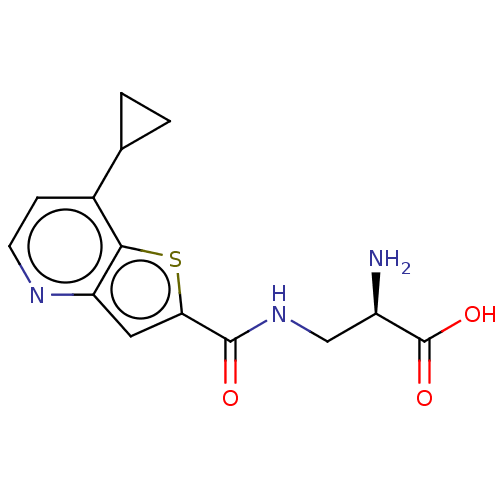
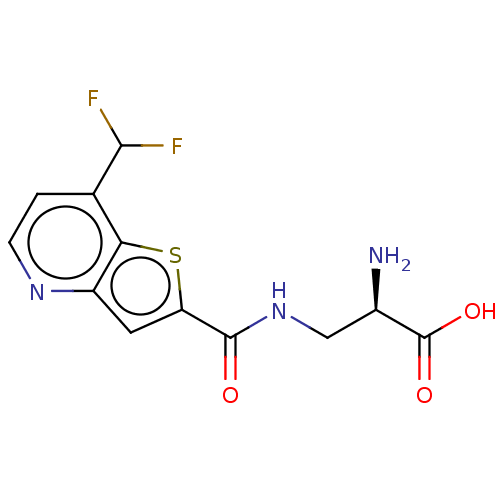
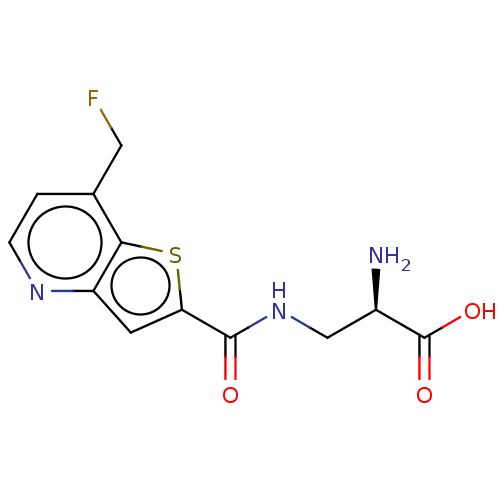
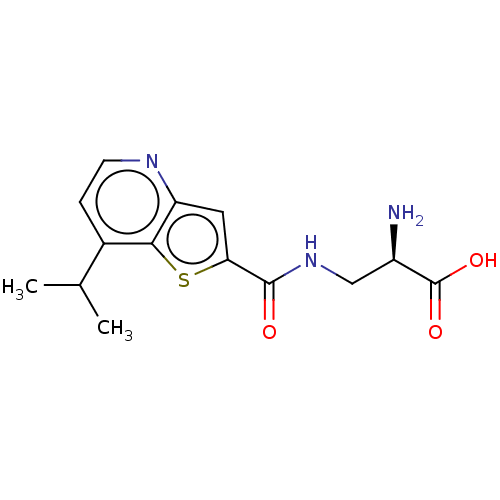
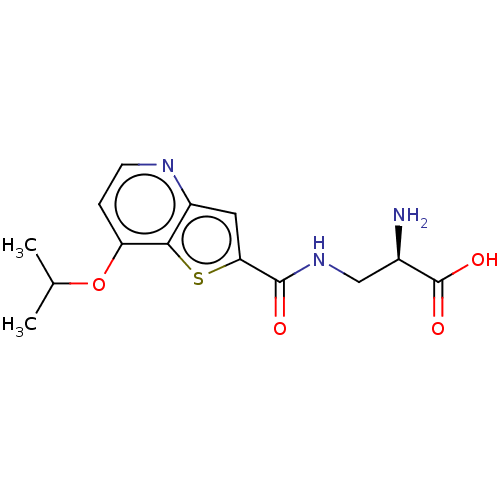
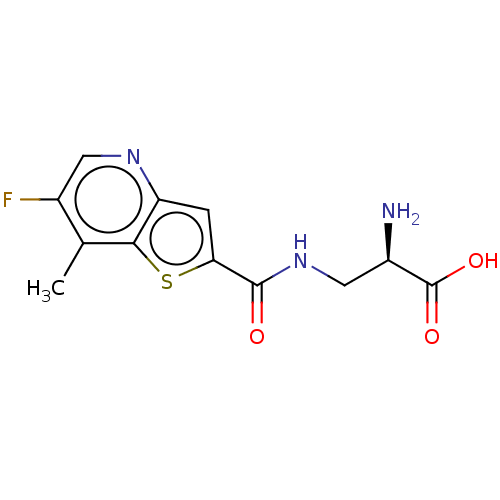
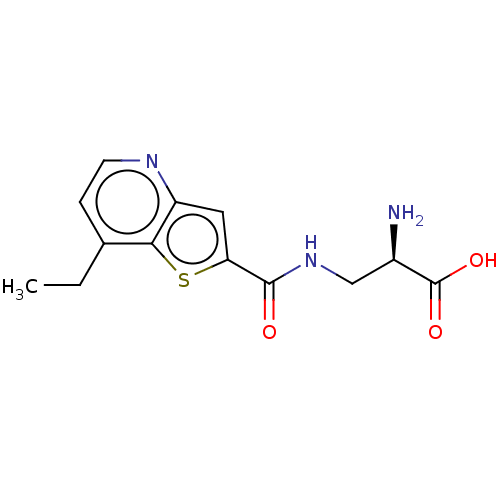
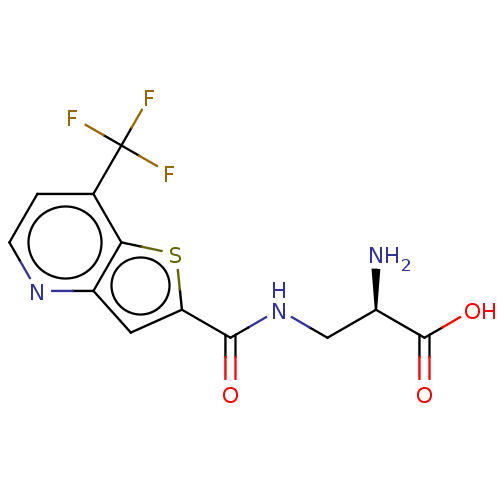
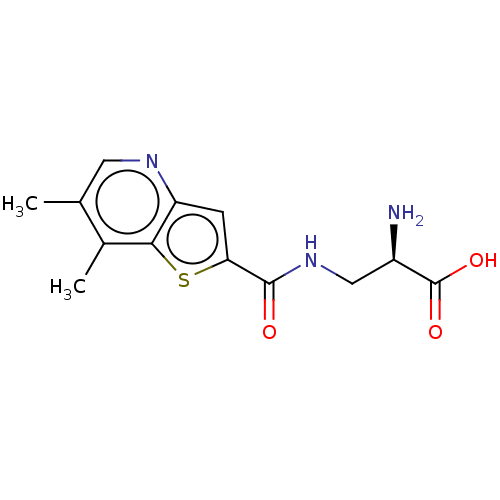
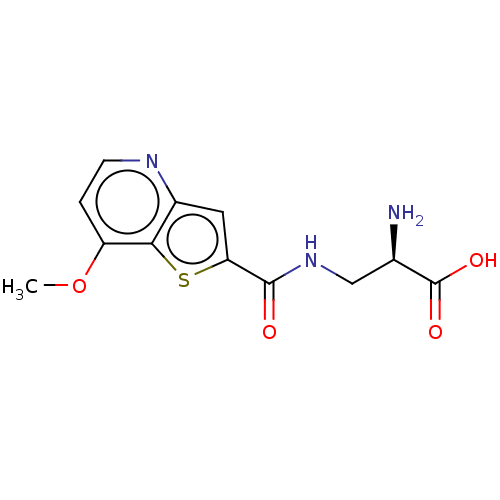
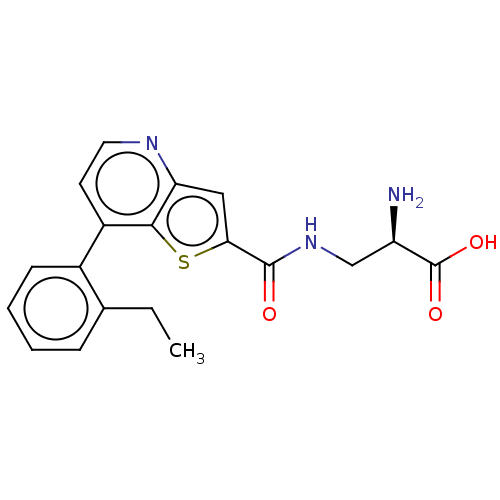
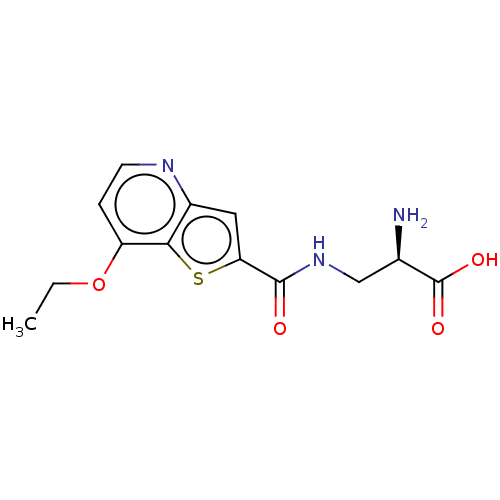
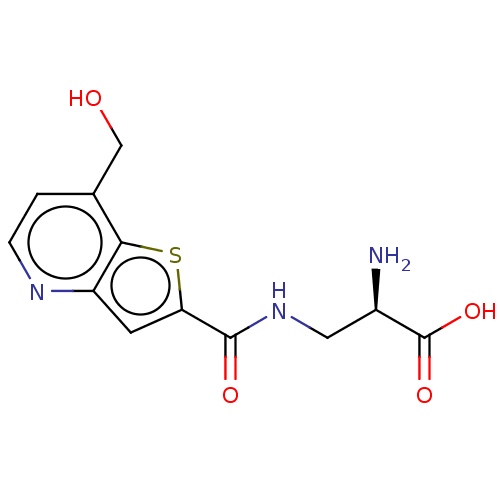
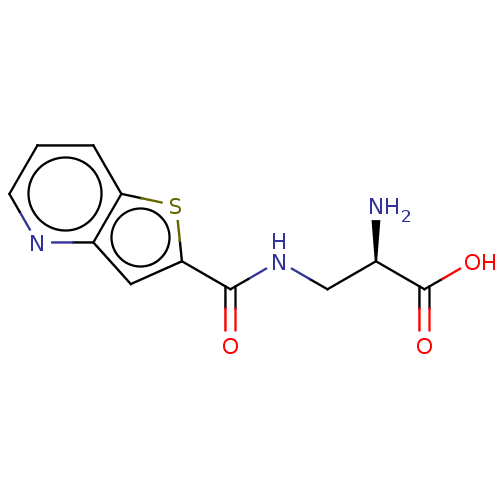
Report error Found 16 Enz. Inhib. hit(s) with all data for entry = 10875
Affinity DataKi: 63nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 96nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 140nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 170nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 180nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 220nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 220nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 260nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 270nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 360nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 490nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 690nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 860nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 870nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 3.90E+3nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 7.40E+3nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
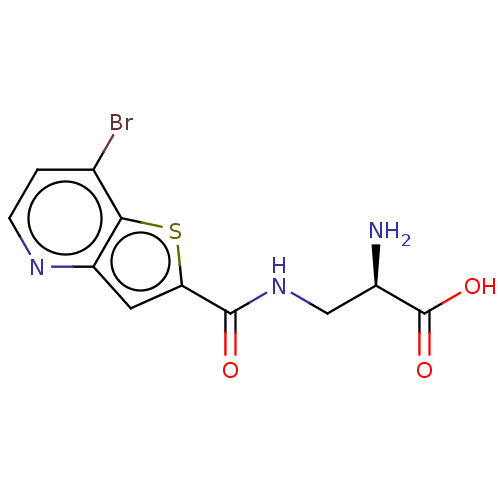
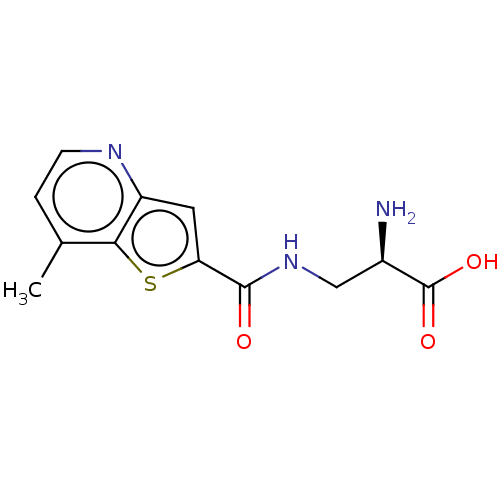
 3D Structure (crystal)
3D Structure (crystal)