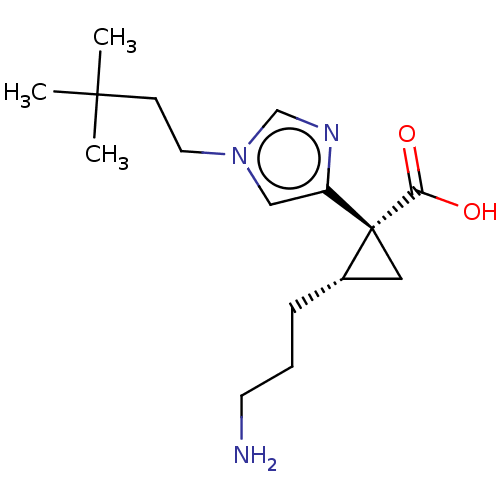
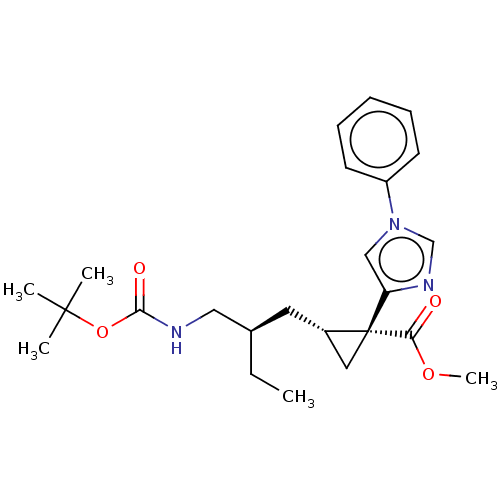
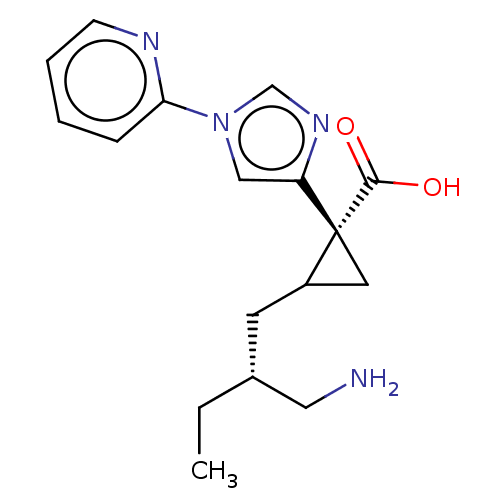
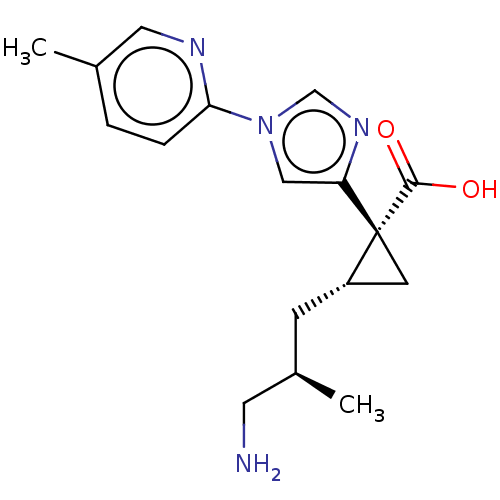
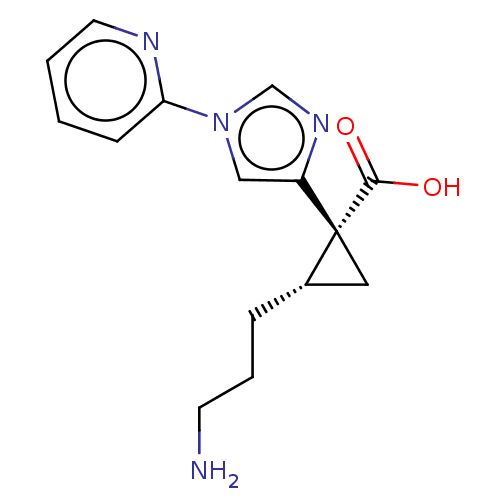
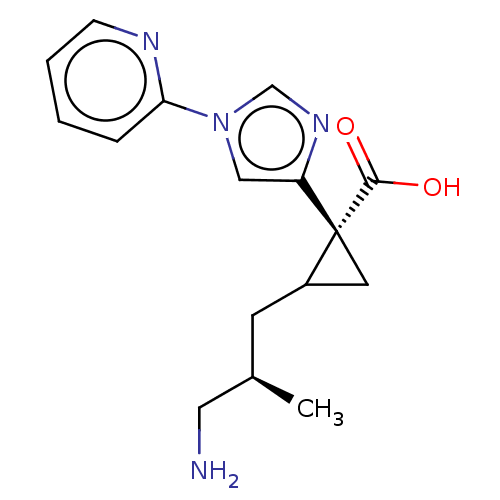
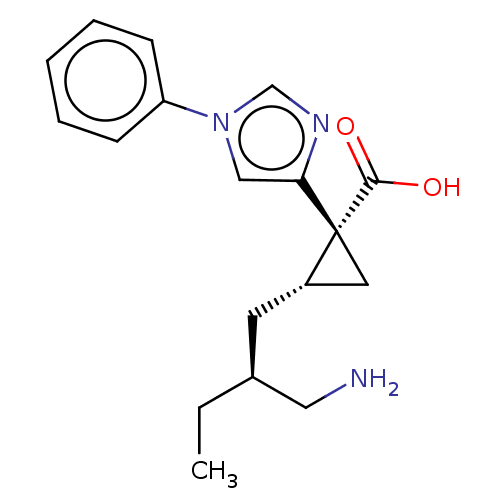
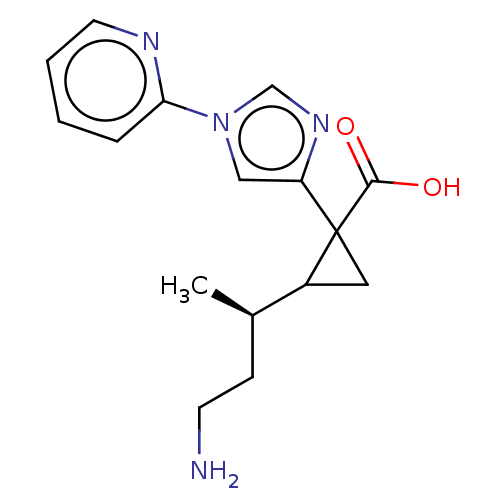
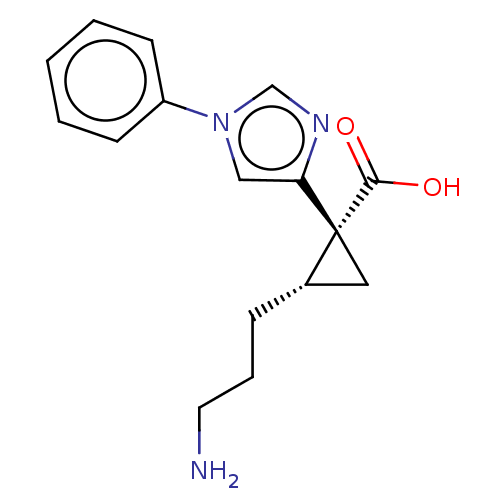
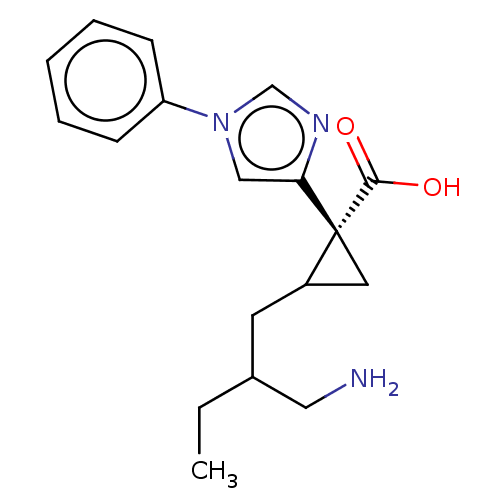
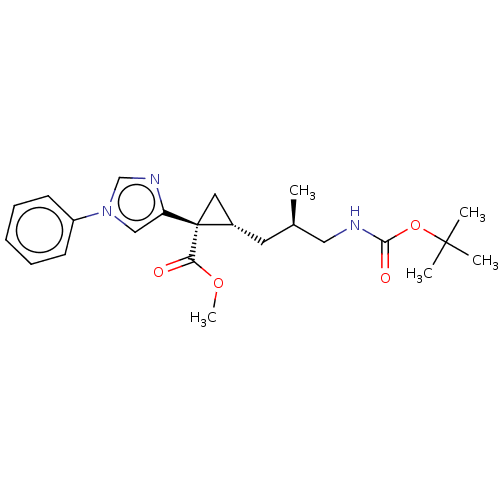
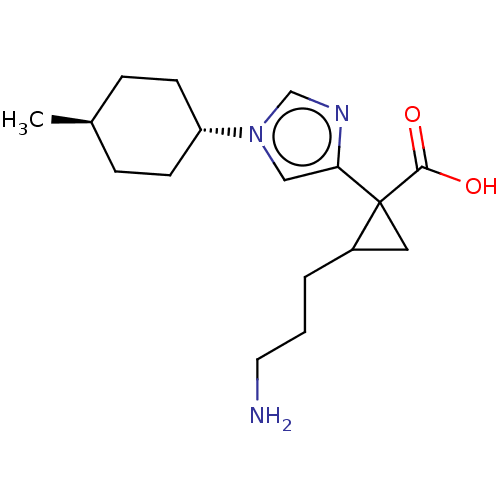
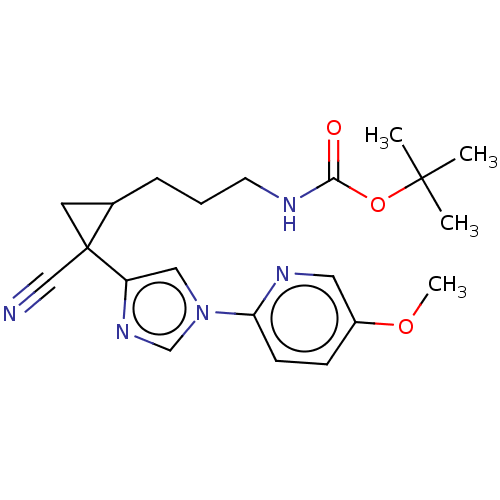
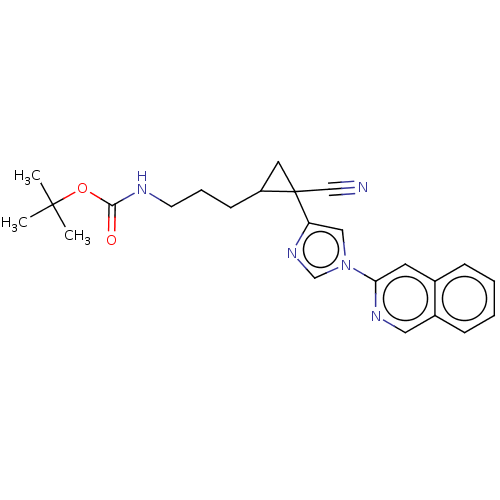
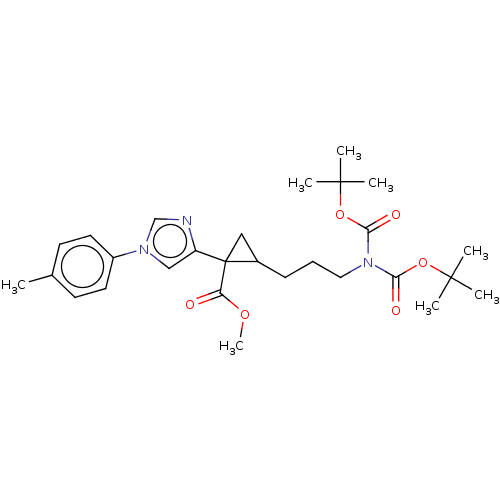
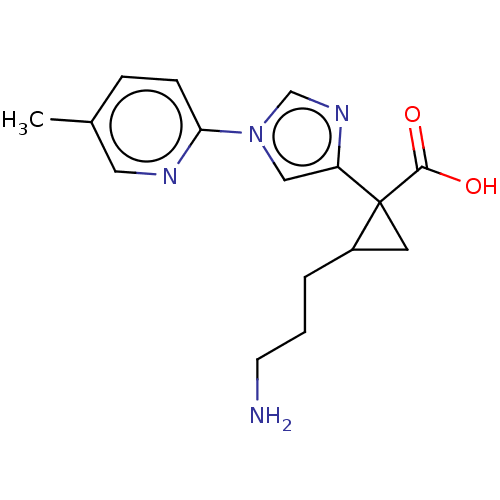
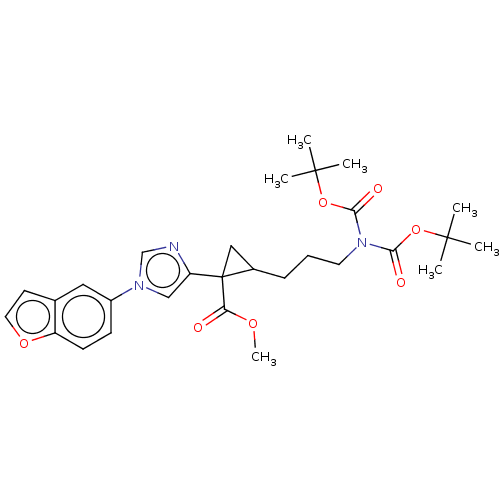
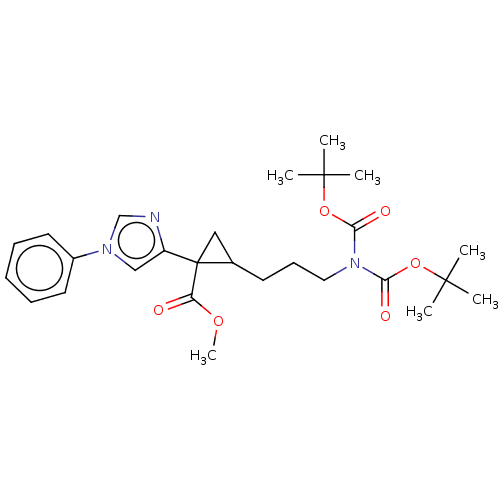
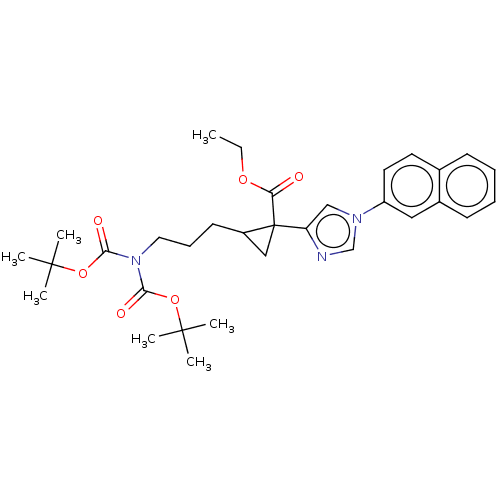
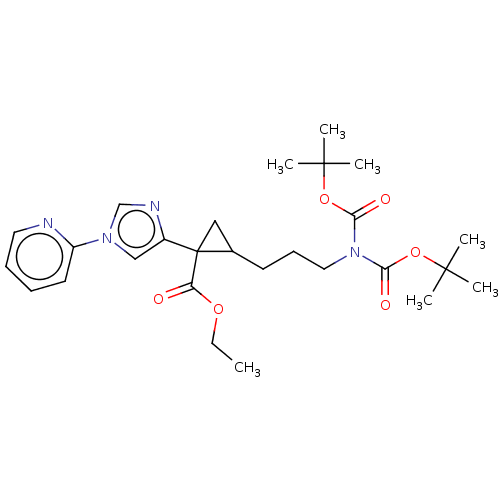
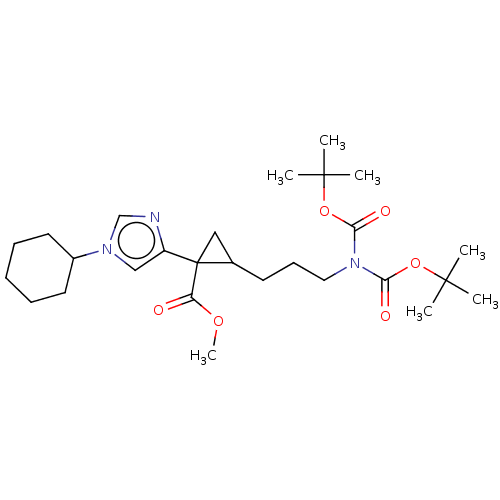
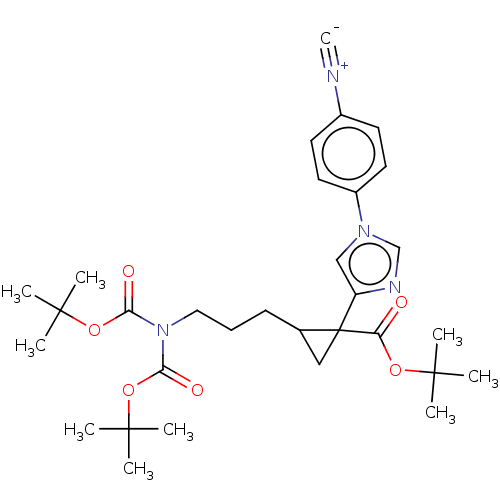
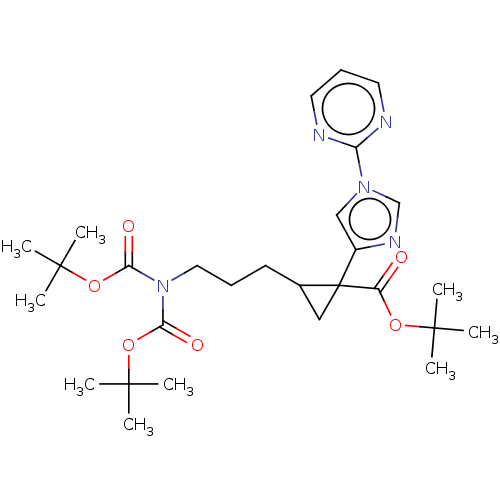
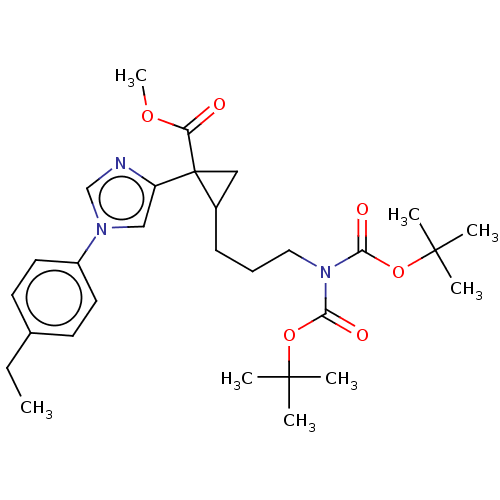
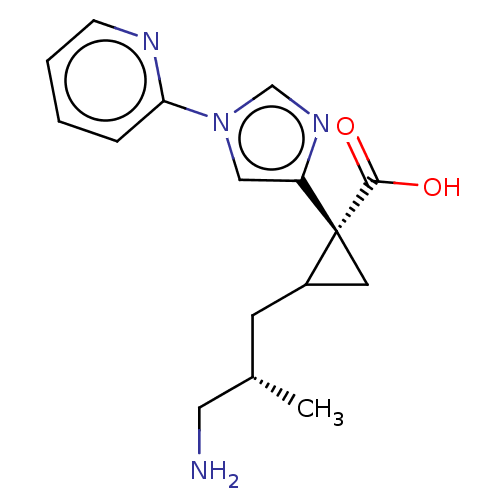
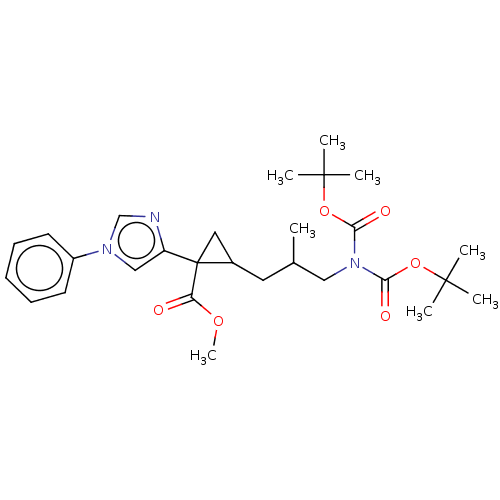
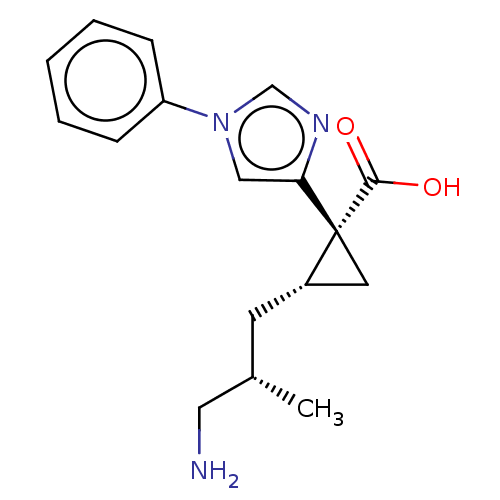
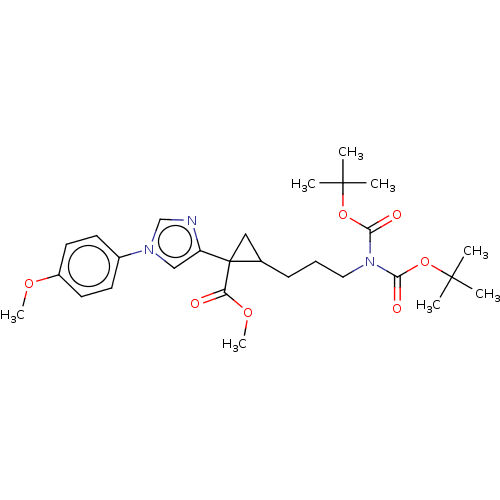
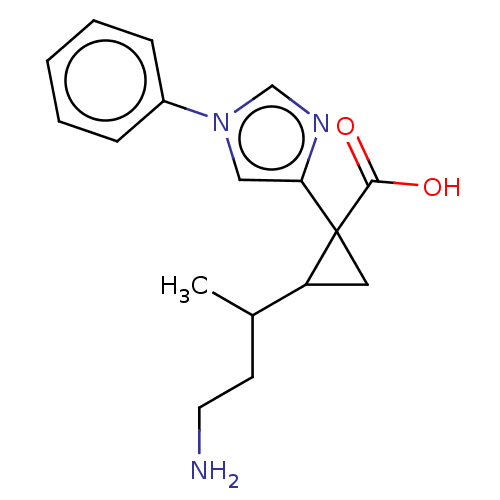
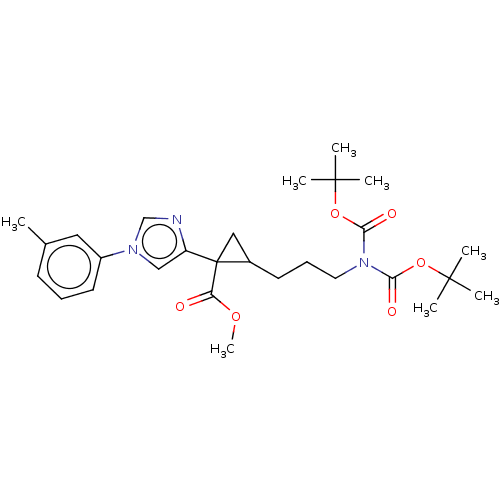
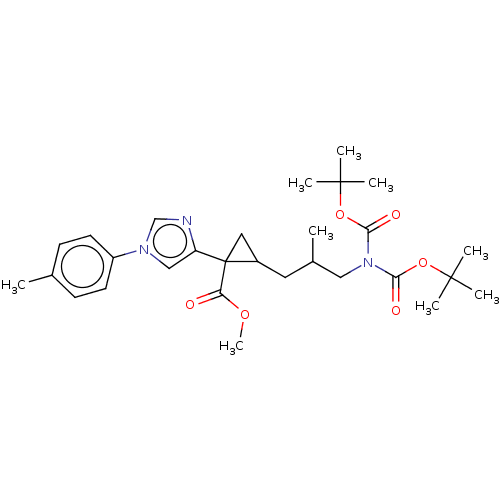
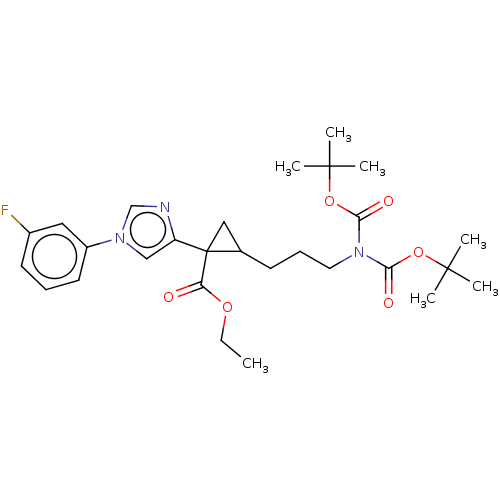
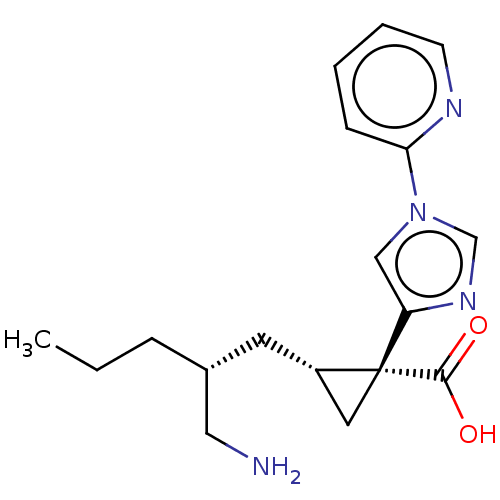
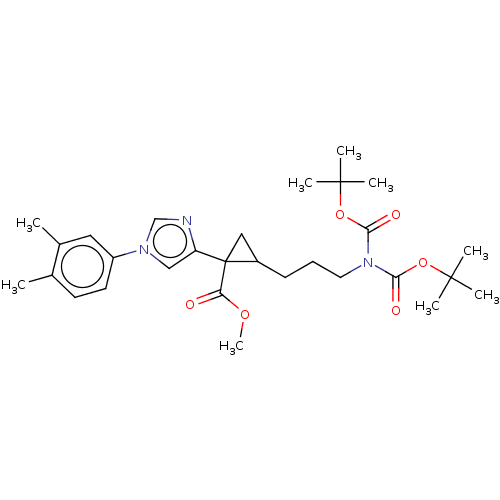
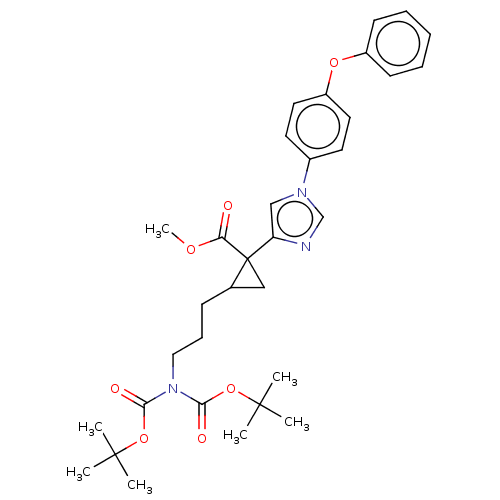
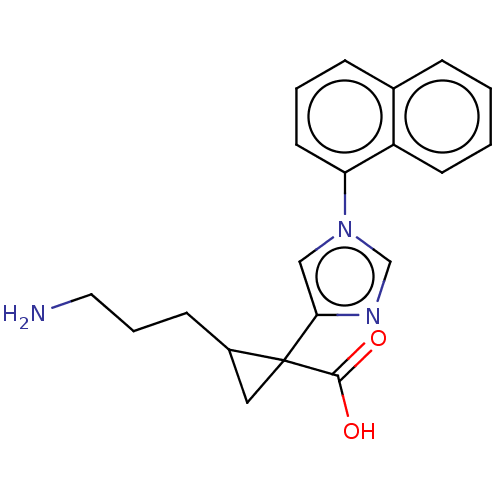
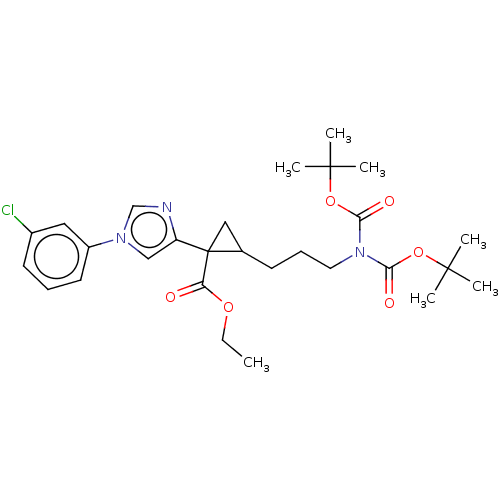
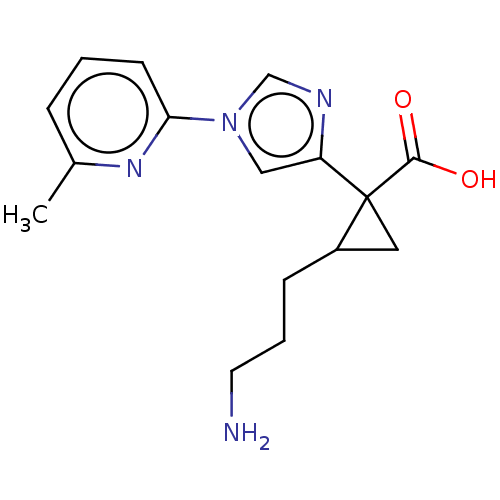
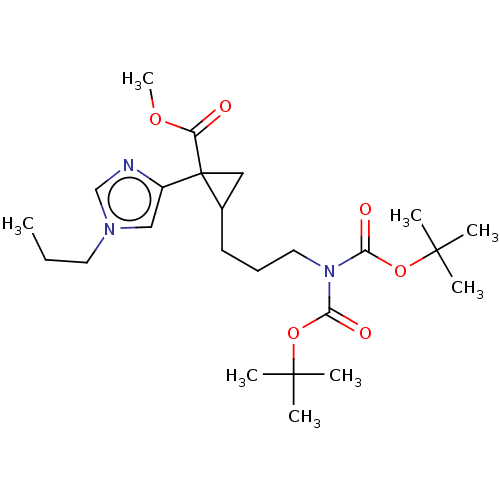
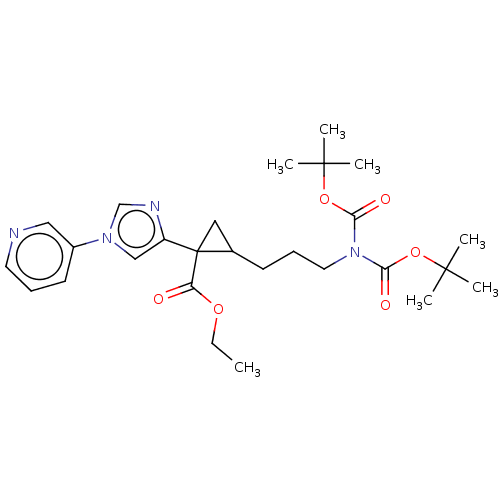
Report error Found 68 Enz. Inhib. hit(s) with all data for entry = 1493
Affinity DataIC50: 4.5nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 7.90nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 8.30nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 8.70nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 8.70nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 15nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 18nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 19nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 19nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 21nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 26nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 27nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 27nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 29nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 29nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 29nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 30nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 31nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 33nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 35nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 35nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 36nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 36nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 37nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 39nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 40nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 41nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 42nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 52nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 54nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 58nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 58nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 61nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 63nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 70nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 73nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 97nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair
Affinity DataIC50: 98nMAssay Description:HEPES buffered saline (20 mM HEPES, 150 mM NaCl, pH 7.4; hereinafter, referred to as HBS) was used in the preparation of a reaction solution. To 12 &...More data for this Ligand-Target Pair