Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 3A4
Ligand
BDBM50343357
Substrate
n/a
Meas. Tech.
ChEMBL_746947 (CHEMBL1777313)
IC50
800±n/a nM
Citation
 Zehnder, L; Bennett, M; Meng, J; Huang, B; Ninkovic, S; Wang, F; Braganza, J; Tatlock, J; Jewell, T; Zhou, JZ; Burke, B; Wang, J; Maegley, K; Mehta, PP; Yin, MJ; Gajiwala, KS; Hickey, MJ; Yamazaki, S; Smith, E; Kang, P; Sistla, A; Dovalsantos, E; Gehring, MR; Kania, R; Wythes, M; Kung, PP Optimization of potent, selective, and orally bioavailable pyrrolodinopyrimidine-containing inhibitors of heat shock protein 90. Identification of development candidate 2-amino-4-{4-chloro-2-[2-(4-fluoro-1H-pyrazol-1-yl)ethoxy]-6-methylphenyl}-N-(2,2-difluoropropyl)-5,7-dihydro-6H-pyrrolo[3,4-d]pyr J Med Chem 54:3368-85 (2011) [PubMed] Article
Zehnder, L; Bennett, M; Meng, J; Huang, B; Ninkovic, S; Wang, F; Braganza, J; Tatlock, J; Jewell, T; Zhou, JZ; Burke, B; Wang, J; Maegley, K; Mehta, PP; Yin, MJ; Gajiwala, KS; Hickey, MJ; Yamazaki, S; Smith, E; Kang, P; Sistla, A; Dovalsantos, E; Gehring, MR; Kania, R; Wythes, M; Kung, PP Optimization of potent, selective, and orally bioavailable pyrrolodinopyrimidine-containing inhibitors of heat shock protein 90. Identification of development candidate 2-amino-4-{4-chloro-2-[2-(4-fluoro-1H-pyrazol-1-yl)ethoxy]-6-methylphenyl}-N-(2,2-difluoropropyl)-5,7-dihydro-6H-pyrrolo[3,4-d]pyr J Med Chem 54:3368-85 (2011) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 3A4
Synonyms:
Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase
Type:
Enzyme
Mol. Mass.:
57349.57
Organism:
Homo sapiens (Human)
Description:
n/a
Residue:
503
Sequence:
MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMFDMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISIAEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYSMDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICVFPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSIIFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVVNETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFSKKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLGGLLQPEKPVVLKVESRDGTVSGA
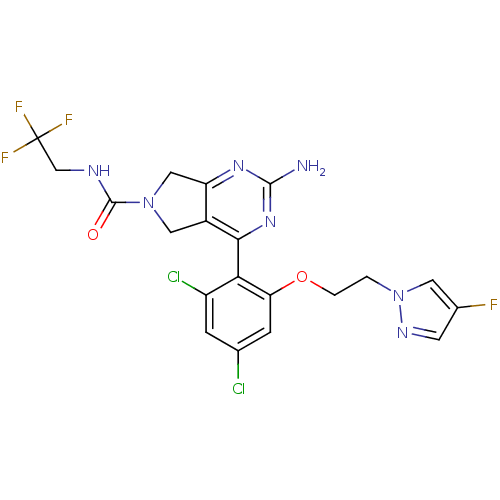
Inhibitor
Name:
BDBM50343357
Synonyms:
2-Amino-4-{2,4-dichloro-6-[2-(4-fluoropyrazol-1-yl)ethoxy]phenyl}-5,7-dihydropyrrolo[3,4-d]pyrimidine-6-carboxylic Acid(2,2,2-Trifluoroethyl)amide | CHEMBL1774930
Type:
Small organic molecule
Emp. Form.:
C20H17Cl2F4N7O2
Mol. Mass.:
534.294
SMILES:
Nc1nc2CN(Cc2c(n1)-c1c(Cl)cc(Cl)cc1OCCn1cc(F)cn1)C(=O)NCC(F)(F)F |(-.23,-41.95,;1.11,-41.19,;1.11,-39.64,;2.44,-38.87,;2.75,-37.37,;4.28,-37.2,;4.91,-38.6,;3.77,-39.63,;3.78,-41.18,;2.44,-41.96,;5.1,-41.95,;5.1,-43.49,;3.77,-44.26,;6.44,-44.26,;7.77,-43.49,;9.11,-44.26,;7.76,-41.94,;6.43,-41.18,;6.42,-39.64,;7.75,-38.86,;9.09,-39.63,;10.42,-38.85,;11.83,-39.47,;12.85,-38.32,;14.39,-38.47,;12.08,-36.99,;10.57,-37.32,;5.04,-35.86,;4.27,-34.53,;6.58,-35.86,;7.35,-34.52,;8.89,-34.51,;9.65,-33.18,;9.66,-35.84,;10.42,-34.51,)|