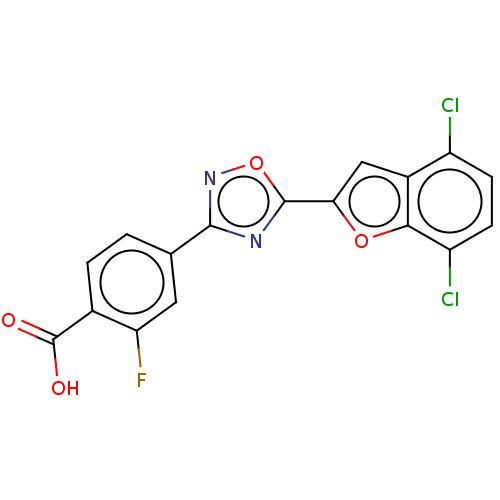
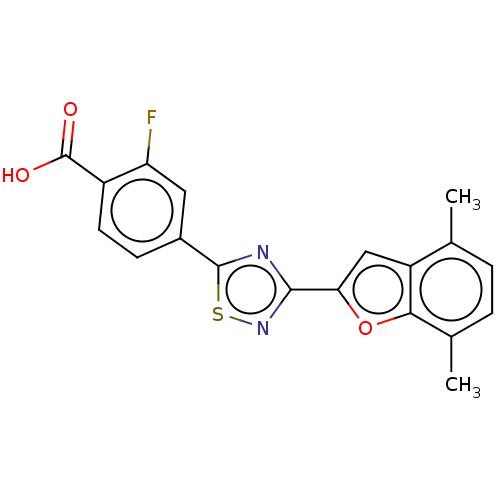
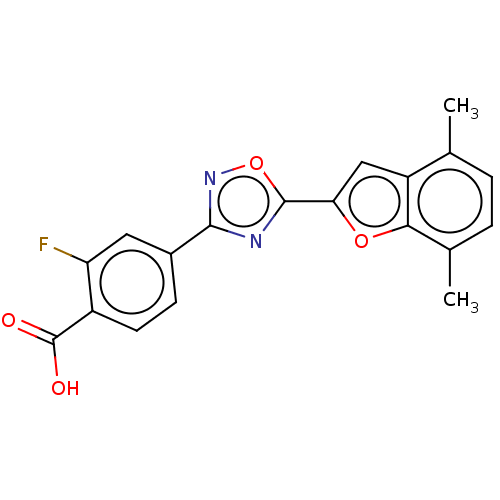
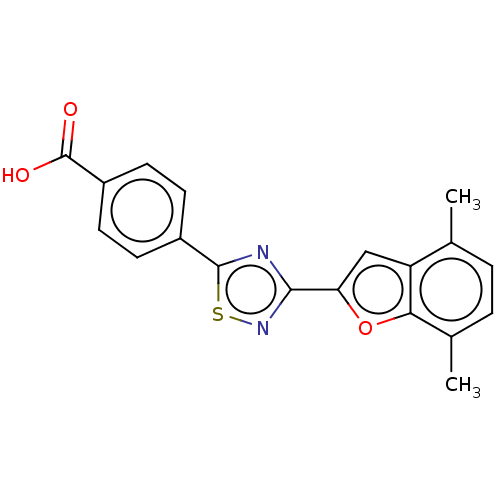
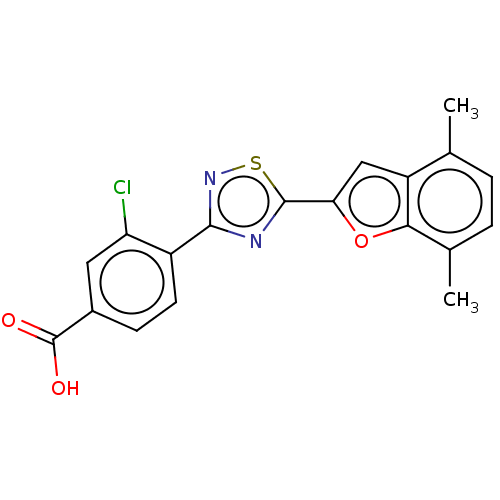
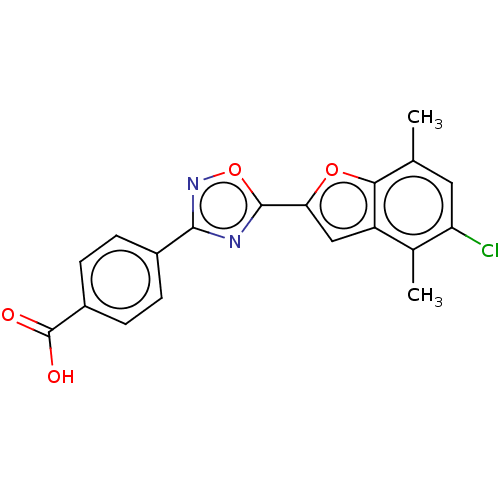
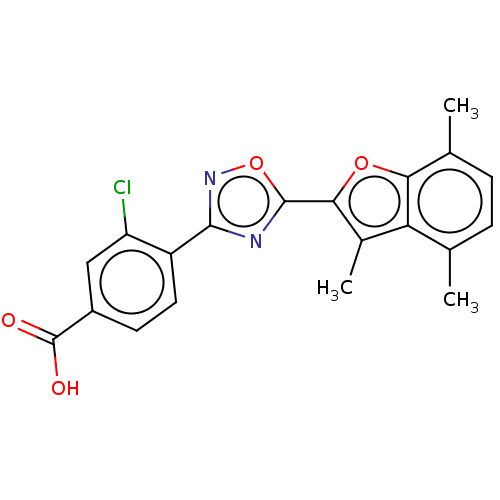
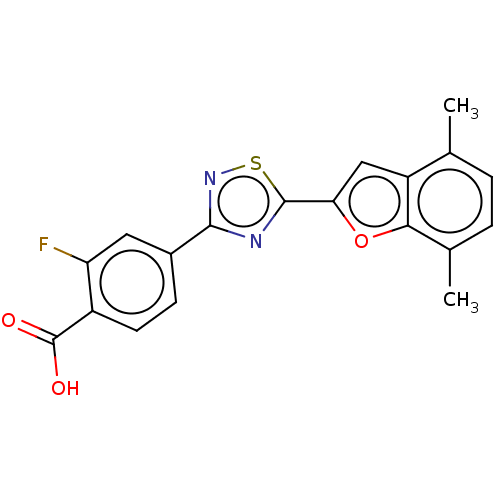
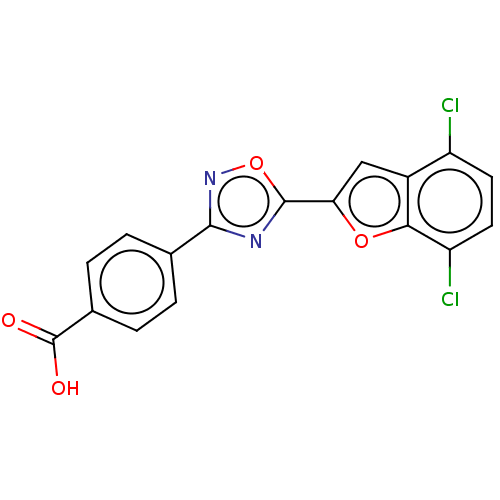
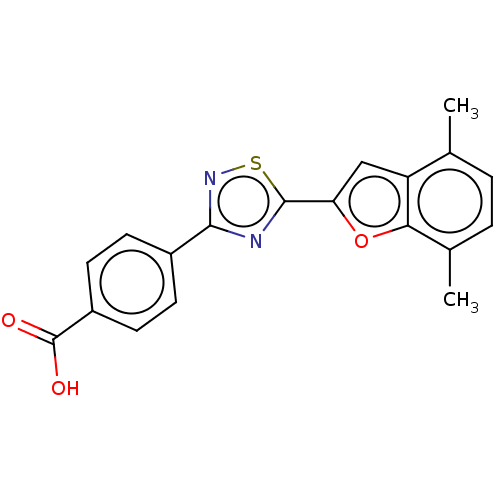
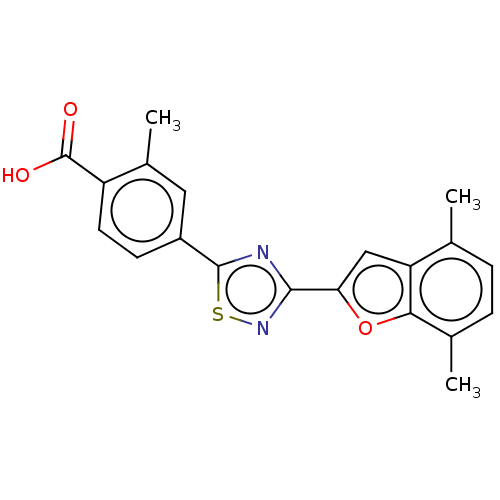
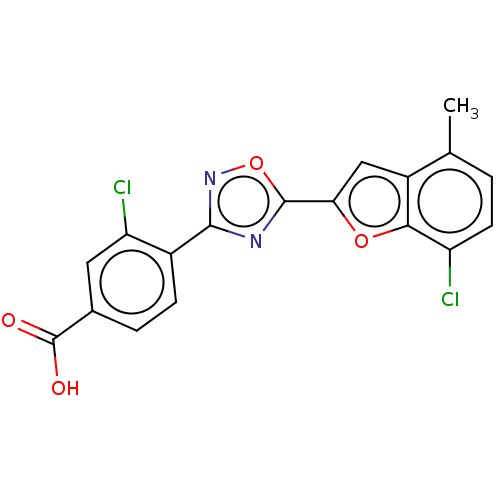
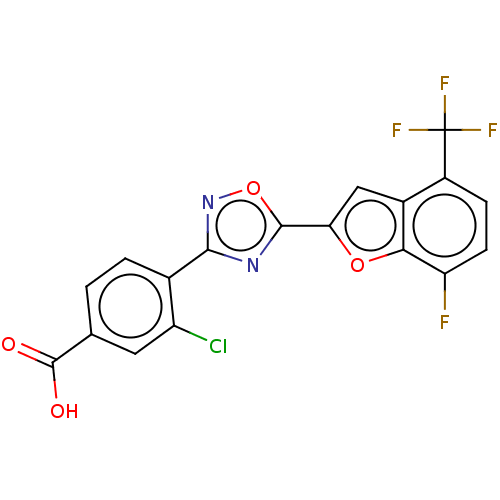
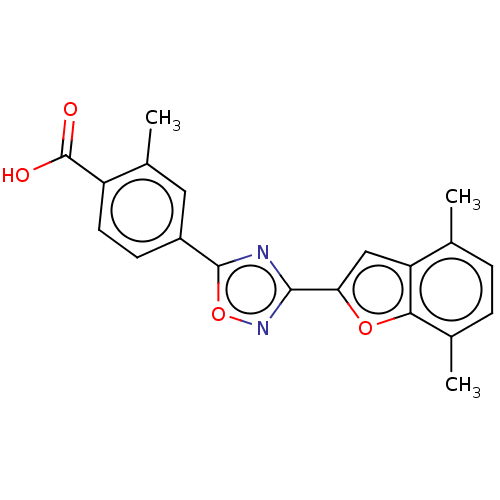
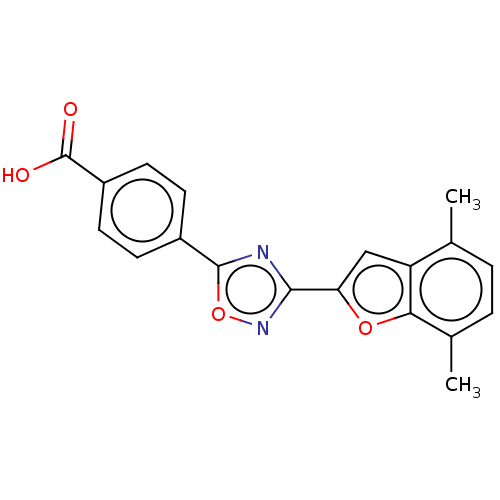
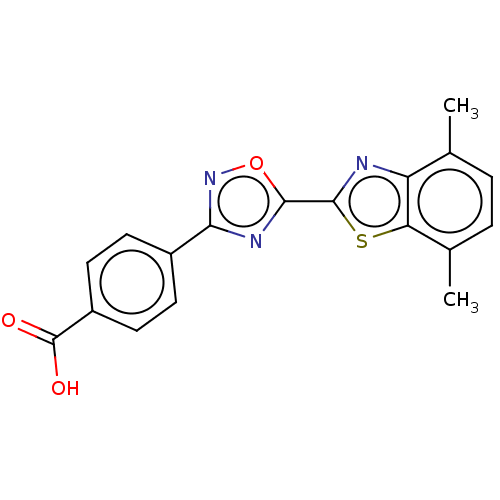
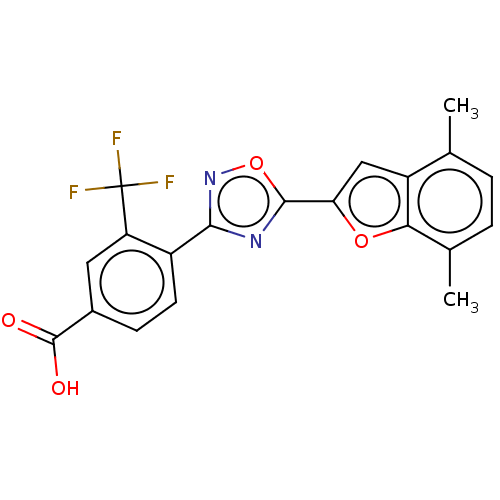
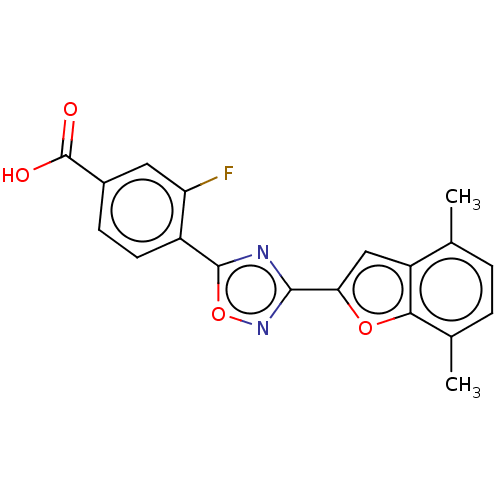
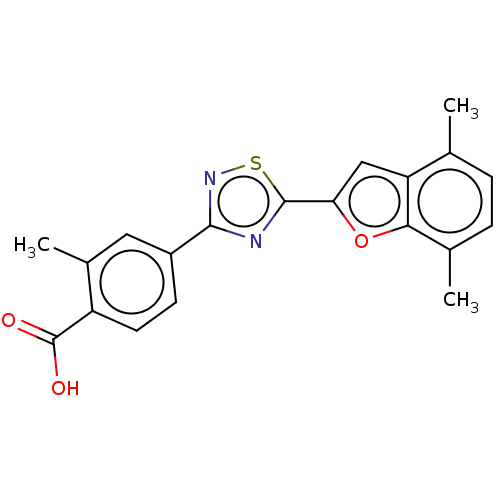
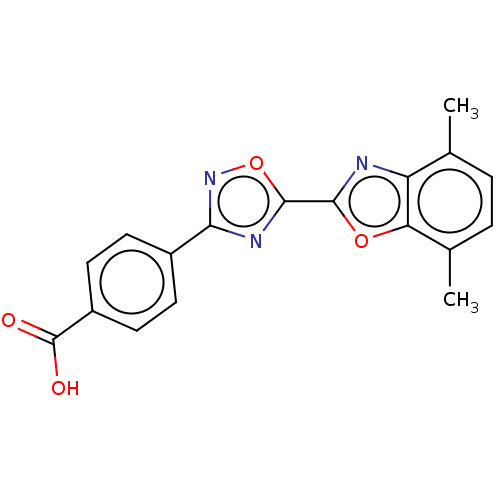
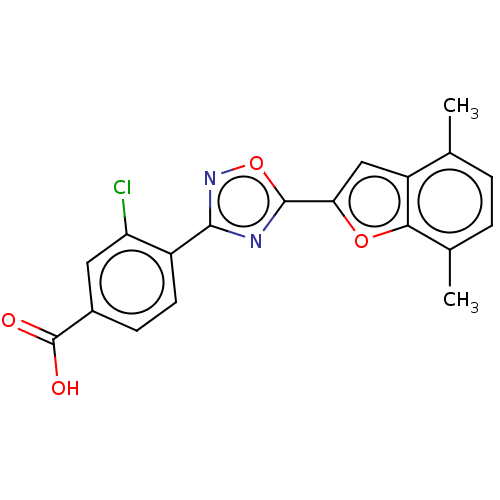
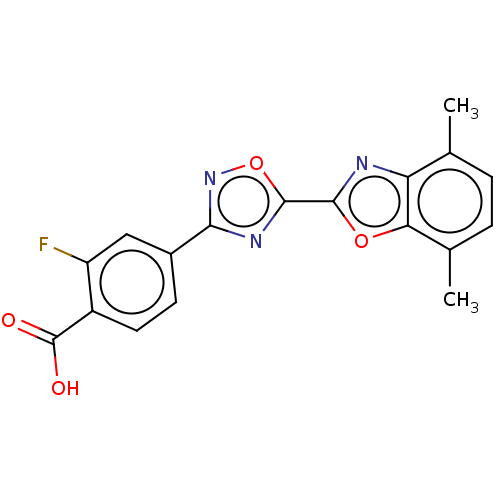
Report error Found 136 Enz. Inhib. hit(s) with all data for entry = 9322
Affinity DataEC50: 0.530nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 0.900nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 1.20nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 1.88nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 1.90nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 1.94nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 2nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 2nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 2.20nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 2.30nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 2.5nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 2.70nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 2.90nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 2.90nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 3.10nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 3.40nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 3.40nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 3.60nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 4nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 4.20nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 4.30nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 4.30nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 4.70nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 5.10nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 5.20nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 5.30nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 6nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 6.10nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 6.30nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 6.80nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 7.40nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 8nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 8.10nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 8.20nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 8.30nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 8.40nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 9nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 9.40nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 10nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 10nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 11nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 11nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 11nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 11nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 11nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 11.4nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 12nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 12nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 13nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair
Affinity DataEC50: 13nMAssay Description:Transcriptional transactivation assays were performed with gal4 fusion receptor constructs, created using each of the RAR ligand binding domains of e...More data for this Ligand-Target Pair