Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Sodium/glucose cotransporter 2
Ligand
BDBM20875
Substrate
n/a
Meas. Tech.
ChEMBL_856134 (CHEMBL2162522)
IC50
38±n/a nM
Citation
 Fushimi, N; Fujikura, H; Shiohara, H; Teranishi, H; Shimizu, K; Yonekubo, S; Ohno, K; Miyagi, T; Itoh, F; Shibazaki, T; Tomae, M; Ishikawa-Takemura, Y; Nakabayashi, T; Kamada, N; Ozawa, T; Kobayashi, S; Isaji, M Structure-activity relationship studies of 4-benzyl-1H-pyrazol-3-ylß-d-glucopyranoside derivatives as potent and selective sodium glucose co-transporter 1 (SGLT1) inhibitors with therapeutic activity on postprandial hyperglycemia. Bioorg Med Chem 20:6598-612 (2012) [PubMed] Article
Fushimi, N; Fujikura, H; Shiohara, H; Teranishi, H; Shimizu, K; Yonekubo, S; Ohno, K; Miyagi, T; Itoh, F; Shibazaki, T; Tomae, M; Ishikawa-Takemura, Y; Nakabayashi, T; Kamada, N; Ozawa, T; Kobayashi, S; Isaji, M Structure-activity relationship studies of 4-benzyl-1H-pyrazol-3-ylß-d-glucopyranoside derivatives as potent and selective sodium glucose co-transporter 1 (SGLT1) inhibitors with therapeutic activity on postprandial hyperglycemia. Bioorg Med Chem 20:6598-612 (2012) [PubMed] Article More Info.:
Target
Name:
Sodium/glucose cotransporter 2
Synonyms:
Na(+)/glucose cotransporter 2 | SC5A2_HUMAN | SGLT2 | SLC5A2 | Sodium-Dependent Glucose Cotransporter 2 (SGLT2) | Sodium/glucose cotransporter 1 (SGLT1) | Solute carrier family 5 member 2
Type:
Protein
Mol. Mass.:
72902.00
Organism:
Homo sapiens (Human)
Description:
P31639
Residue:
672
Sequence:
MEEHTEAGSAPEMGAQKALIDNPADILVIAAYFLLVIGVGLWSMCRTNRGTVGGYFLAGRSMVWWPVGASLFASNIGSGHFVGLAGTGAASGLAVAGFEWNALFVVLLLGWLFAPVYLTAGVITMPQYLRKRFGGRRIRLYLSVLSLFLYIFTKISVDMFSGAVFIQQALGWNIYASVIALLGITMIYTVTGGLAALMYTDTVQTFVILGGACILMGYAFHEVGGYSGLFDKYLGAATSLTVSEDPAVGNISSFCYRPRPDSYHLLRHPVTGDLPWPALLLGLTIVSGWYWCSDQVIVQRCLAGKSLTHIKAGCILCGYLKLTPMFLMVMPGMISRILYPDEVACVVPEVCRRVCGTEVGCSNIAYPRLVVKLMPNGLRGLMLAVMLAALMSSLASIFNSSSTLFTMDIYTRLRPRAGDRELLLVGRLWVVFIVVVSVAWLPVVQAAQGGQLFDYIQAVSSYLAPPVSAVFVLALFVPRVNEQGAFWGLIGGLLMGLARLIPEFSFGSGSCVQPSACPAFLCGVHYLYFAIVLFFCSGLLTLTVSLCTAPIPRKHLHRLVFSLRHSKEEREDLDADEQQGSSLPVQNGCPESAMEMNEPQAPAPSLFRQCLLWFCGMSRGGVGSPPPLTQEEAAAAARRLEDISEDPSWARVVNLNALLMMAVAVFLWGFYA
Inhibitor
Name:
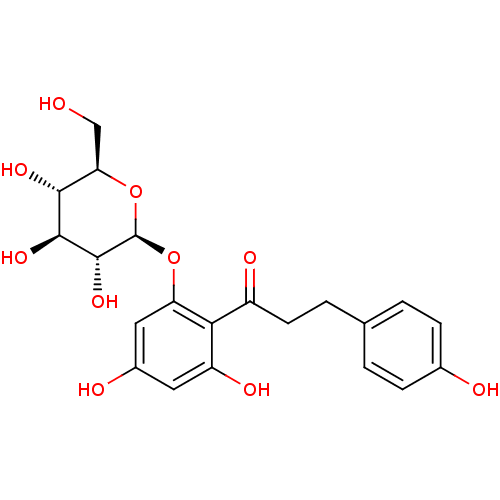
BDBM20875
Synonyms:
1-(2,4-dihydroxy-6-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}phenyl)-3-(4-hydroxyphenyl)propan-1-one | CHEMBL241384 | CHEMBL245067 | JMC511145 Compound 1 | O-glucoside, 1 | Phlorizin
Type:
Natural product
Emp. Form.:
C21H24O10
Mol. Mass.:
436.4093
SMILES:
OC[C@H]1O[C@@H](Oc2cc(O)cc(O)c2C(=O)CCc2ccc(O)cc2)[C@H](O)[C@@H](O)[C@@H]1O