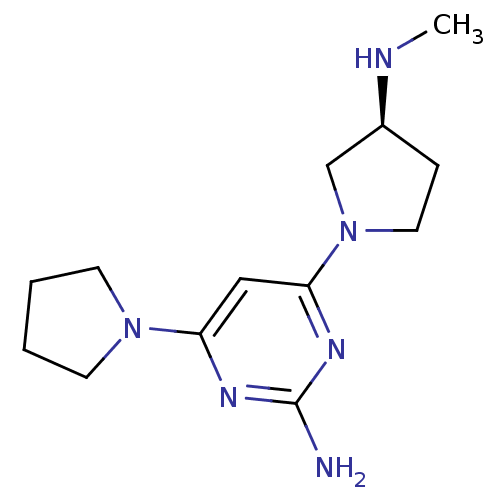
Affinity DataKi: 0.0400nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
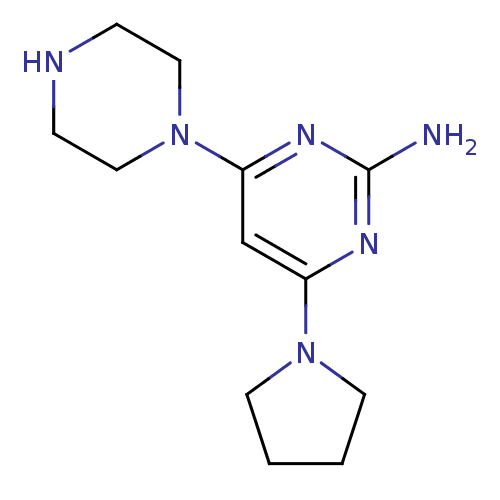
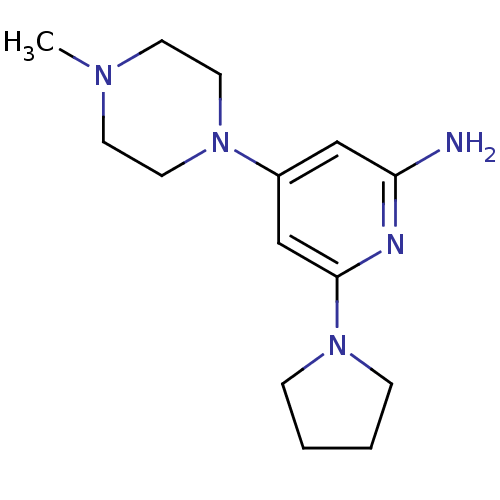
Affinity DataKi: 0.200nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
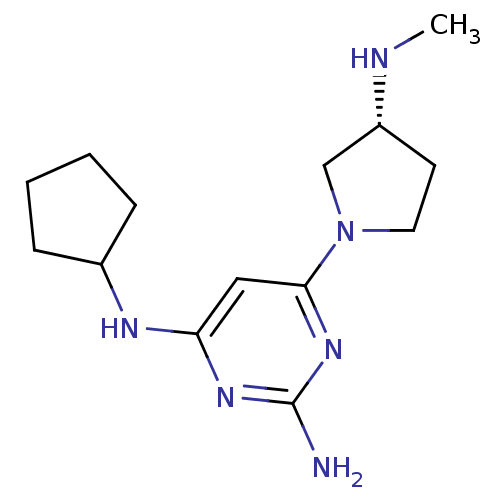
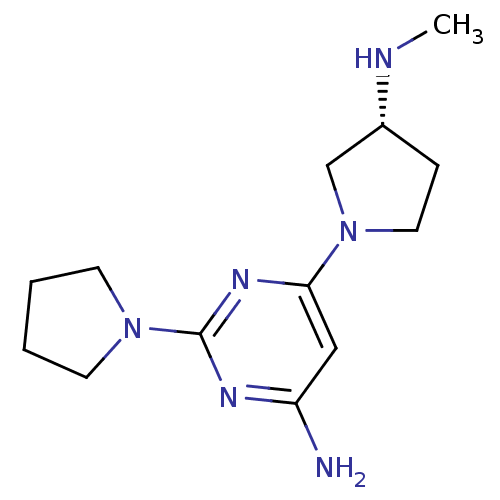
Affinity DataKi: 0.300nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
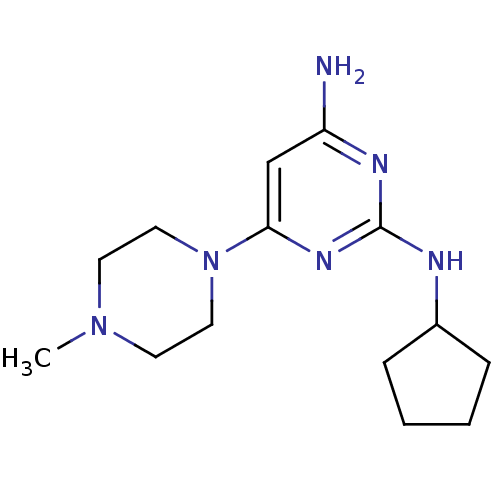
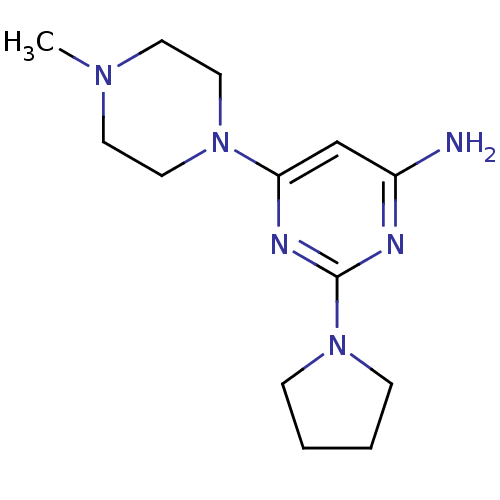
Affinity DataKi: 0.900nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 1nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 1nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 2nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 2nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 3nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 3nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 3nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 3nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 4nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 4nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 6nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 6nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 6nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 8nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 9nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 10nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataEC50: 11nMAssay Description:Agonist activity at human histamine H4 receptor expressed in human SK-N-MC cells assessed as effect on forskolin-induced cAMP accumulation after 6 hr...More data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataEC50: 12nMAssay Description:Agonist activity at human histamine H4 receptor expressed in human SK-N-MC cells assessed as effect on forskolin-induced cAMP accumulation after 6 hr...More data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 15nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataEC50: 16nMAssay Description:Agonist activity at human histamine H4 receptor expressed in human SK-N-MC cells assessed as effect on forskolin-induced cAMP accumulation after 6 hr...More data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 20nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 24nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 25nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 26nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 28nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 29nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 36nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 40nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 44nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 50nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataEC50: 50nMAssay Description:Agonist activity at human histamine H4 receptor expressed in human SK-N-MC cells assessed as effect on forskolin-induced cAMP accumulation after 6 hr...More data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 51nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 58nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 64nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 69nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 77nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 83nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataEC50: 86nMAssay Description:Agonist activity at human histamine H4 receptor expressed in human SK-N-MC cells assessed as effect on forskolin-induced cAMP accumulation after 6 hr...More data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 100nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataEC50: 101nMAssay Description:Agonist activity at human histamine H4 receptor expressed in human SK-N-MC cells assessed as effect on forskolin-induced cAMP accumulation after 6 hr...More data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 127nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 132nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 140nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 148nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 162nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 189nMAssay Description:Displacement of [3H]histamine from human histamine H4 receptorMore data for this Ligand-Target Pair