Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Coagulation factor X
Ligand
BDBM50193842
Substrate
n/a
Meas. Tech.
ChEMBL_499722 (CHEMBL968695)
IC50
0.9±n/a nM
Citation
 Zhang, P; Huang, W; Wang, L; Bao, L; Jia, ZJ; Bauer, SM; Goldman, EA; Probst, GD; Song, Y; Su, T; Fan, J; Wu, Y; Li, W; Woolfrey, J; Sinha, U; Wong, PW; Edwards, ST; Arfsten, AE; Clizbe, LA; Kanter, J; Pandey, A; Park, G; Hutchaleelaha, A; Lambing, JL; Hollenbach, SJ; Scarborough, RM; Zhu, BY Discovery of betrixaban (PRT054021), N-(5-chloropyridin-2-yl)-2-(4-(N,N-dimethylcarbamimidoyl)benzamido)-5-methoxybenzamide, a highly potent, selective, and orally efficacious factor Xa inhibitor. Bioorg Med Chem Lett 19:2179-85 (2009) [PubMed] Article
Zhang, P; Huang, W; Wang, L; Bao, L; Jia, ZJ; Bauer, SM; Goldman, EA; Probst, GD; Song, Y; Su, T; Fan, J; Wu, Y; Li, W; Woolfrey, J; Sinha, U; Wong, PW; Edwards, ST; Arfsten, AE; Clizbe, LA; Kanter, J; Pandey, A; Park, G; Hutchaleelaha, A; Lambing, JL; Hollenbach, SJ; Scarborough, RM; Zhu, BY Discovery of betrixaban (PRT054021), N-(5-chloropyridin-2-yl)-2-(4-(N,N-dimethylcarbamimidoyl)benzamido)-5-methoxybenzamide, a highly potent, selective, and orally efficacious factor Xa inhibitor. Bioorg Med Chem Lett 19:2179-85 (2009) [PubMed] Article More Info.:
Target
Name:
Coagulation factor X
Synonyms:
Activated coagulation factor X (FXa) | Activated factor Xa heavy chain | Coagulation factor X precursor | Coagulation factor Xa | F10 | FA10_HUMAN | Factor X heavy chain | Factor X light chain | Factor Xa | Stuart factor | Stuart-Prower factor
Type:
Enzyme
Mol. Mass.:
54726.60
Organism:
Homo sapiens (Human)
Description:
n/a
Residue:
488
Sequence:
MGRPLHLVLLSASLAGLLLLGESLFIRREQANNILARVTRANSFLEEMKKGHLERECMEETCSYEEAREVFEDSDKTNEFWNKYKDGDQCETSPCQNQGKCKDGLGEYTCTCLEGFEGKNCELFTRKLCSLDNGDCDQFCHEEQNSVVCSCARGYTLADNGKACIPTGPYPCGKQTLERRKRSVAQATSSSGEAPDSITWKPYDAADLDPTENPFDLLDFNQTQPERGDNNLTRIVGGQECKDGECPWQALLINEENEGFCGGTILSEFYILTAAHCLYQAKRFKVRVGDRNTEQEEGGEAVHEVEVVIKHNRFTKETYDFDIAVLRLKTPITFRMNVAPACLPERDWAESTLMTQKTGIVSGFGRTHEKGRQSTRLKMLEVPYVDRNSCKLSSSFIITQNMFCAGYDTKQEDACQGDSGGPHVTRFKDTYFVTGIVSWGEGCARKGKYGIYTKVTAFLKWIDRSMKTRGLPKAKSHAPEVITSSPLK
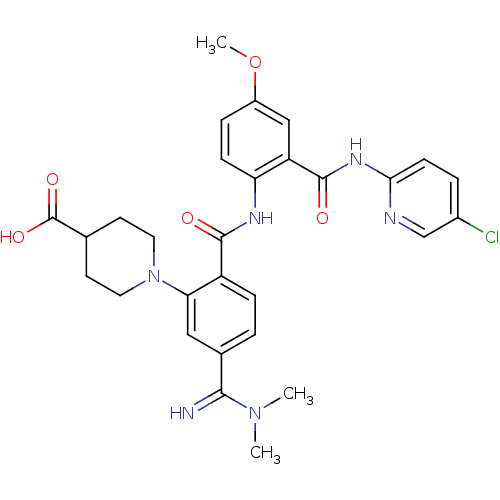
Inhibitor
Name:
BDBM50193842
Synonyms:
1-(2-(2-(5-chloropyridin-2-ylcarbamoyl)-4-methoxyphenylcarbamoyl)-5-(N,N-dimethylcarbamimidoyl)phenyl)piperidine-4-carboxylic acid | 1-[2-[2-(5-chloro-pyridin-2-ylcarbamoyl)-4-methoxy-phenylcarbamoyl]-5-(N,N-dimethyl-carbamimidoyl)-phenyl]-piperidine-4-carboxylic acid | CHEMBL218315
Type:
Small organic molecule
Emp. Form.:
C29H31ClN6O5
Mol. Mass.:
579.047
SMILES:
COc1ccc(NC(=O)c2ccc(cc2N2CCC(CC2)C(O)=O)C(=N)N(C)C)c(c1)C(=O)Nc1ccc(Cl)cn1