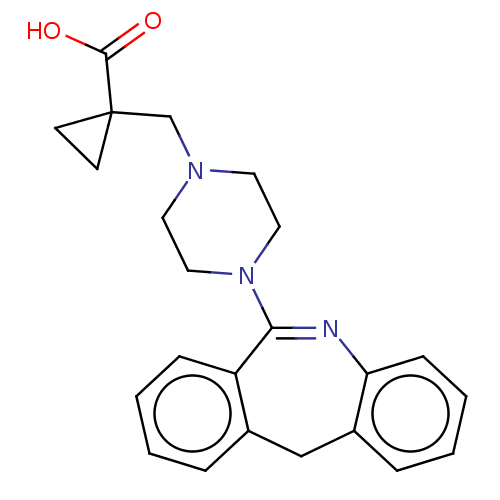
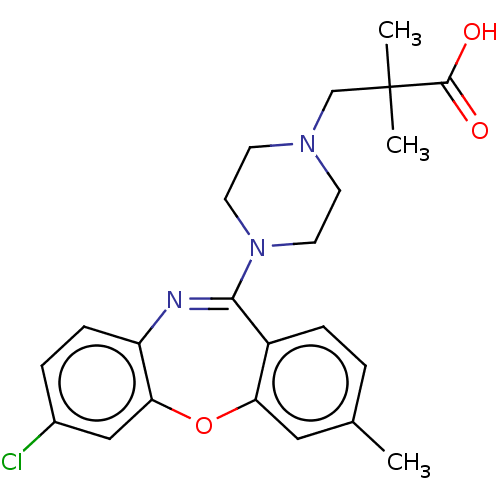
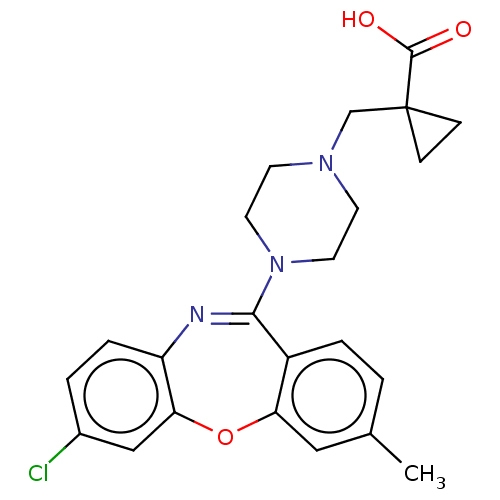
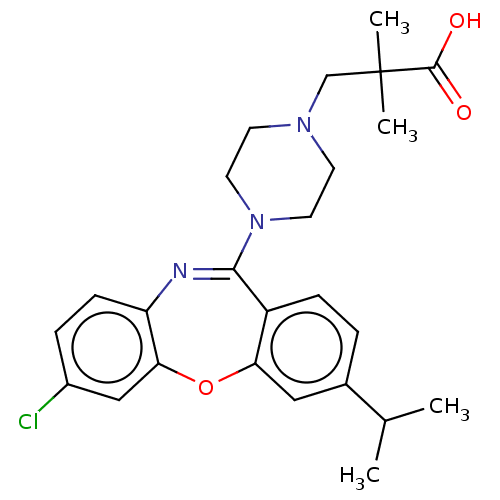
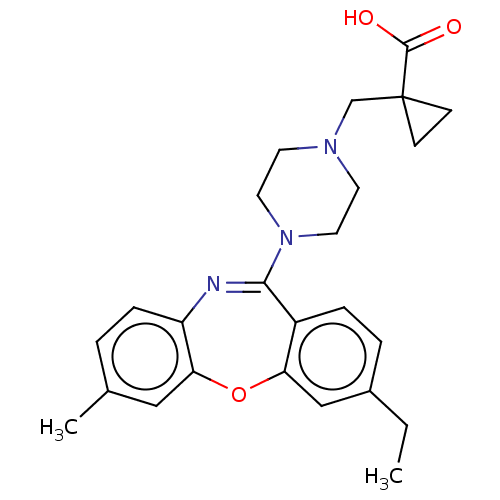
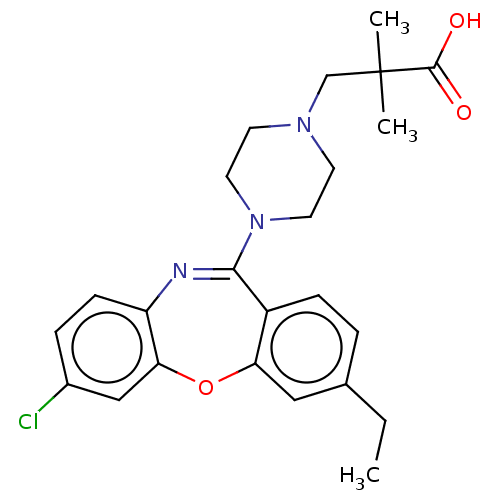
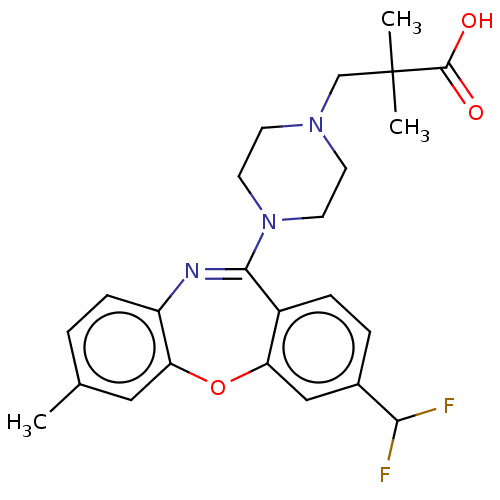
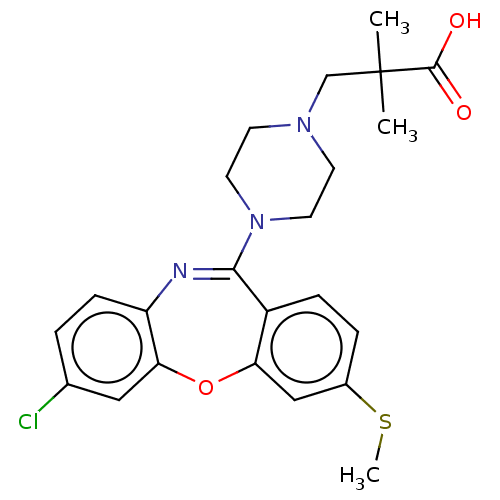
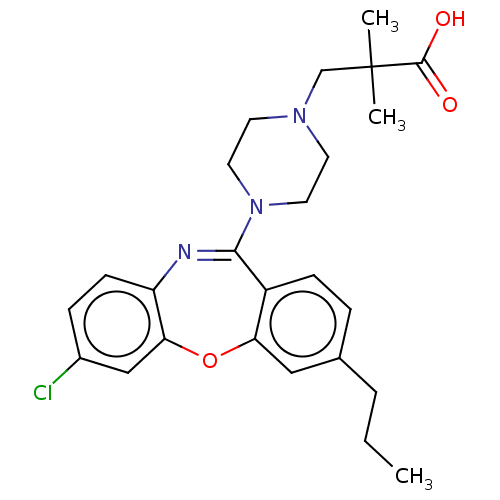
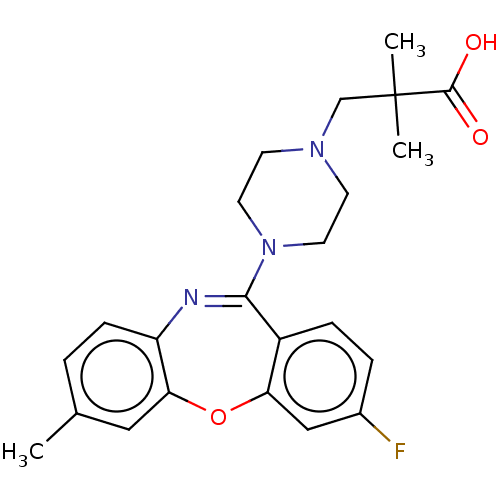
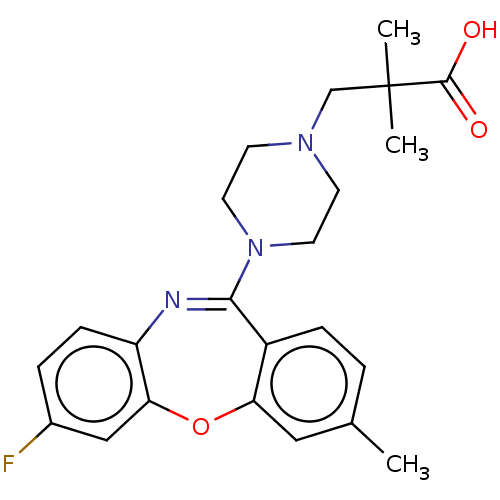
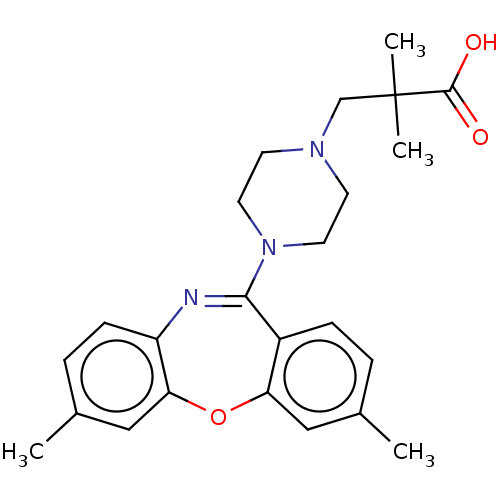
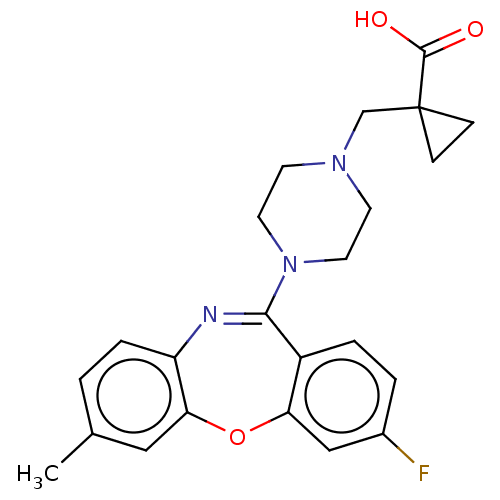
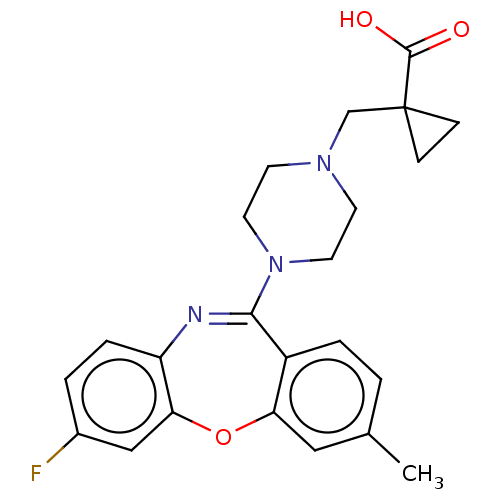
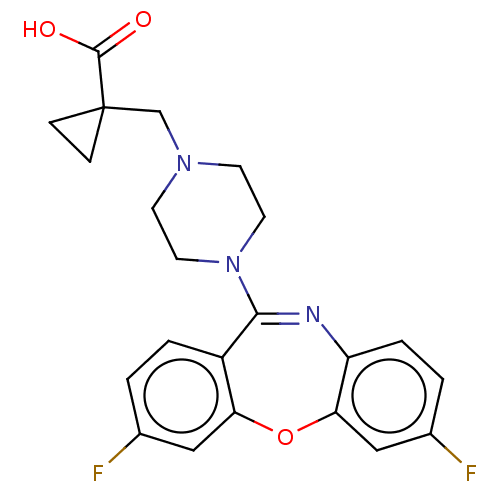
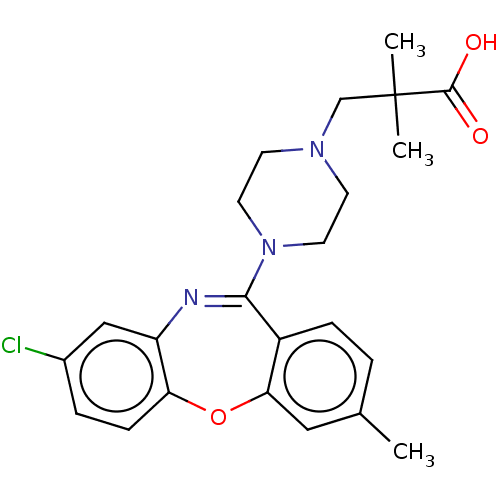
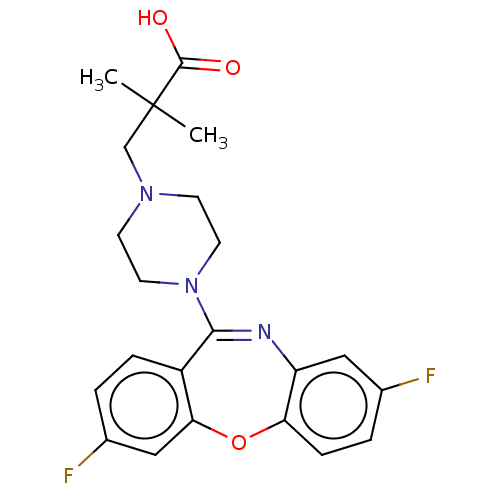
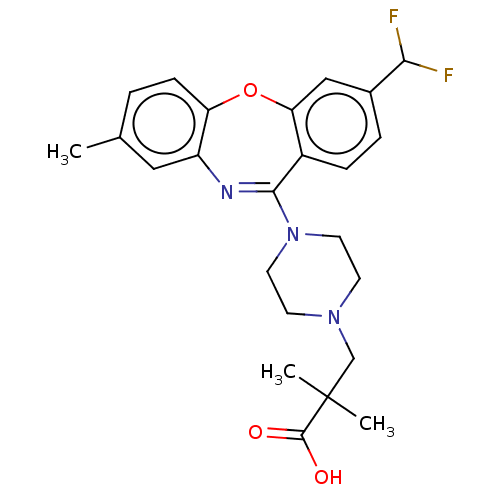
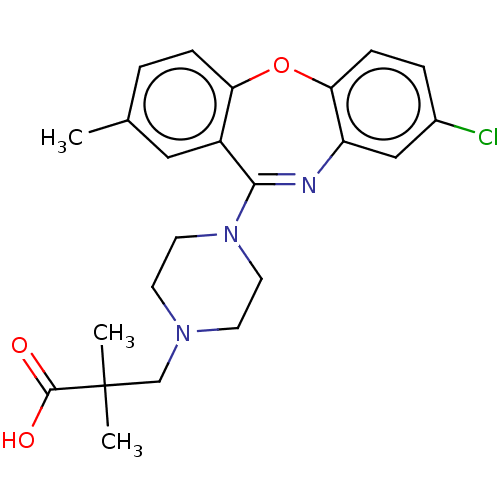
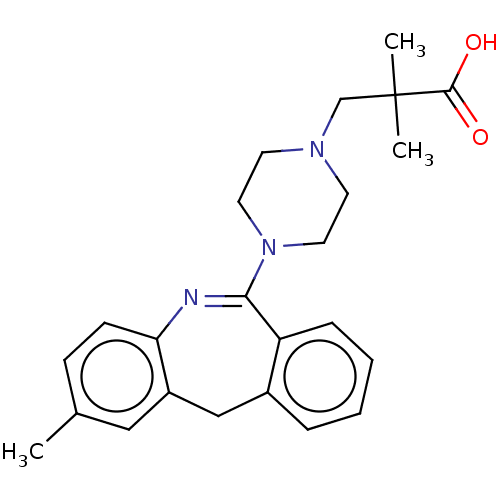
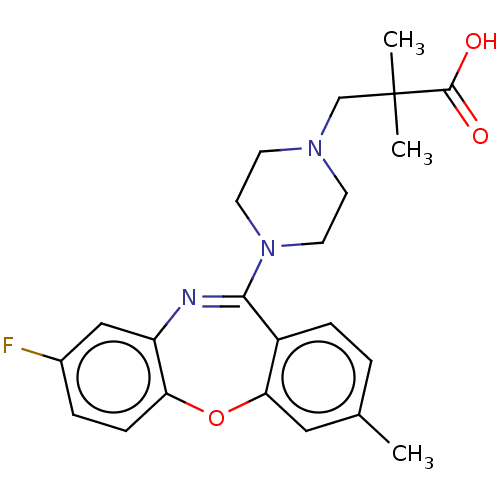
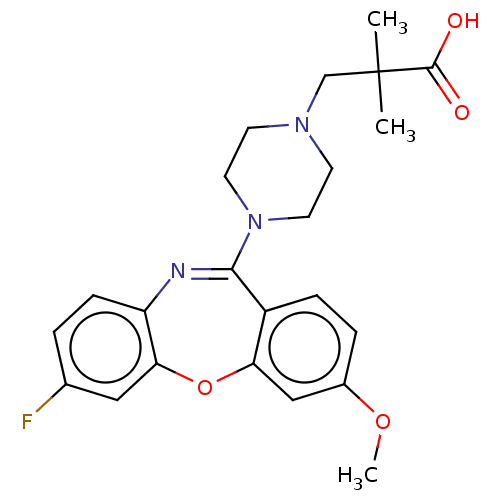
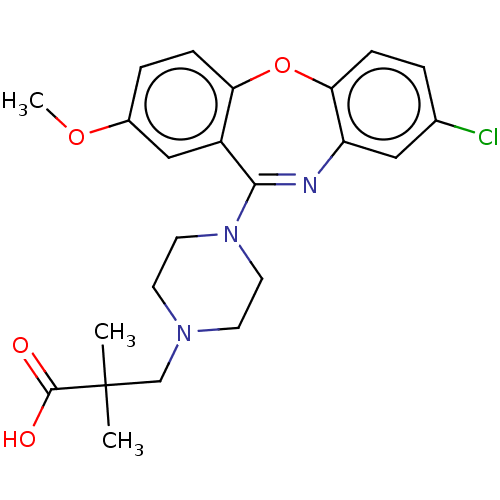
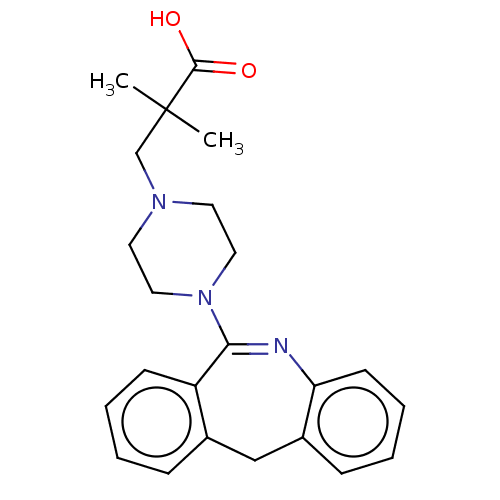
Report error Found 75 Enz. Inhib. hit(s) with all data for entry = 9998
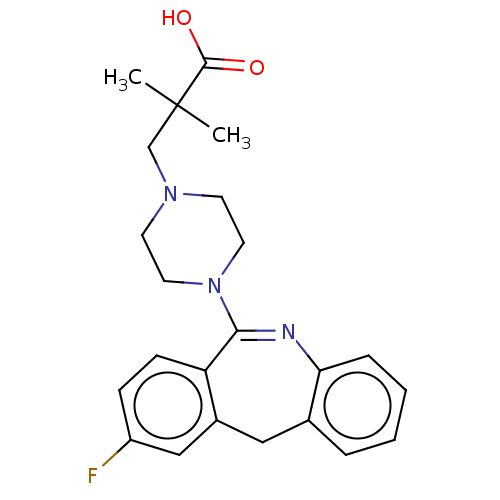
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
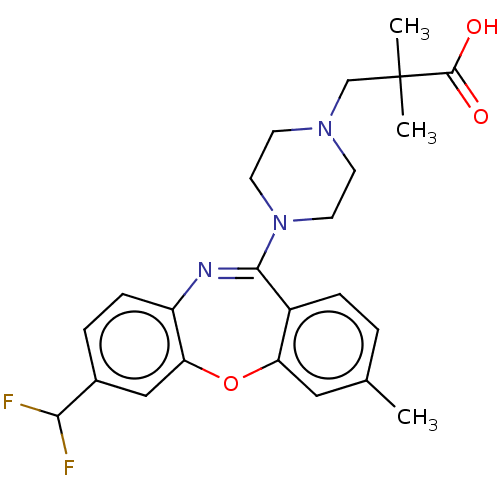
Affinity DataKi: 13nMAssay Description:1 μL of a compound of the present disclosure the reference compound (Table J) was transferred to an assay plate. 1 μL of 0.2 mM SB-206533 w...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μL of a compound of the present disclosure the reference compound (Table J) was transferred to an assay plate. 1 μL of 0.2 mM SB-206533 w...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μL of a compound of the present disclosure the reference compound (Table J) was transferred to an assay plate. 1 μL of 0.2 mM SB-206533 w...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μL of a compound of the present disclosure the reference compound (Table J) was transferred to an assay plate. 1 μL of 0.2 mM SB-206533 w...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μL of a compound of the present disclosure the reference compound (Table J) was transferred to an assay plate. 1 μL of 0.2 mM SB-206533 w...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μL of a compound of the present disclosure the reference compound (Table J) was transferred to an assay plate. 1 μL of 0.2 mM SB-206533 w...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 37.5nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 37.5nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 37.5nMAssay Description:1 μL of a compound of the present disclosure the reference compound (Table J) was transferred to an assay plate. 1 μL of 0.2 mM SB-206533 w...More data for this Ligand-Target Pair
Affinity DataKi: 37.5nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 37.5nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 37.5nMAssay Description:1 μL of a compound of the present disclosure the reference compound (Table J) was transferred to an assay plate. 1 μL of 0.2 mM SB-206533 w...More data for this Ligand-Target Pair
Affinity DataKi: 37.5nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 37.5nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 37.5nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 37.5nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 37.5nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: 46nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: >50nMAssay Description:1 μL of a compound of the present disclosure the reference compound (Table J) was transferred to an assay plate. 1 μL of 0.2 mM SB-206533 w...More data for this Ligand-Target Pair
Affinity DataKi: >50nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: >50nMAssay Description:1 μL of a compound of the present disclosure the reference compound (Table J) was transferred to an assay plate. 1 μL of 0.2 mM SB-206533 w...More data for this Ligand-Target Pair
Affinity DataKi: >50nMAssay Description:1 μL of a compound of the present disclosure the reference compound (Table J) was transferred to an assay plate. 1 μL of 0.2 mM SB-206533 w...More data for this Ligand-Target Pair
Affinity DataKi: >50nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: >50nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: >50nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: >50nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: >50nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair
Affinity DataKi: >50nMAssay Description:1 μl of a compounds of the present disclosure and either reference compound (Table C) were transferred to assay plates. 1 μl of 0.2 mM Keta...More data for this Ligand-Target Pair