Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Enoyl-[acyl-carrier-protein] reductase [NADH]
Ligand
BDBM16300
Substrate
BDBM16293
Meas. Tech.
Measurement of Inhibition Constants
pH
6.8±n/a
Temperature
295.15±n/a K
Ki
0.75±0.08 nM
Km
29000±n/a nM
Comments
Ki for the inhibition of InhA by the INH-NAD adduct was from Rawat et al., Proc. Natl. Acad. Sci. U.S.A. 100, 13881-13886.
Citation
 Sullivan, TJ; Truglio, JJ; Boyne, ME; Novichenok, P; Zhang, X; Stratton, CF; Li, HJ; Kaur, T; Amin, A; Johnson, F; Slayden, RA; Kisker, C; Tonge, PJ High affinity InhA inhibitors with activity against drug-resistant strains of Mycobacterium tuberculosis. ACS Chem Biol 1:43-53 (2006) [PubMed] Article
Sullivan, TJ; Truglio, JJ; Boyne, ME; Novichenok, P; Zhang, X; Stratton, CF; Li, HJ; Kaur, T; Amin, A; Johnson, F; Slayden, RA; Kisker, C; Tonge, PJ High affinity InhA inhibitors with activity against drug-resistant strains of Mycobacterium tuberculosis. ACS Chem Biol 1:43-53 (2006) [PubMed] Article Target
Name:
Enoyl-[acyl-carrier-protein] reductase [NADH]
Synonyms:
Enoyl-ACP Reductase (InhA) | Enoyl-[acyl-carrier-protein] reductase | Enoyl-[acyl-carrier-protein] reductase [NADH] | INHA_MYCTU | NADH-dependent enoyl-ACP reductase | inhA
Type:
Enzyme
Mol. Mass.:
28526.00
Organism:
Mycobacterium tuberculosis (strain ATCC 25618 / H37Rv)
Description:
P9WGR1
Residue:
269
Sequence:
MTGLLDGKRILVSGIITDSSIAFHIARVAQEQGAQLVLTGFDRLRLIQRITDRLPAKAPLLELDVQNEEHLASLAGRVTEAIGAGNKLDGVVHSIGFMPQTGMGINPFFDAPYADVSKGIHISAYSYASMAKALLPIMNPGGSIVGMDFDPSRAMPAYNWMTVAKSALESVNRFVAREAGKYGVRSNLVAAGPIRTLAMSAIVGGALGEEAGAQIQLLEEGWDQRAPIGWNMKDATPVAKTVCALLSDWLPATTGDIIYADGGAHTQLL
Inhibitor
Name:
BDBM16300
Synonyms:
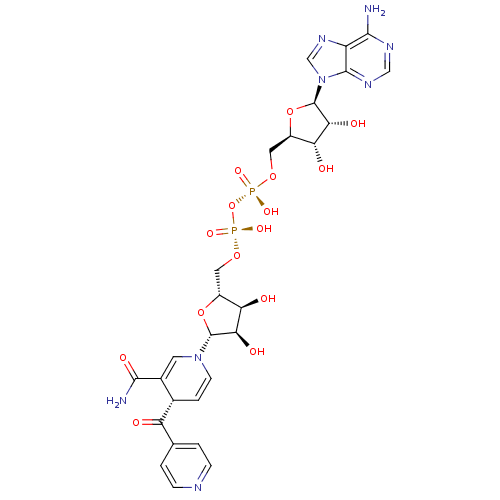
INH-NAD Adduct | ISONICOTINIC-ACETYL-NICOTINAMIDE-ADENINE DINUCLEOTIDE | [({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}(hydroxy)phosphoryl)oxy]({[(2R,3S,4R,5R)-5-[(4S)-3-carbamoyl-4-(pyridin-4-ylcarbonyl)-1,4-dihydropyridin-1-yl]-3,4-dihydroxyoxolan-2-yl]methoxy})phosphinic acid | [[(2R,3S,4R,5R)-5-(6-aminopurin-9-yl)-3,4-dihydroxy-oxolan-2-yl]methoxy-hydroxy-phosphoryl]oxy-[[(2R,3S,4R,5R)-5-[(4S)-3-carbamoyl-4-(pyridine-4-carbonyl)-4H-pyridin-1-yl]-3,4-dihydroxy-oxolan-2-yl]methoxy]phosphinic acid
Type:
n/a
Emp. Form.:
C27H32N8O15P2
Mol. Mass.:
770.5351
SMILES:
NC(=O)C1=CN(C=C[C@@H]1C(=O)c1ccncc1)[C@@H]1O[C@H](CO[P@](O)(=O)O[P@](O)(=O)OC[C@H]2O[C@H]([C@H](O)[C@@H]2O)n2cnc3c(N)ncnc23)[C@@H](O)[C@H]1O |r,c:6,t:3|