Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cyclin-dependent kinase 12
Ligand
BDBM50367857
Substrate
n/a
Meas. Tech.
ChEMBL_1735959 (CHEMBL4151495)
IC50
13±n/a nM
Citation
 Ito, M; Tanaka, T; Toita, A; Uchiyama, N; Kokubo, H; Morishita, N; Klein, MG; Zou, H; Murakami, M; Kondo, M; Sameshima, T; Araki, S; Endo, S; Kawamoto, T; Morin, GB; Aparicio, SA; Nakanishi, A; Maezaki, H; Imaeda, Y Discovery of 3-Benzyl-1-( trans-4-((5-cyanopyridin-2-yl)amino)cyclohexyl)-1-arylurea Derivatives as Novel and Selective Cyclin-Dependent Kinase 12 (CDK12) Inhibitors. J Med Chem 61:7710-7728 (2018) [PubMed] Article
Ito, M; Tanaka, T; Toita, A; Uchiyama, N; Kokubo, H; Morishita, N; Klein, MG; Zou, H; Murakami, M; Kondo, M; Sameshima, T; Araki, S; Endo, S; Kawamoto, T; Morin, GB; Aparicio, SA; Nakanishi, A; Maezaki, H; Imaeda, Y Discovery of 3-Benzyl-1-( trans-4-((5-cyanopyridin-2-yl)amino)cyclohexyl)-1-arylurea Derivatives as Novel and Selective Cyclin-Dependent Kinase 12 (CDK12) Inhibitors. J Med Chem 61:7710-7728 (2018) [PubMed] Article More Info.:
Target
Name:
Cyclin-dependent kinase 12
Synonyms:
2.7.11.22 | 2.7.11.23 | CDC2-related protein kinase 7 | CDK12 | CDK12_HUMAN | CRK7 | CRKRS | Cdc2-related kinase, arginine/serine-rich | Cell division cycle 2-related protein kinase 7 | Cell division protein kinase 12 | Cyclin-dependent kinase 12 | KIAA0904 | hCDK12
Type:
PROTEIN
Mol. Mass.:
164218.64
Organism:
Human
Description:
ChEMBL_117739
Residue:
1490
Sequence:
MPNSERHGGKKDGSGGASGTLQPSSGGGSSNSRERHRLVSKHKRHKSKHSKDMGLVTPEAASLGTVIKPLVEYDDISSDSDTFSDDMAFKLDRRENDERRGSDRSDRLHKHRHHQHRRSRDLLKAKQTEKEKSQEVSSKSGSMKDRISGSSKRSNEETDDYGKAQVAKSSSKESRSSKLHKEKTRKERELKSGHKDRSKSHRKRETPKSYKTVDSPKRRSRSPHRKWSDSSKQDDSPSGASYGQDYDLSPSRSHTSSNYDSYKKSPGSTSRRQSVSPPYKEPSAYQSSTRSPSPYSRRQRSVSPYSRRRSSSYERSGSYSGRSPSPYGRRRSSSPFLSKRSLSRSPLPSRKSMKSRSRSPAYSRHSSSHSKKKRSSSRSRHSSISPVRLPLNSSLGAELSRKKKERAAAAAAAKMDGKESKGSPVFLPRKENSSVEAKDSGLESKKLPRSVKLEKSAPDTELVNVTHLNTEVKNSSDTGKVKLDENSEKHLVKDLKAQGTRDSKPIALKEEIVTPKETETSEKETPPPLPTIASPPPPLPTTTPPPQTPPLPPLPPIPALPQQPPLPPSQPAFSQVPASSTSTLPPSTHSKTSAVSSQANSQPPVQVSVKTQVSVTAAIPHLKTSTLPPLPLPPLLPGDDDMDSPKETLPSKPVKKEKEQRTRHLLTDLPLPPELPGGDLSPPDSPEPKAITPPQQPYKKRPKICCPRYGERRQTESDWGKRCVDKFDIIGIIGEGTYGQVYKAKDKDTGELVALKKVRLDNEKEGFPITAIREIKILRQLIHRSVVNMKEIVTDKQDALDFKKDKGAFYLVFEYMDHDLMGLLESGLVHFSEDHIKSFMKQLMEGLEYCHKKNFLHRDIKCSNILLNNSGQIKLADFGLARLYNSEESRPYTNKVITLWYRPPELLLGEERYTPAIDVWSCGCILGELFTKKPIFQANLELAQLELISRLCGSPCPAVWPDVIKLPYFNTMKPKKQYRRRLREEFSFIPSAALDLLDHMLTLDPSKRCTAEQTLQSDFLKDVELSKMAPPDLPHWQDCHELWSKKRRRQRQSGVVVEEPPPSKTSRKETTSGTSTEPVKNSSPAPPQPAPGKVESGAGDAIGLADITQQLNQSELAVLLNLLQSQTDLSIPQMAQLLNIHSNPEMQQQLEALNQSISALTEATSQQQDSETMAPEESLKEAPSAPVILPSAEQTTLEASSTPADMQNILAVLLSQLMKTQEPAGSLEENNSDKNSGPQGPRRTPTMPQEEAAACPPHILPPEKRPPEPPGPPPPPPPPPLVEGDLSSAPQELNPAVTAALLQLLSQPEAEPPGHLPHEHQALRPMEYSTRPRPNRTYGNTDGPETGFSAIDTDERNSGPALTESLVQTLVKNRTFSGSLSHLGESSSYQGTGSVQFPGDQDLRFARVPLALHPVVGQPFLKAEGSSNSVVHAETKLQNYGELGPGTTGASSSGAGLHWGGPTQSSAYGKLYRGPTRVPPRGGRGRGVPY
Inhibitor
Name:
BDBM50367857
Synonyms:
CHEMBL4159417
Type:
Small organic molecule
Emp. Form.:
C26H28N6O
Mol. Mass.:
440.5401
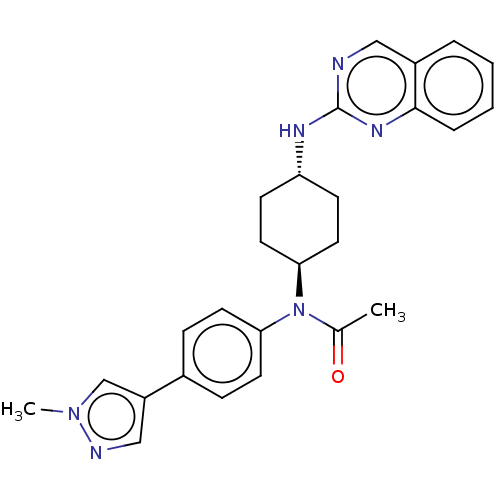
SMILES:
CC(=O)N([C@H]1CC[C@@H](CC1)Nc1ncc2ccccc2n1)c1ccc(cc1)-c1cnn(C)c1 |r,wU:4.3,wD:7.10,(61.19,-8.94,;59.85,-9.71,;58.52,-8.94,;59.85,-11.25,;58.51,-12.02,;58.51,-13.56,;57.18,-14.32,;55.86,-13.55,;55.83,-12.02,;57.18,-11.24,;54.52,-14.32,;53.19,-13.56,;51.85,-14.33,;50.51,-13.56,;50.51,-12.01,;49.19,-11.25,;49.18,-9.72,;50.51,-8.95,;51.84,-9.71,;51.84,-11.24,;53.18,-12.01,;61.19,-12.03,;61.17,-13.56,;62.51,-14.33,;63.84,-13.57,;63.84,-12.02,;62.51,-11.25,;65.18,-14.34,;65.34,-15.87,;66.85,-16.19,;67.62,-14.85,;69.15,-14.69,;66.59,-13.71,)|