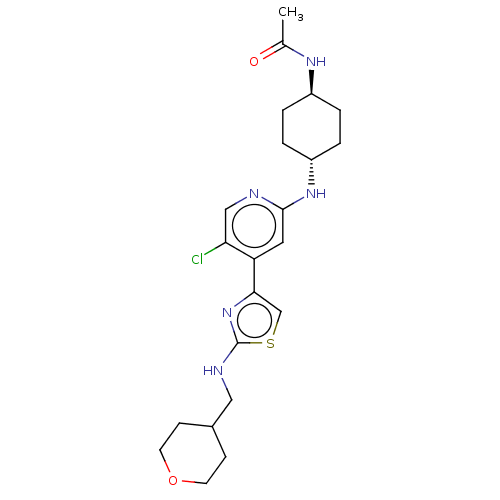
Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cyclin-K
Ligand
BDBM50466229
Substrate
n/a
Meas. Tech.
ChEMBL_1792118 (CHEMBL4264037)
IC50
101±n/a nM
Citation
 Wang, B; Wu, J; Wu, Y; Chen, C; Zou, F; Wang, A; Wu, H; Hu, Z; Jiang, Z; Liu, Q; Wang, W; Zhang, Y; Liu, F; Zhao, M; Hu, J; Huang, T; Ge, J; Wang, L; Ren, T; Wang, Y; Liu, J; Liu, Q Discovery of 4-(((4-(5-chloro-2-(((1s,4s)-4-((2-methoxyethyl)amino)cyclohexyl)amino)pyridin-4-yl)thiazol-2-yl)amino)methyl)tetrahydro-2H-pyran-4-carbonitrile (JSH-150) as a novel highly selective and potent CDK9 kinase inhibitor. Eur J Med Chem 158:896-916 (2018) [PubMed] Article
Wang, B; Wu, J; Wu, Y; Chen, C; Zou, F; Wang, A; Wu, H; Hu, Z; Jiang, Z; Liu, Q; Wang, W; Zhang, Y; Liu, F; Zhao, M; Hu, J; Huang, T; Ge, J; Wang, L; Ren, T; Wang, Y; Liu, J; Liu, Q Discovery of 4-(((4-(5-chloro-2-(((1s,4s)-4-((2-methoxyethyl)amino)cyclohexyl)amino)pyridin-4-yl)thiazol-2-yl)amino)methyl)tetrahydro-2H-pyran-4-carbonitrile (JSH-150) as a novel highly selective and potent CDK9 kinase inhibitor. Eur J Med Chem 158:896-916 (2018) [PubMed] Article More Info.:
Target
Name:
Cyclin-K
Synonyms:
CCNK | CCNK_HUMAN | CPR4
Type:
PROTEIN
Mol. Mass.:
64254.10
Organism:
Homo sapiens (Human)
Description:
ChEMBL_107901
Residue:
580
Sequence:
MKENKENSSPSVTSANLDHTKPCWYWDKKDLAHTPSQLEGLDPATEARYRREGARFIFDVGTRLGLHYDTLATGIIYFHRFYMFHSFKQFPRYVTGACCLFLAGKVEETPKKCKDIIKTARSLLNDVQFGQFGDDPKEEVMVLERILLQTIKFDLQVEHPYQFLLKYAKQLKGDKNKIQKLVQMAWTFVNDSLCTTLSLQWEPEIIAVAVMYLAGRLCKFEIQEWTSKPMYRRWWEQFVQDVPVDVLEDICHQILDLYSQGKQQMPHHTPHQLQQPPSLQPTPQVPQVQQSQPSQSSEPSQPQQKDPQQPAQQQQPAQQPKKPSPQPSSPRQVKRAVVVSPKEENKAAEPPPPKIPKIETTHPPLPPAHPPPDRKPPLAAALGEAEPPGPVDATDLPKVQIPPPAHPAPVHQPPPLPHRPPPPPPSSYMTGMSTTSSYMSGEGYQSLQSMMKTEGPSYGALPPAYGPPAHLPYHPHVYPPNPPPPPVPPPPASFPPPAIPPPTPGYPPPPPTYNPNFPPPPPRLPPTHAVPPHPPPGLGLPPASYPPPAVPPGGQPPVPPPIPPPGMPPVGGLGRAAWMR
Inhibitor
Name:
BDBM50466229
Synonyms:
CHEMBL4279380
Type:
Small organic molecule
Emp. Form.:
C22H30ClN5O2S
Mol. Mass.:
464.024
SMILES:
CC(=O)N[C@H]1CC[C@@H](CC1)Nc1cc(-c2csc(NCC3CCOCC3)n2)c(Cl)cn1 |r,wU:4.3,wD:7.10,(19.81,-28.2,;19.8,-29.74,;21.13,-30.52,;18.46,-30.49,;18.45,-32.03,;19.77,-32.82,;19.76,-34.35,;18.42,-35.11,;17.09,-34.34,;17.11,-32.8,;18.41,-36.65,;19.74,-37.43,;21.09,-36.65,;22.42,-37.42,;23.75,-36.64,;23.8,-35.1,;25.27,-34.66,;26.13,-35.95,;27.67,-35.95,;28.45,-34.61,;29.99,-34.61,;30.75,-35.95,;32.29,-35.95,;33.07,-34.61,;32.29,-33.27,;30.75,-33.27,;25.19,-37.16,;22.42,-38.97,;23.76,-39.73,;21.09,-39.73,;19.74,-38.97,)|