Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Tyrosine-protein kinase BTK
Ligand
BDBM468000
Substrate
n/a
Meas. Tech.
ChEMBL_2238755 (CHEMBL5152651)
Ki
28±n/a nM
Citation
 Tichenor, MS; Wiener, JJM; Rao, NL; Bacani, GM; Wei, J; Pooley Deckhut, C; Barbay, JK; Kreutter, KD; Chang, L; Clancy, KW; Murrey, HE; Wang, W; Ahn, K; Huber, M; Rex, E; Coe, KJ; Wu, J; Rui, H; Sepassi, K; Gaudiano, M; Bekkers, M; Cornelissen, I; Packman, K; Seierstad, M; Xiouras, C; Bembenek, SD; Alexander, R; Milligan, C; Balasubramanian, S; Lebsack, AD; Venable, JD; Philippar, U; Edwards, JP; Hirst, G Discovery of JNJ-64264681: A Potent and Selective Covalent Inhibitor of Bruton's Tyrosine Kinase. J Med Chem 65:14326-14336 (2022) [PubMed]
Tichenor, MS; Wiener, JJM; Rao, NL; Bacani, GM; Wei, J; Pooley Deckhut, C; Barbay, JK; Kreutter, KD; Chang, L; Clancy, KW; Murrey, HE; Wang, W; Ahn, K; Huber, M; Rex, E; Coe, KJ; Wu, J; Rui, H; Sepassi, K; Gaudiano, M; Bekkers, M; Cornelissen, I; Packman, K; Seierstad, M; Xiouras, C; Bembenek, SD; Alexander, R; Milligan, C; Balasubramanian, S; Lebsack, AD; Venable, JD; Philippar, U; Edwards, JP; Hirst, G Discovery of JNJ-64264681: A Potent and Selective Covalent Inhibitor of Bruton's Tyrosine Kinase. J Med Chem 65:14326-14336 (2022) [PubMed] More Info.:
Target
Name:
Tyrosine-protein kinase BTK
Synonyms:
AGMX1 | ATK | Agammaglobulinaemia tyrosine kinase | Agammaglobulinemia tyrosine kinase | B cell progenitor kinase | B-cell progenitor kinase | BPK | BTK | BTK_HUMAN | Bruton tyrosine kinase | Tyrosine Kinase BTK | Tyrosine-protein kinase (BTK) | Tyrosine-protein kinase BTK (BTK)
Type:
Enzyme
Mol. Mass.:
76289.95
Organism:
Homo sapiens (Human)
Description:
Q06187
Residue:
659
Sequence:
MAAVILESIFLKRSQQKKKTSPLNFKKRLFLLTVHKLSYYEYDFERGRRGSKKGSIDVEKITCVETVVPEKNPPPERQIPRRGEESSEMEQISIIERFPYPFQVVYDEGPLYVFSPTEELRKRWIHQLKNVIRYNSDLVQKYHPCFWIDGQYLCCSQTAKNAMGCQILENRNGSLKPGSSHRKTKKPLPPTPEEDQILKKPLPPEPAAAPVSTSELKKVVALYDYMPMNANDLQLRKGDEYFILEESNLPWWRARDKNGQEGYIPSNYVTEAEDSIEMYEWYSKHMTRSQAEQLLKQEGKEGGFIVRDSSKAGKYTVSVFAKSTGDPQGVIRHYVVCSTPQSQYYLAEKHLFSTIPELINYHQHNSAGLISRLKYPVSQQNKNAPSTAGLGYGSWEIDPKDLTFLKELGTGQFGVVKYGKWRGQYDVAIKMIKEGSMSEDEFIEEAKVMMNLSHEKLVQLYGVCTKQRPIFIITEYMANGCLLNYLREMRHRFQTQQLLEMCKDVCEAMEYLESKQFLHRDLAARNCLVNDQGVVKVSDFGLSRYVLDDEYTSSVGSKFPVRWSPPEVLMYSKFSSKSDIWAFGVLMWEIYSLGKMPYERFTNSETAEHIAQGLRLYRPHLASEKVYTIMYSCWHEKADERPTFKILLSNILDVMDEES
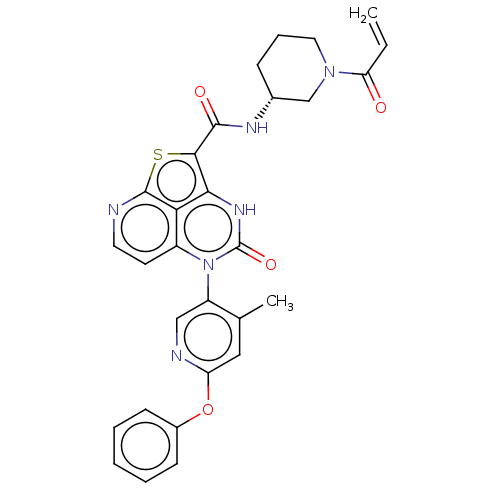
Inhibitor
Name:
BDBM468000
Synonyms:
(R)-N-(1-Acryloylpiperidin-3-yl)-5-(*S)-(4-methyl-6- phenoxypyridin-3-yl)-4-oxo-4,5-dihydro-3H-1-thia-3,5,8- triazaacenaphthylene-2-carboxamide; | US10800792, Example 662 | US10822348, Example 691
Type:
Small organic molecule
Emp. Form.:
C29H26N6O4S
Mol. Mass.:
554.62
SMILES:
Cc1cc(Oc2ccccc2)ncc1-n1c2ccnc3sc(C(=O)N[C@@H]4CCCN(C4)C(=O)C=C)c([nH]c1=O)c23 |r,wU:25.26,(-4.48,-.77,;-4.48,.77,;-5.82,1.54,;-5.82,3.08,;-7.15,3.85,;-8.48,3.08,;-9.82,3.85,;-11.15,3.08,;-11.15,1.54,;-9.82,.77,;-8.48,1.54,;-4.48,3.85,;-3.15,3.08,;-3.15,1.54,;-1.82,.77,;-1.82,-.77,;-3.15,-1.54,;-3.15,-3.08,;-1.82,-3.85,;-.48,-3.08,;1.01,-3.48,;1.94,-1.86,;3.48,-1.86,;4.25,-3.19,;4.25,-.53,;5.79,-.53,;6.56,-1.86,;8.1,-1.86,;8.87,-.53,;8.1,.81,;6.56,.81,;8.87,2.14,;8.1,3.48,;10.41,2.14,;11.15,3.43,;.85,-.77,;.85,.77,;-.48,1.54,;-.48,3.08,;-.48,-1.54,)|