Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Peroxisome proliferator-activated receptor alpha
Ligand
BDBM28749
Substrate
BDBM10852
Meas. Tech.
Cell-Based Transcription Assay
EC50
468±10 nM
Comments
42% of maximal activation.
Citation
 Shen, L; Zhang, Y; Wang, A; Sieber-McMaster, E; Chen, X; Pelton, P; Xu, JZ; Yang, M; Zhu, P; Zhou, L; Reuman, M; Hu, Z; Russell, R; Gibbs, AC; Ross, H; Demarest, K; Murray, WV; Kuo, GH Synthesis and identification of [1,2,4]thiadiazole derivatives as a new series of potent and orally active dual agonists of peroxisome proliferator-activated receptors alpha and delta. J Med Chem 50:3954-63 (2007) [PubMed] Article
Shen, L; Zhang, Y; Wang, A; Sieber-McMaster, E; Chen, X; Pelton, P; Xu, JZ; Yang, M; Zhu, P; Zhou, L; Reuman, M; Hu, Z; Russell, R; Gibbs, AC; Ross, H; Demarest, K; Murray, WV; Kuo, GH Synthesis and identification of [1,2,4]thiadiazole derivatives as a new series of potent and orally active dual agonists of peroxisome proliferator-activated receptors alpha and delta. J Med Chem 50:3954-63 (2007) [PubMed] Article More Info.:
Target
Name:
Peroxisome proliferator-activated receptor alpha
Synonyms:
NR1C1 | Nuclear receptor subfamily 1 group C member 1 | PPAR | PPAR alpha/gamma | PPAR-alpha | PPARA | PPARA_HUMAN | Peroxisome Proliferator-Activated Receptor alpha | Peroxisome proliferator-activated receptor | Peroxisome proliferator-activated receptor alpha (PPAR alpha)
Type:
Enzyme
Mol. Mass.:
52222.08
Organism:
Homo sapiens (Human)
Description:
Q07869
Residue:
468
Sequence:
MVDTESPLCPLSPLEAGDLESPLSEEFLQEMGNIQEISQSIGEDSSGSFGFTEYQYLGSCPGSDGSVITDTLSPASSPSSVTYPVVPGSVDESPSGALNIECRICGDKASGYHYGVHACEGCKGFFRRTIRLKLVYDKCDRSCKIQKKNRNKCQYCRFHKCLSVGMSHNAIRFGRMPRSEKAKLKAEILTCEHDIEDSETADLKSLAKRIYEAYLKNFNMNKVKARVILSGKASNNPPFVIHDMETLCMAEKTLVAKLVANGIQNKEAEVRIFHCCQCTSVETVTELTEFAKAIPGFANLDLNDQVTLLKYGVYEAIFAMLSSVMNKDGMLVAYGNGFITREFLKSLRKPFCDIMEPKFDFAMKFNALELDDSDISLFVAAIICCGDRPGLLNVGHIEKMQEGIVHVLRLHLQSNHPDDIFLFPKLLQKMADLRQLVTEHAQLVQIIKKTESDAALHPLLQEIYRDMY
Inhibitor
Name:
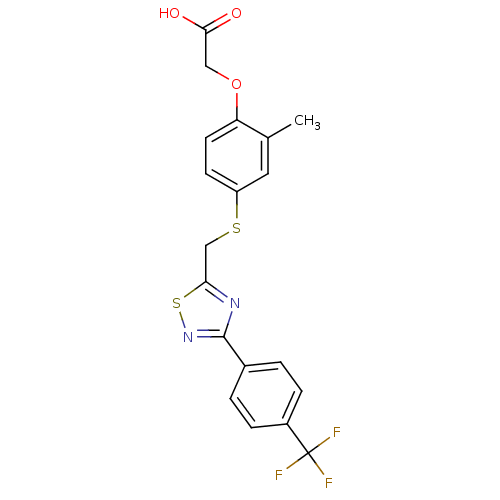
BDBM28749
Synonyms:
2-{2-methyl-4-[({3-[4-(trifluoromethyl)phenyl]-1,2,4-thiadiazol-5-yl}methyl)sulfanyl]phenoxy}acetic acid | [1,2,4]thiadiazole derivative, 13
Type:
Small organic molecule
Emp. Form.:
C19H15F3N2O3S2
Mol. Mass.:
440.459
SMILES:
Cc1cc(SCc2nc(ns2)-c2ccc(cc2)C(F)(F)F)ccc1OCC(O)=O
Substrate