Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Enoyl-[acyl-carrier-protein] reductase [NADH] FabI
Ligand
BDBM8726
Substrate
BDBM8737
Meas. Tech.
Enzyme Inhibition Assay
pH
6.5±n/a
Temperature
303.15±n/a K
IC50
430±400 nM
Citation
 Seefeld, MA; Miller, WH; Newlander, KA; Burgess, WJ; DeWolf, WE; Elkins, PA; Head, MS; Jakas, DR; Janson, CA; Keller, PM; Manley, PJ; Moore, TD; Payne, DJ; Pearson, S; Polizzi, BJ; Qiu, X; Rittenhouse, SF; Uzinskas, IN; Wallis, NG; Huffman, WF Indole naphthyridinones as inhibitors of bacterial enoyl-ACP reductases FabI and FabK. J Med Chem 46:1627-35 (2003) [PubMed] Article
Seefeld, MA; Miller, WH; Newlander, KA; Burgess, WJ; DeWolf, WE; Elkins, PA; Head, MS; Jakas, DR; Janson, CA; Keller, PM; Manley, PJ; Moore, TD; Payne, DJ; Pearson, S; Polizzi, BJ; Qiu, X; Rittenhouse, SF; Uzinskas, IN; Wallis, NG; Huffman, WF Indole naphthyridinones as inhibitors of bacterial enoyl-ACP reductases FabI and FabK. J Med Chem 46:1627-35 (2003) [PubMed] Article Target
Name:
Enoyl-[acyl-carrier-protein] reductase [NADH] FabI
Synonyms:
Enoyl - (acyl carrier protein) reductase | Enoyl-ACP Reductase (FabI) | Enoyl-[acyl-carrier-protein] reductase | Enoyl-acyl carrier protein reductase (FabI) | FABI_ECOLI | NADH-dependent enoyl-ACP reductase | envM | fabI
Type:
Enzyme
Mol. Mass.:
27861.12
Organism:
Escherichia coli
Description:
n/a
Residue:
262
Sequence:
MGFLSGKRILVTGVASKLSIAYGIAQAMHREGAELAFTYQNDKLKGRVEEFAAQLGSDIVLQCDVAEDASIDTMFAELGKVWPKFDGFVHSIGFAPGDQLDGDYVNAVTREGFKIAHDISSYSFVAMAKACRSMLNPGSALLTLSYLGAERAIPNYNVMGLAKASLEANVRYMANAMGPEGVRVNAISAGPIRTLAASGIKDFRKMLAHCEAVTPIRRTVTIEDVGNSAAFLCSDLSAGISGEVVHVDGGFSIAAMNELELK
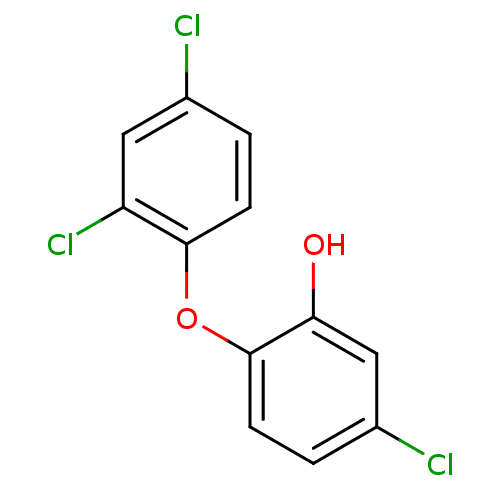
Inhibitor
Name:
BDBM8726
Synonyms:
5-chloro-2-(2,4-dichlorophenoxy)phenol | CHEMBL849 | TCL | Triclosan | US20230414581, Compound 35
Type:
Small organic molecule
Emp. Form.:
C12H7Cl3O2
Mol. Mass.:
289.542
SMILES:
Oc1cc(Cl)ccc1Oc1ccc(Cl)cc1Cl
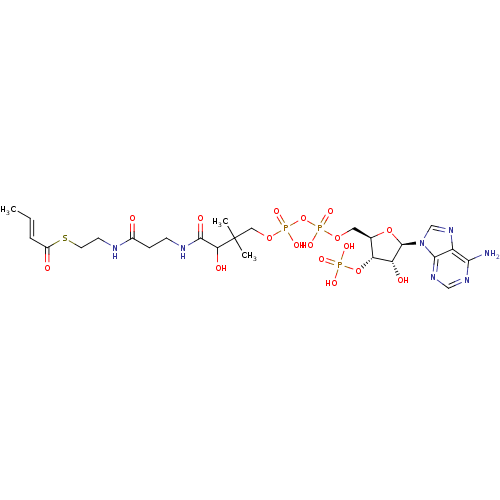
Substrate
Name:
BDBM8737
Synonyms:
2-Butenoyl-CoA | But-2-enoyl-CoA | Crotonoyl-CoA | trans-butyr-2-enoyl-CoA | {[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-2-{[({[(3-{[2-({2-[(2E)-but-2-enoylsulfanyl]ethyl}carbamoyl)ethyl]carbamoyl}-3-hydroxy-2,2-dimethylpropoxy)(hydroxy)phosphoryl]oxy}(hydroxy)phosphoryl)oxy]methyl}-4-hydroxyoxolan-3-yl]oxy}phosphonic acid
Type:
Small organic molecule
Emp. Form.:
C25H40N7O17P3S
Mol. Mass.:
835.608
SMILES:
C\C=C\C(=O)SCCNC(=O)CCNC(=O)C(O)C(C)(C)COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1OP(O)(O)=O)n1cnc2c(N)ncnc12 |r|