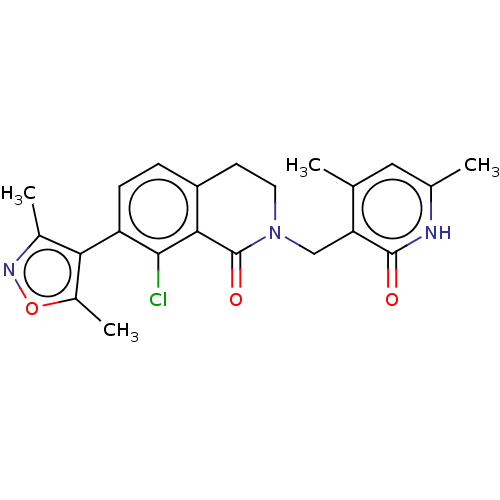
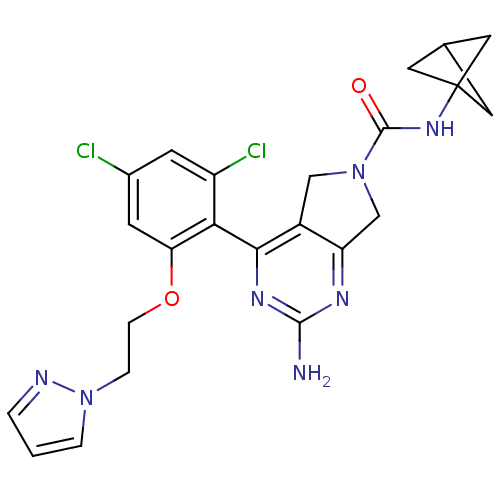
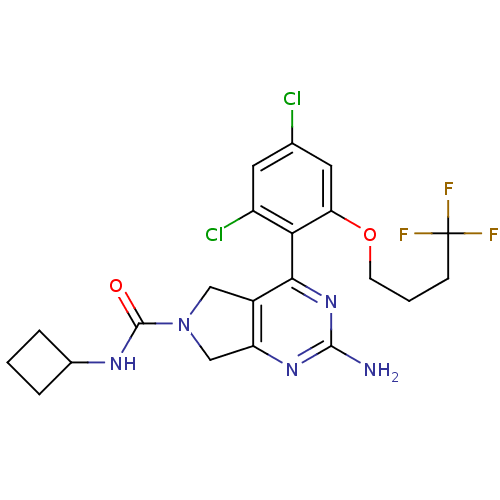
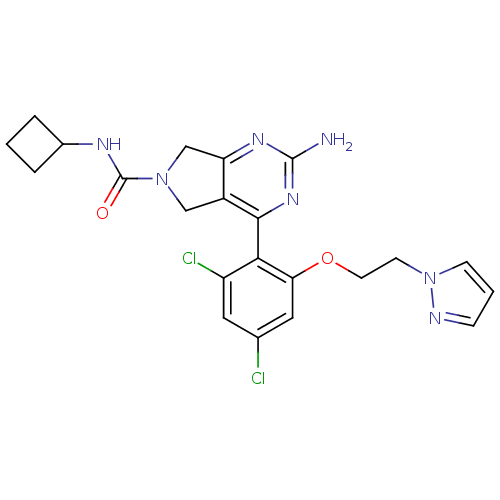
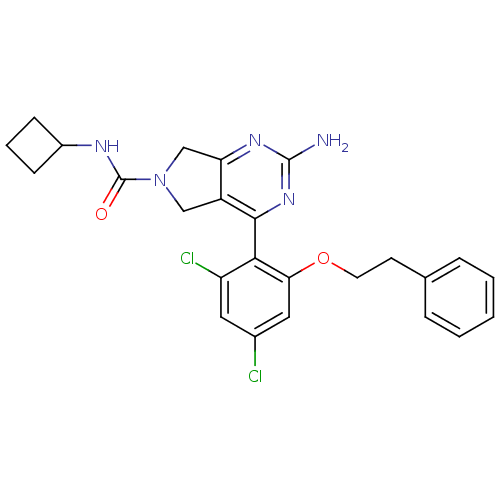
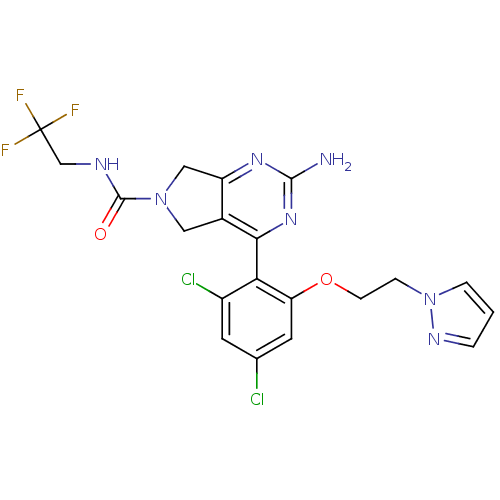
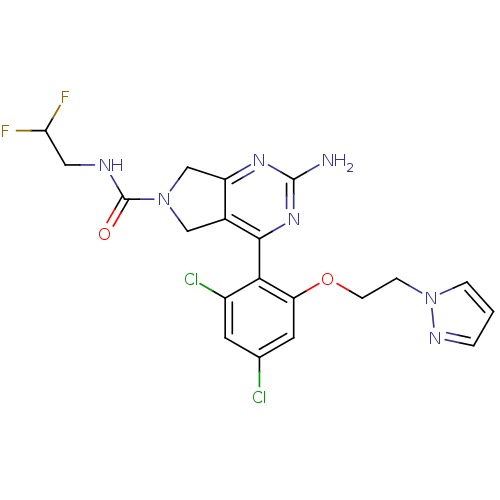
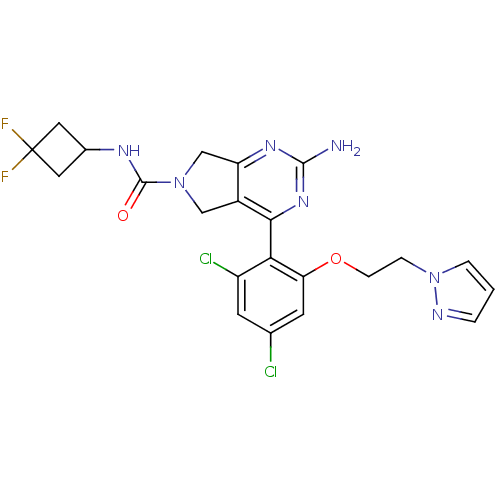
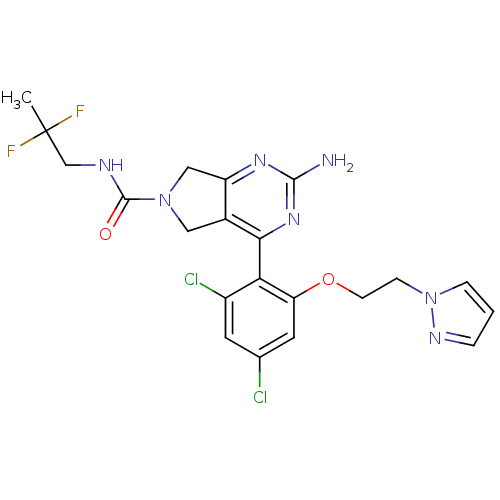
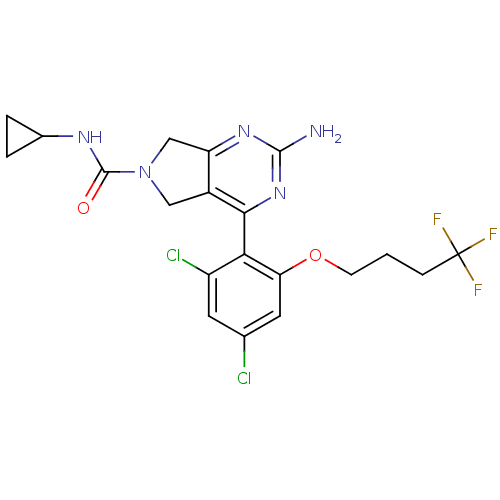
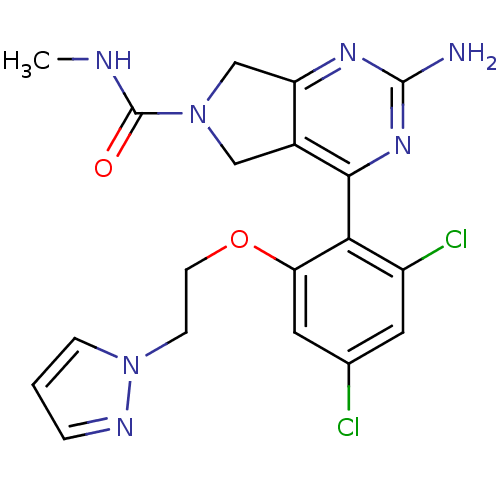
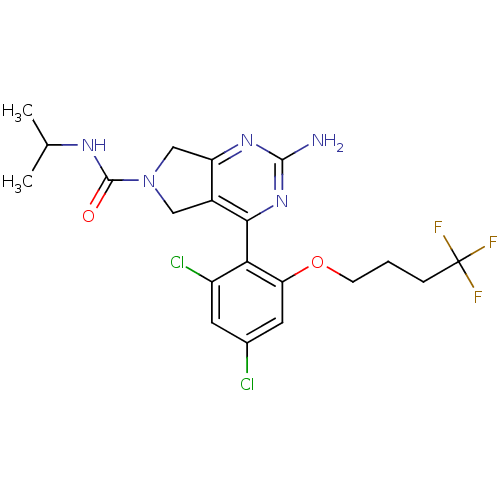
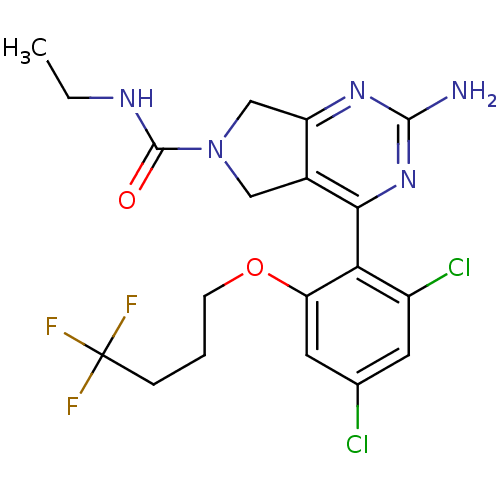
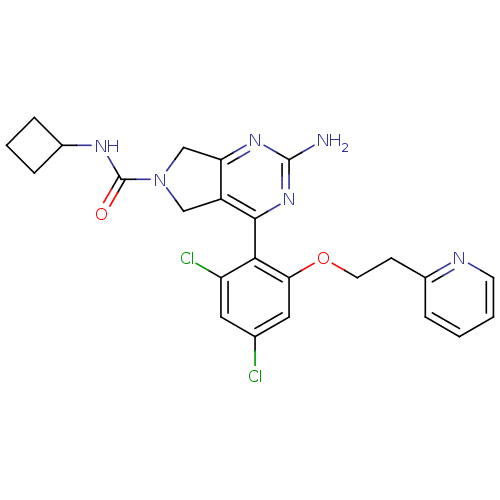
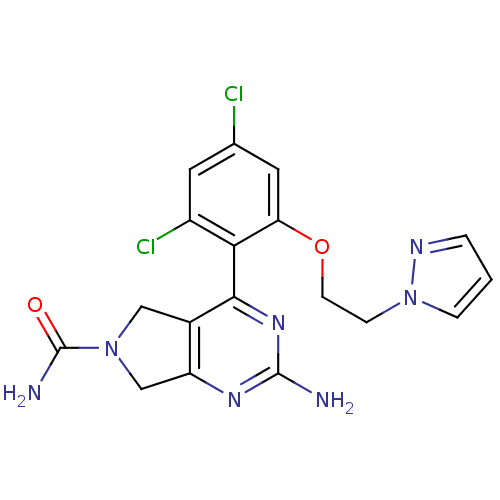
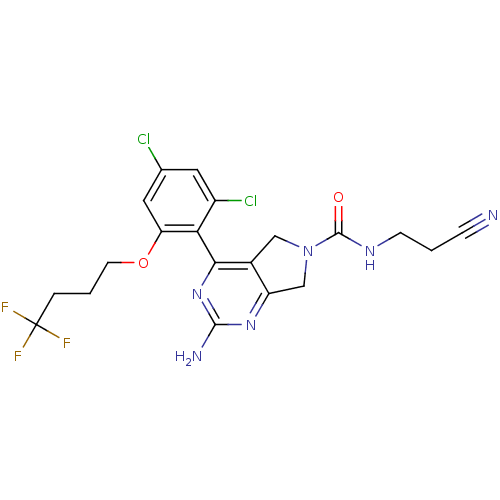
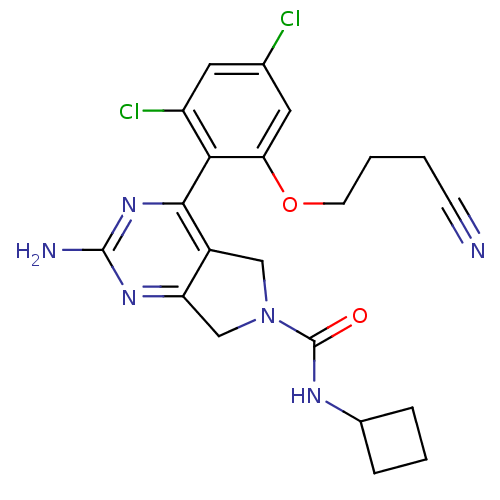
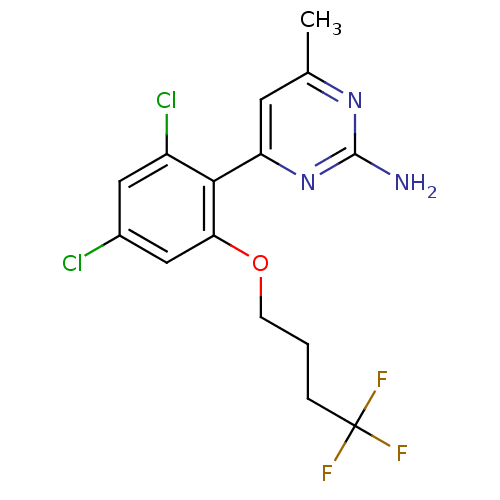
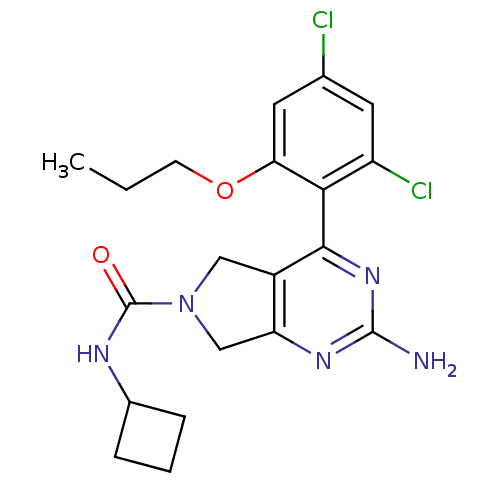
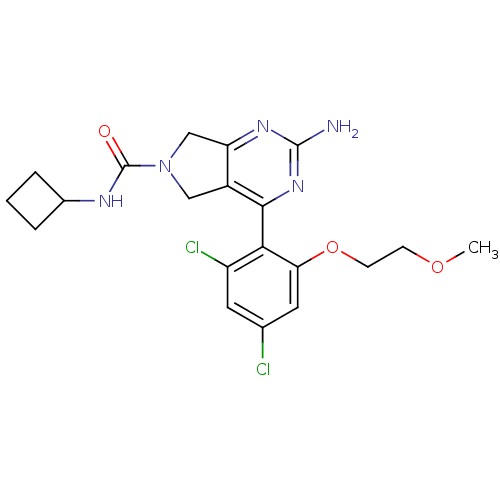
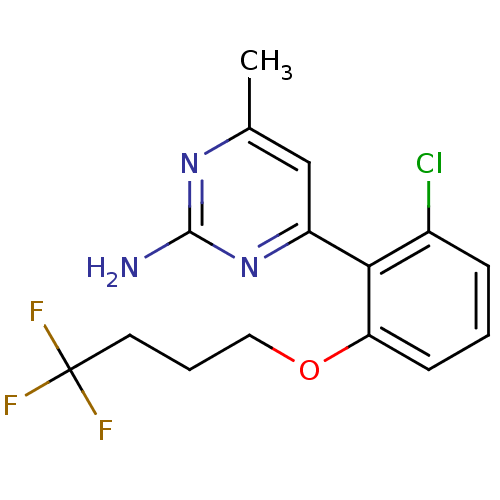
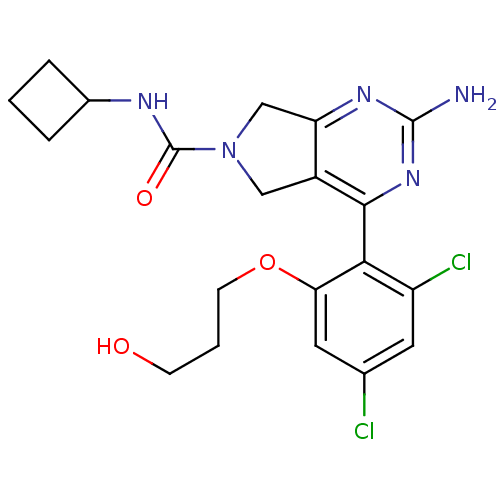
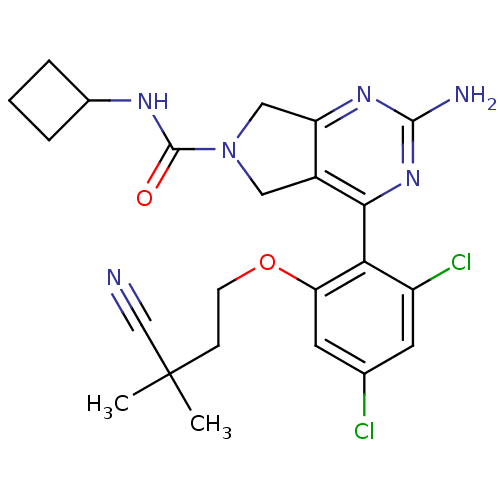
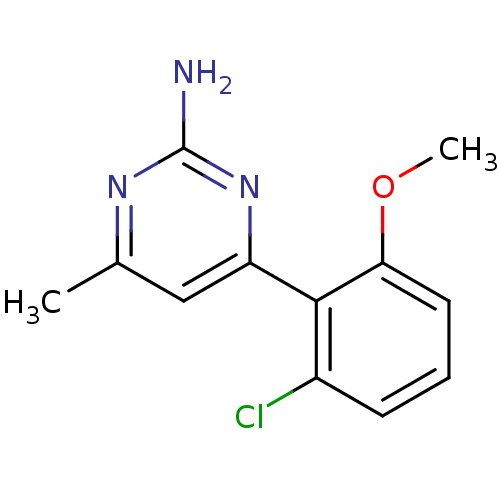
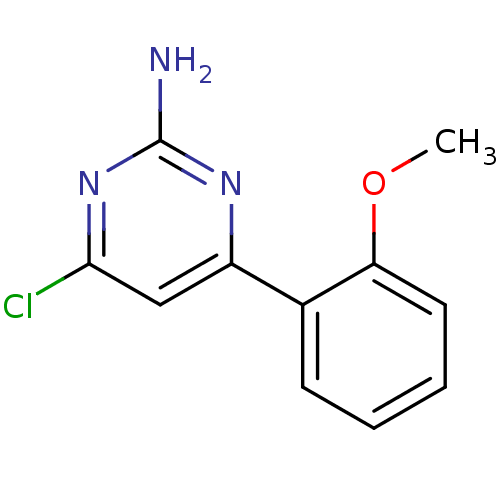
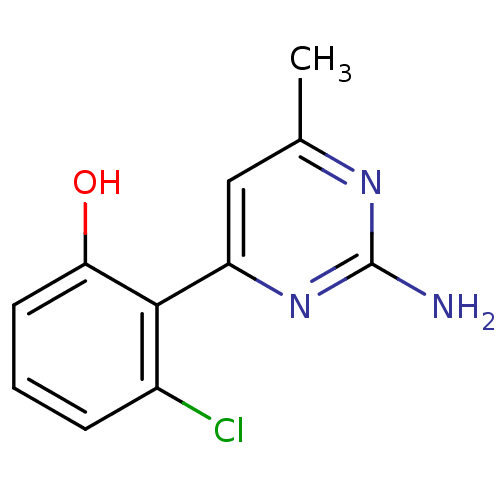
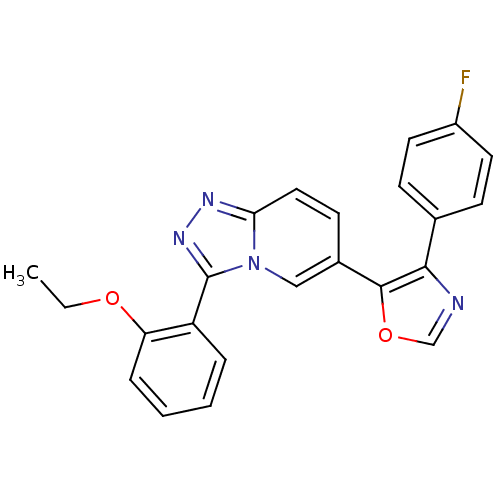
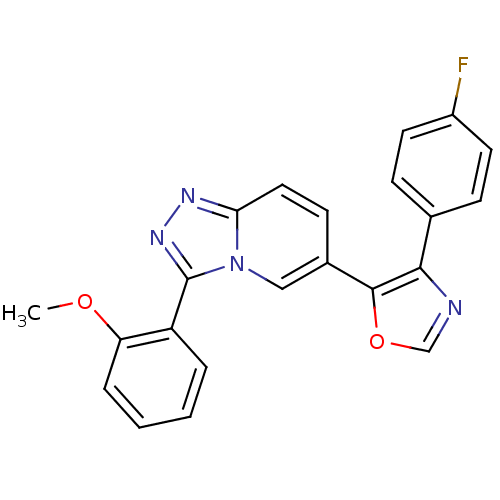
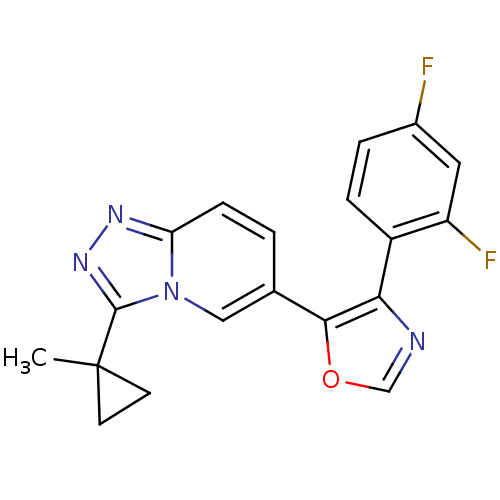
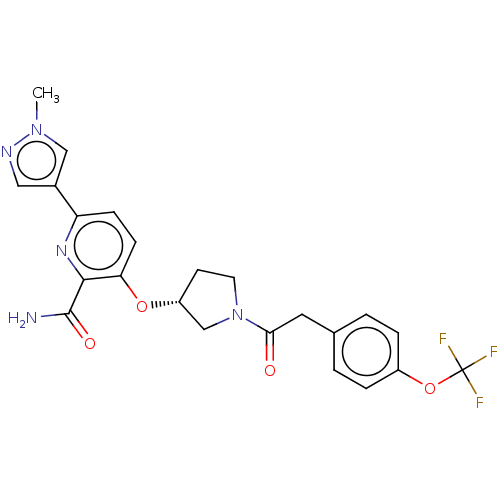
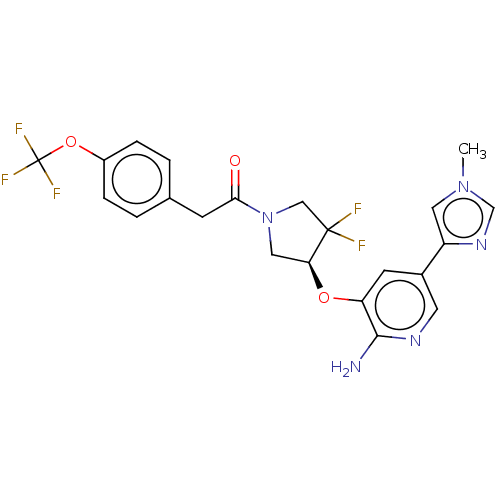
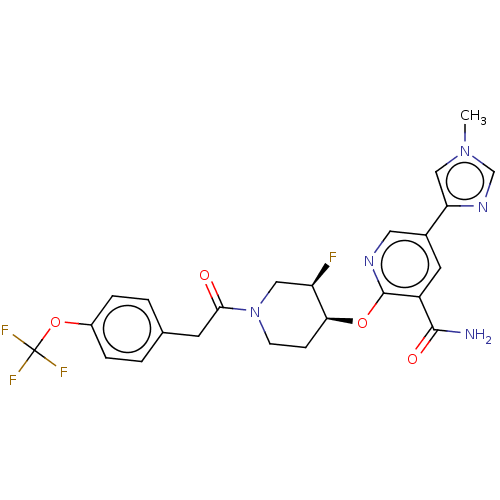
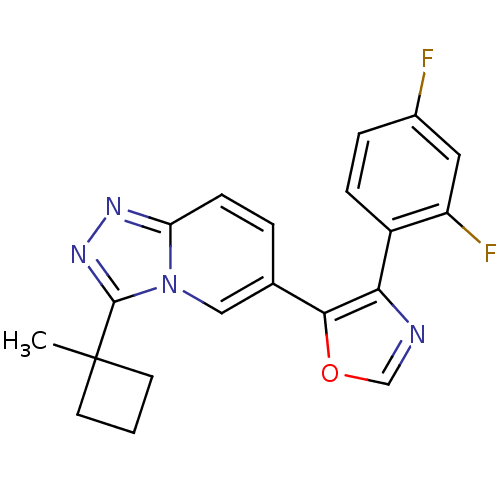
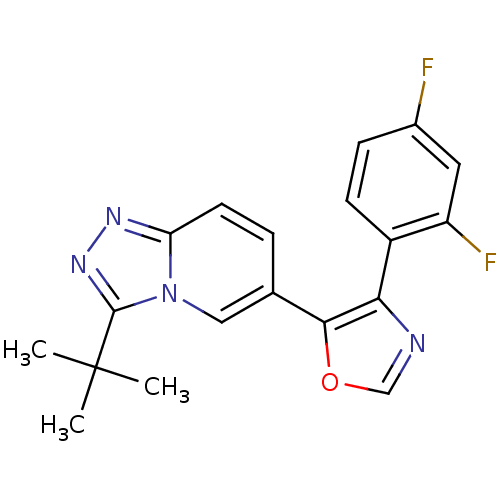
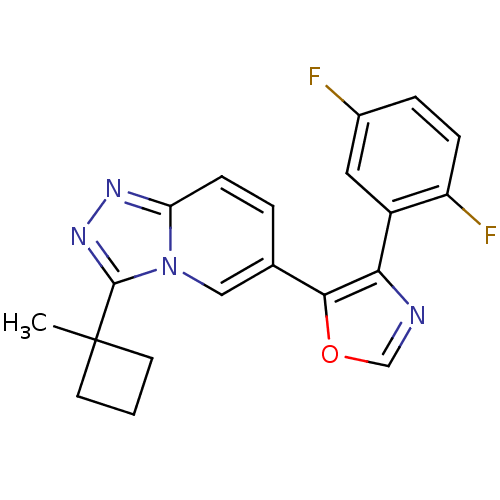
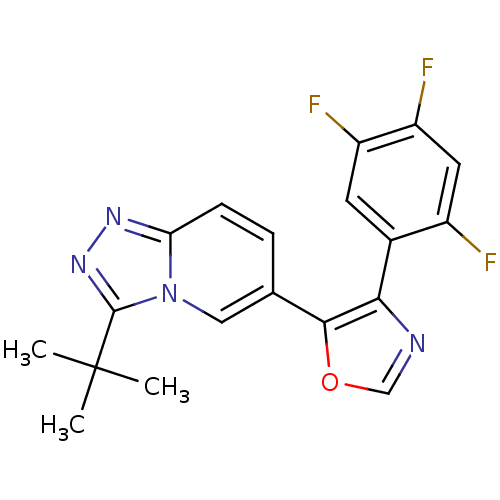
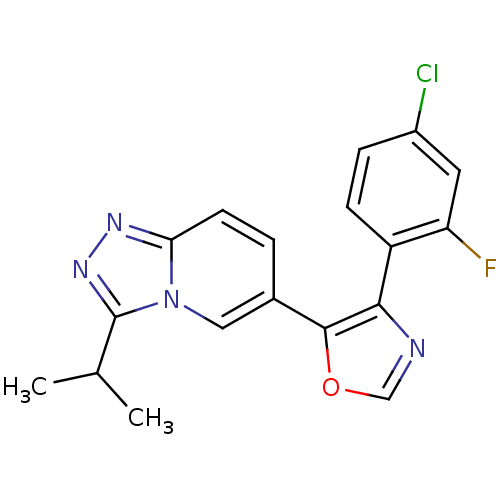
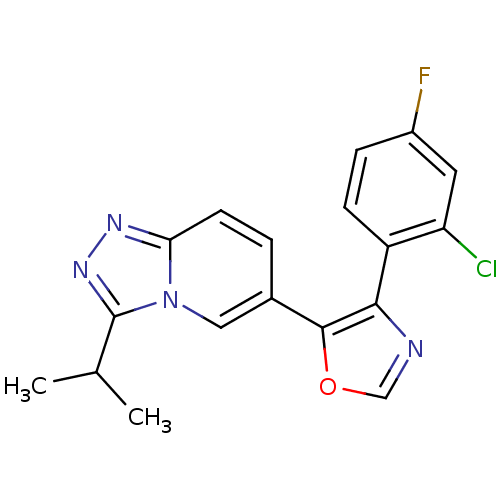
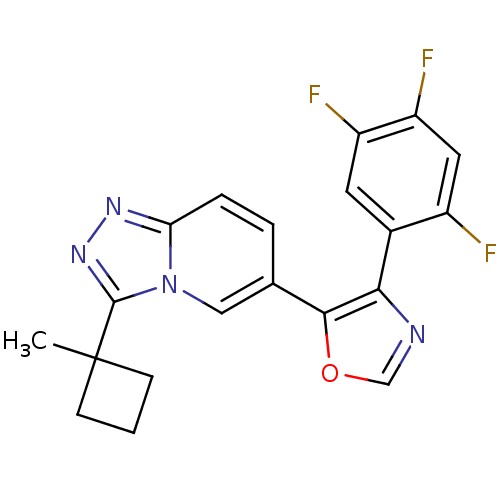
Affinity DataKi: 0.700nMAssay Description:Inhibition of wild-type EZH2 (unknown origin) expressed in baculovirus infected SF9 cells co-expressing SUZ12/EED/RbAp48 complex using HeLa cells der...More data for this Ligand-Target Pair
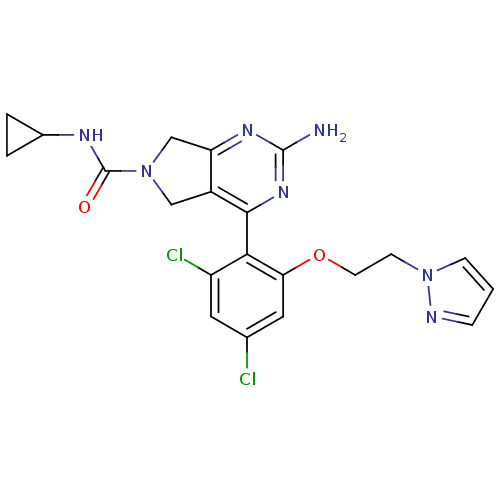
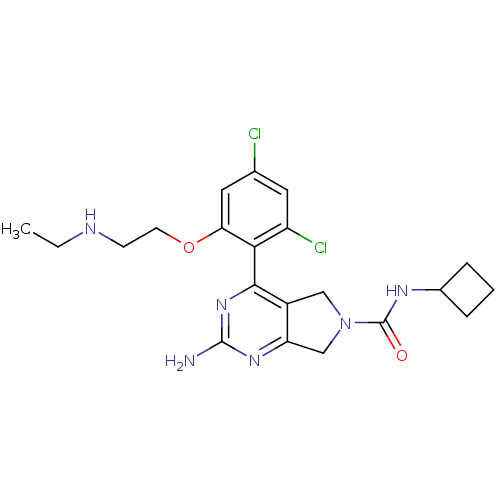
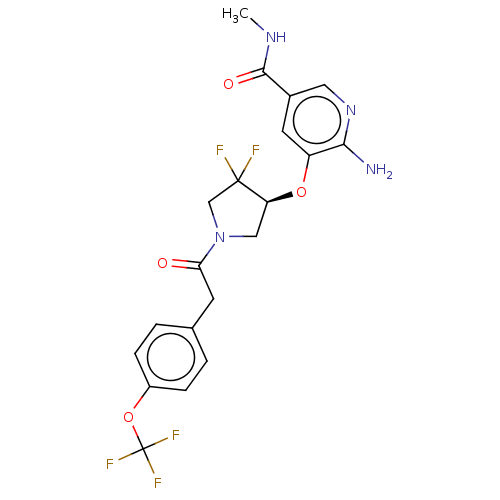
Affinity DataKi: 2nMAssay Description:Inhibition of wild-type EZH2 (unknown origin) expressed in baculovirus infected SF9 cells co-expressing SUZ12/EED/RbAp48 complex using HeLa cells der...More data for this Ligand-Target Pair
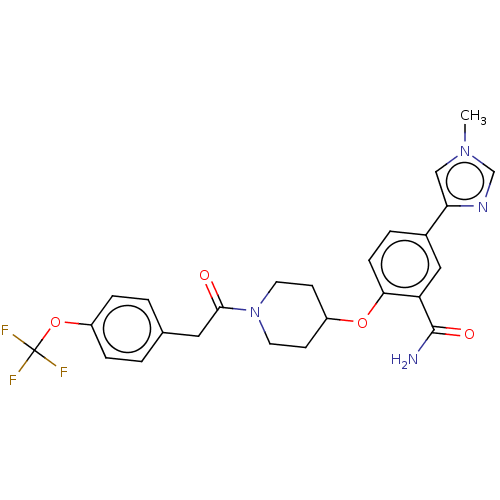
Affinity DataKi: 6nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
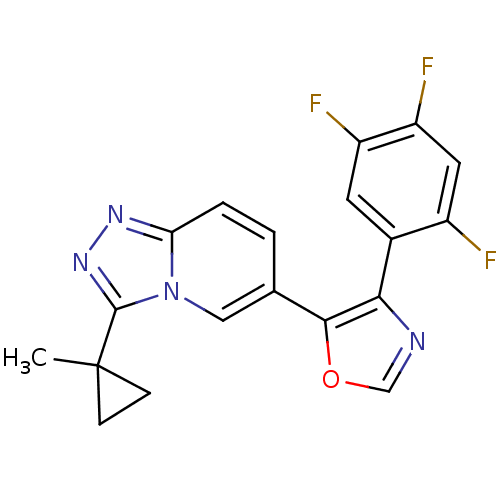
Affinity DataKi: 7nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 7nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 7nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 7nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 9nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 9nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 12nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 16nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 18nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 20nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 21nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 26nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 27nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 35nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 38nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 40nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 50nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 110nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 120nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 130nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 190nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 250nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 300nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 700nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 1.20E+3nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 1.40E+3nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 8.30E+3nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: 1.16E+4nMAssay Description:Displacement of [3H]dofetilide from human ERG channel expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 0.400nMAssay Description:The IC50s were determined by incubating each test compound with activated p38-alpha in a 96-well plate that had been pre-treated with ATF-2-GST to al...More data for this Ligand-Target Pair
Affinity DataIC50: 0.400nMAssay Description:The IC50s were determined by incubating each test compound with activated p38-alpha in a 96-well plate that had been pre-treated with ATF-2-GST to al...More data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:The IC50s were determined by incubating each test compound with activated p38-alpha in a 96-well plate that had been pre-treated with ATF-2-GST to al...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Antagonist activity at prolink-tagged TrkC in human U2OS cells assessed as inhibition of NT3-induced receptor phosphorylation by measuring reduction ...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Antagonist activity at prolink-tagged TrkA in human U2OS cells assessed as inhibition of beta-NGF-induced receptor phosphorylation by measuring reduc...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMAssay Description:Antagonist activity at prolink-tagged TrkC in human U2OS cells assessed as inhibition of NT3-induced receptor phosphorylation by measuring reduction ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMAssay Description:The IC50s were determined by incubating each test compound with activated p38-alpha in a 96-well plate that had been pre-treated with ATF-2-GST to al...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMAssay Description:The IC50s were determined by incubating each test compound with activated p38-alpha in a 96-well plate that had been pre-treated with ATF-2-GST to al...More data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:The IC50s were determined by incubating each test compound with activated p38-alpha in a 96-well plate that had been pre-treated with ATF-2-GST to al...More data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMAssay Description:The IC50s were determined by incubating each test compound with activated p38-alpha in a 96-well plate that had been pre-treated with ATF-2-GST to al...More data for this Ligand-Target Pair
Affinity DataIC50: 2.20nMpH: 7.2 T: 2°CAssay Description:The IC50s were determined by incubating each test compound with activated p38-alpha in a 96-well plate that had been pre-treated with ATF-2-GST to al...More data for this Ligand-Target Pair
Affinity DataIC50: 2.20nMAssay Description:Antagonist activity at prolink-tagged TrkC in human U2OS cells assessed as inhibition of NT3-induced receptor phosphorylation by measuring reduction ...More data for this Ligand-Target Pair
Affinity DataIC50: 2.30nMAssay Description:Antagonist activity at prolink-tagged TrkC in human U2OS cells assessed as inhibition of NT3-induced receptor phosphorylation by measuring reduction ...More data for this Ligand-Target Pair
Affinity DataIC50: 2.30nMAssay Description:The IC50s were determined by incubating each test compound with activated p38-alpha in a 96-well plate that had been pre-treated with ATF-2-GST to al...More data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMAssay Description:The IC50s were determined by incubating each test compound with activated p38-alpha in a 96-well plate that had been pre-treated with ATF-2-GST to al...More data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMAssay Description:The IC50s were determined by incubating each test compound with activated p38-alpha in a 96-well plate that had been pre-treated with ATF-2-GST to al...More data for this Ligand-Target Pair
Affinity DataIC50: 2.60nMAssay Description:Antagonist activity at prolink-tagged TrkB in human U2OS cells assessed as inhibition of BDNF-induced receptor phosphorylation by measuring reduction...More data for this Ligand-Target Pair

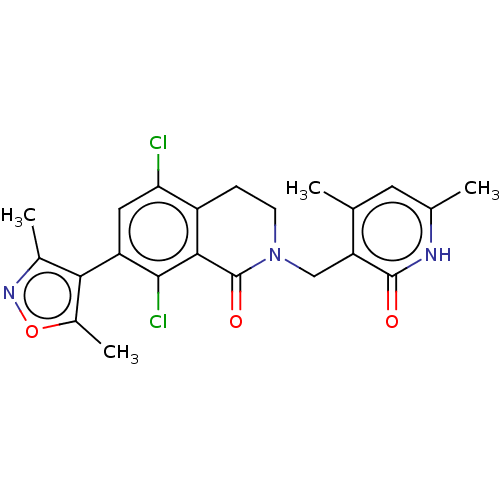
 3D Structure (crystal)
3D Structure (crystal)