Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 2C8
Ligand
BDBM50247629
Substrate
n/a
Meas. Tech.
ChEMBL_567198 (CHEMBL1029110)
IC50
9800±n/a nM
Citation
 O'Neill, PM; Shone, AE; Stanford, D; Nixon, G; Asadollahy, E; Park, BK; Maggs, JL; Roberts, P; Stocks, PA; Biagini, G; Bray, PG; Davies, J; Berry, N; Hall, C; Rimmer, K; Winstanley, PA; Hindley, S; Bambal, RB; Davis, CB; Bates, M; Gresham, SL; Brigandi, RA; Gomez-de-Las-Heras, FM; Gargallo, DV; Parapini, S; Vivas, L; Lander, H; Taramelli, D; Ward, SA Synthesis, antimalarial activity, and preclinical pharmacology of a novel series of 4'-fluoro and 4'-chloro analogues of amodiaquine. Identification of a suitable"back-up" compound for N-tert-butyl isoquine. J Med Chem 52:1828-44 (2009) [PubMed] Article
O'Neill, PM; Shone, AE; Stanford, D; Nixon, G; Asadollahy, E; Park, BK; Maggs, JL; Roberts, P; Stocks, PA; Biagini, G; Bray, PG; Davies, J; Berry, N; Hall, C; Rimmer, K; Winstanley, PA; Hindley, S; Bambal, RB; Davis, CB; Bates, M; Gresham, SL; Brigandi, RA; Gomez-de-Las-Heras, FM; Gargallo, DV; Parapini, S; Vivas, L; Lander, H; Taramelli, D; Ward, SA Synthesis, antimalarial activity, and preclinical pharmacology of a novel series of 4'-fluoro and 4'-chloro analogues of amodiaquine. Identification of a suitable"back-up" compound for N-tert-butyl isoquine. J Med Chem 52:1828-44 (2009) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 2C8
Synonyms:
CP2C8_HUMAN | CYP2C8 | CYPIIC8 | Cytochrome P450 2C8 (CYP2C8) | P450 IIC2 | P450 MP-12/MP-20 | P450 form 1 | S-mephenytoin 4-hydroxylase
Type:
Protein
Mol. Mass.:
55839.23
Organism:
Homo sapiens (Human)
Description:
P10632
Residue:
490
Sequence:
MEPFVVLVLCLSFMLLFSLWRQSCRRRKLPPGPTPLPIIGNMLQIDVKDICKSFTNFSKVYGPVFTVYFGMNPIVVFHGYEAVKEALIDNGEEFSGRGNSPISQRITKGLGIISSNGKRWKEIRRFSLTTLRNFGMGKRSIEDRVQEEAHCLVEELRKTKASPCDPTFILGCAPCNVICSVVFQKRFDYKDQNFLTLMKRFNENFRILNSPWIQVCNNFPLLIDCFPGTHNKVLKNVALTRSYIREKVKEHQASLDVNNPRDFIDCFLIKMEQEKDNQKSEFNIENLVGTVADLFVAGTETTSTTLRYGLLLLLKHPEVTAKVQEEIDHVIGRHRSPCMQDRSHMPYTDAVVHEIQRYSDLVPTGVPHAVTTDTKFRNYLIPKGTTIMALLTSVLHDDKEFPNPNIFDPGHFLDKNGNFKKSDYFMPFSAGKRICAGEGLARMELFLFLTTILQNFNLKSVDDLKNLNTTAVTKGIVSLPPSYQICFIPV
Inhibitor
Name:
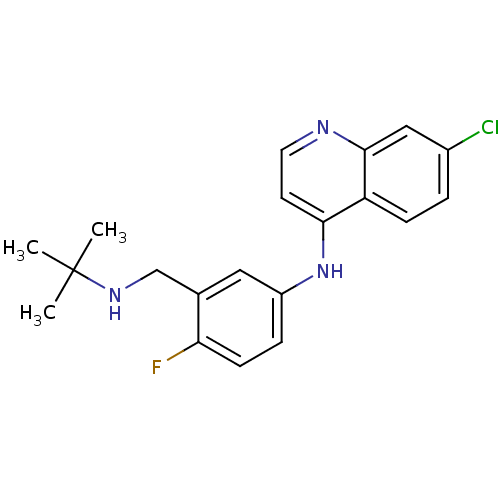
BDBM50247629
Synonyms:
CHEMBL453384 | N-(3-((tert-butylamino)methyl)-4-fluorophenyl)-7-chloroquinolin-4-amine | TCMDC-135909 | [3-(tert-Butylaminomethyl)-4-fluorophenyl](7-chloro-quinolin-4-yl)amine
Type:
Small organic molecule
Emp. Form.:
C20H21ClFN3
Mol. Mass.:
357.852
SMILES:
CC(C)(C)NCc1cc(Nc2ccnc3cc(Cl)ccc23)ccc1F