Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Presenilin-1
Ligand
BDBM50485435
Substrate
n/a
Meas. Tech.
ChEMBL_829023 (CHEMBL2060673)
IC50
7.5±n/a nM
Citation
 Stepan, AF; Subramanyam, C; Efremov, IV; Dutra, JK; O'Sullivan, TJ; DiRico, KJ; McDonald, WS; Won, A; Dorff, PH; Nolan, CE; Becker, SL; Pustilnik, LR; Riddell, DR; Kauffman, GW; Kormos, BL; Zhang, L; Lu, Y; Capetta, SH; Green, ME; Karki, K; Sibley, E; Atchison, KP; Hallgren, AJ; Oborski, CE; Robshaw, AE; Sneed, B; O'Donnell, CJ Application of the bicyclo[1.1.1]pentane motif as a nonclassical phenyl ring bioisostere in the design of a potent and orally active ?-secretase inhibitor. J Med Chem 55:3414-24 (2012) [PubMed] Article
Stepan, AF; Subramanyam, C; Efremov, IV; Dutra, JK; O'Sullivan, TJ; DiRico, KJ; McDonald, WS; Won, A; Dorff, PH; Nolan, CE; Becker, SL; Pustilnik, LR; Riddell, DR; Kauffman, GW; Kormos, BL; Zhang, L; Lu, Y; Capetta, SH; Green, ME; Karki, K; Sibley, E; Atchison, KP; Hallgren, AJ; Oborski, CE; Robshaw, AE; Sneed, B; O'Donnell, CJ Application of the bicyclo[1.1.1]pentane motif as a nonclassical phenyl ring bioisostere in the design of a potent and orally active ?-secretase inhibitor. J Med Chem 55:3414-24 (2012) [PubMed] Article More Info.:
Target
Name:
Presenilin-1
Synonyms:
3.4.23.- | AD3 | Gamma-Secretase Subunit Presenilin-1 | PS-1 | PS1 | PS1-CTF12 | PSEN1 | PSN1_HUMAN | PSNL1 | Presenilin-1 CTF subunit | Presenilin-1 CTF12 | Presenilin-1 NTF subunit | Protein S182
Type:
n/a
Mol. Mass.:
52657.13
Organism:
Human
Description:
P49768
Residue:
467
Sequence:
MTELPAPLSYFQNAQMSEDNHLSNTVRSQNDNRERQEHNDRRSLGHPEPLSNGRPQGNSRQVVEQDEEEDEELTLKYGAKHVIMLFVPVTLCMVVVVATIKSVSFYTRKDGQLIYTPFTEDTETVGQRALHSILNAAIMISVIVVMTILLVVLYKYRCYKVIHAWLIISSLLLLFFFSFIYLGEVFKTYNVAVDYITVALLIWNFGVVGMISIHWKGPLRLQQAYLIMISALMALVFIKYLPEWTAWLILAVISVYDLVAVLCPKGPLRMLVETAQERNETLFPALIYSSTMVWLVNMAEGDPEAQRRVSKNSKYNAESTERESQDTVAENDDGGFSEEWEAQRDSHLGPHRSTPESRAAVQELSSSILAGEDPEERGVKLGLGDFIFYSVLVGKASATASGDWNTTIACFVAILIGLCLTLLLLAIFKKALPALPISITFGLVFYFATDYLVQPFMDQLAFHQFYI
Inhibitor
Name:
BDBM50485435
Synonyms:
CHEMBL2059014
Type:
Small organic molecule
Emp. Form.:
C18H21ClF3N3O3S
Mol. Mass.:
451.891
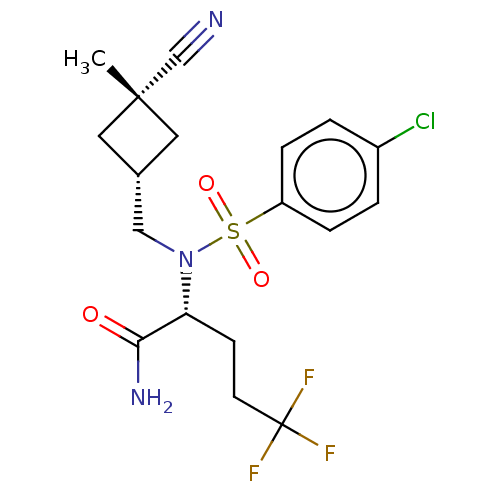
SMILES:
C[C@]1(C[C@H](CN([C@H](CCC(F)(F)F)C(N)=O)S(=O)(=O)c2ccc(Cl)cc2)C1)C#N |r,wU:3.3,1.28,wD:6.6,(7.47,-26.36,;7.49,-27.9,;7.49,-29.44,;9.02,-29.44,;10.37,-30.19,;11.7,-29.41,;13.07,-30.19,;14.4,-29.41,;15.73,-30.18,;17.07,-29.41,;18.4,-30.18,;17.06,-27.87,;18.39,-28.63,;13.07,-31.73,;14.4,-32.49,;11.74,-32.5,;11.7,-27.86,;10.93,-26.53,;10.16,-27.86,;13.03,-27.1,;13.06,-25.57,;14.41,-24.82,;15.73,-25.61,;17.07,-24.85,;15.71,-27.14,;14.36,-27.9,;9.03,-27.9,;6.4,-26.81,;5.31,-25.73,)|