Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Glycogen phosphorylase, liver form
Ligand
BDBM50256072
Substrate
n/a
Meas. Tech.
ChEMBL_515052 (CHEMBL1034794)
IC50
845±n/a nM
Citation
 Sparks, SM; Banker, P; Bickett, DM; Carter, HL; Clancy, DC; Dickerson, SH; Dwornik, KA; Garrido, DM; Golden, PL; Nolte, RT; Peat, AJ; Sheckler, LR; Tavares, FX; Thomson, SA; Wang, L; Weiel, JE Anthranilimide-based glycogen phosphorylase inhibitors for the treatment of type 2 diabetes: 1. Identification of 1-amino-1-cycloalkyl carboxylic acid headgroups. Bioorg Med Chem Lett 19:976-80 (2009) [PubMed] Article
Sparks, SM; Banker, P; Bickett, DM; Carter, HL; Clancy, DC; Dickerson, SH; Dwornik, KA; Garrido, DM; Golden, PL; Nolte, RT; Peat, AJ; Sheckler, LR; Tavares, FX; Thomson, SA; Wang, L; Weiel, JE Anthranilimide-based glycogen phosphorylase inhibitors for the treatment of type 2 diabetes: 1. Identification of 1-amino-1-cycloalkyl carboxylic acid headgroups. Bioorg Med Chem Lett 19:976-80 (2009) [PubMed] Article More Info.:
Target
Name:
Glycogen phosphorylase, liver form
Synonyms:
Glycogen Phosphorylase (PYGL) | Glycogen Phosphorylase, liver form | Liver glycogen phosphorylase | PYGL | PYGL_HUMAN
Type:
Homodimer
Mol. Mass.:
97153.98
Organism:
Homo sapiens (Human)
Description:
Dimers associate into a tetramer to form the enzymatically active phosphorylase A.
Residue:
847
Sequence:
MAKPLTDQEKRRQISIRGIVGVENVAELKKSFNRHLHFTLVKDRNVATTRDYYFALAHTVRDHLVGRWIRTQQHYYDKCPKRVYYLSLEFYMGRTLQNTMINLGLQNACDEAIYQLGLDIEELEEIEEDAGLGNGGLGRLAACFLDSMATLGLAAYGYGIRYEYGIFNQKIRDGWQVEEADDWLRYGNPWEKSRPEFMLPVHFYGKVEHTNTGTKWIDTQVVLALPYDTPVPGYMNNTVNTMRLWSARAPNDFNLRDFNVGDYIQAVLDRNLAENISRVLYPNDNFFEGKELRLKQEYFVVAATLQDIIRRFKASKFGSTRGAGTVFDAFPDQVAIQLNDTHPALAIPELMRIFVDIEKLPWSKAWELTQKTFAYTNHTVLPEALERWPVDLVEKLLPRHLEIIYEINQKHLDRIVALFPKDVDRLRRMSLIEEEGSKRINMAHLCIVGSHAVNGVAKIHSDIVKTKVFKDFSELEPDKFQNKTNGITPRRWLLLCNPGLAELIAEKIGEDYVKDLSQLTKLHSFLGDDVFLRELAKVKQENKLKFSQFLETEYKVKINPSSMFDVQVKRIHEYKRQLLNCLHVITMYNRIKKDPKKLFVPRTVIIGGKAAPGYHMAKMIIKLITSVADVVNNDPMVGSKLKVIFLENYRVSLAEKVIPATDLSEQISTAGTEASGTGNMKFMLNGALTIGTMDGANVEMAEEAGEENLFIFGMRIDDVAALDKKGYEAKEYYEALPELKLVIDQIDNGFFSPKQPDLFKDIINMLFYHDRFKVFADYEAYVKCQDKVSQLYMNPKAWNTMVLKNIAASGKFSSDRTIKEYAQNIWNVEPSDLKISLSNESNKVNGN
Inhibitor
Name:
BDBM50256072
Synonyms:
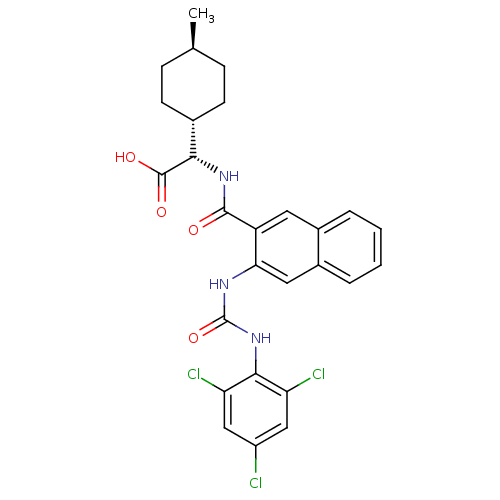
(S)-2-((1r,4S)-4-methylcyclohexyl)-2-(3-(3-(2,4,6-trichlorophenyl)ureido)-2-naphthamido)acetic acid | CHEMBL480732
Type:
Small organic molecule
Emp. Form.:
C27H26Cl3N3O4
Mol. Mass.:
562.872
SMILES:
C[C@H]1CC[C@@H](CC1)[C@H](NC(=O)c1cc2ccccc2cc1NC(=O)Nc1c(Cl)cc(Cl)cc1Cl)C(O)=O |r,wU:7.8,4.4,wD:1.0,(-2.28,-28.91,;-2.28,-30.45,;-3.61,-31.23,;-3.61,-32.77,;-2.28,-33.53,;-.94,-32.75,;-.95,-31.22,;-2.27,-35.07,;-3.6,-35.84,;-3.59,-37.38,;-2.25,-38.15,;-4.92,-38.16,;-6.26,-37.4,;-7.58,-38.17,;-8.92,-37.41,;-10.25,-38.18,;-10.25,-39.73,;-8.91,-40.5,;-7.58,-39.72,;-6.25,-40.48,;-4.91,-39.71,;-3.57,-40.48,;-3.57,-42.02,;-4.9,-42.79,;-2.23,-42.78,;-2.23,-44.32,;-3.56,-45.09,;-4.89,-44.32,;-3.55,-46.63,;-2.21,-47.4,;-2.21,-48.94,;-.88,-46.61,;-.89,-45.08,;.44,-44.3,;-.93,-35.83,;-.92,-37.37,;.4,-35.05,)|